Le Infezioni in Medicina, n. 3, 303-310, 2025
doi: 10.53854/liim-3303-7
ORIGINAL ARTICLES
Validation of a clinical scoring system to differentiate mycobacterial and bacterial causes of spondylodiscitis: a retrospective cohort study from Kerala, India
Vettakkara Kandy Muhammed Niyas1, Rajalakshmi Ananthanarayanan1, Mohamed Puthiyaveettil Zunimol2, Dheeraj Mohan3, Sundeep Malla4, Steven Van Den Broucke5, Erika Vlieghe5,6, Emmanuel Bottieau5, Nitin Gupta5-7
1Department of Infectious Diseases, KIMSHEALTH, Thiruvananthapuram, Kerala, India;
2Department of Radiology, KIMSHEALTH, Thiruvananthapuram, Kerala, India;
3Department of Family Medicine, KIMSHEALTH, Thiruvananthapuram, Kerala, India;
4Department of Radiodiagnosis, Amrita Institute of Medical Sciences, Faridabad, India;
5Department of Clinical Sciences, Institute of Tropical Medicine, Antwerp, Belgium;
6University of Antwerp, Antwerp, Belgium;
7Department of Infectious Diseases, Kasturba Medical College, Manipal, Manipal Academy of Higher Education, Manipal, India.
Article received 4 June 2025 and accepted 24 July 2025
Corresponding author
Nitin Gupta
E-mail: nityn.gupta@manipal.edu
SummaRY
Background: Mycobacterial and bacterial spondylodiscitis require distinct therapeutic approaches. However, microbiological diagnosis is often delayed or unavailable in resource-limited settings. This study aimed to validate a clinical scoring system developed in a prior retrospective cohort study in Karnataka (India) to differentiate these aetiologies.
Methods: We conducted a retrospective cohort study of patients with microbiologically confirmed spondylodiscitis admitted between January 2017 and August 2024 in a tertiary care centre in Kerala, India. Demographic, clinical, laboratory, imaging, and treatment data were compared between the two groups, and the scoring system’s diagnostic performance was assessed.
Results: Of 151 patients with infectious spondylodiscitis, 73 patients with community-acquired microbiologically confirmed infection were included. Of these, 32 (44%) had mycobacterial, and 41 (56%) had bacterial spondylodiscitis [Staphylococcus aureus (37%), Escherichia coli (11%), Klebsiella pneumoniae (11%), Streptococcus spp (4.1%), Salmonella spp (1.4%)]. The scoring system demonstrated good diagnostic accuracy for bacterial aetiology (AUC: 0.78; 95% CI: 0.67–0.88). Mycobacterial cases were younger, had longer symptom duration, lower C-reactive protein and leukocyte counts, and more frequent vertebral height loss on imaging. The rate of clinical improvement was similar between groups.
Conclusions: The validated clinical scoring system reliably distinguishes mycobacterial from bacterial spondylodiscitis and may guide empirical management in settings where timely microbiological diagnosis is not feasible.
Keywords: spondylodiscitis, mycobacterial infection, bacterial infection, clinical scoring system.
INTRODUCTION
Infectious involvement of the spine is associated with significant long-term neurological morbidity if not diagnosed early [1]. In India, the most common cause of infectious spondylodiscitis is tuberculosis (TB), but recent reports indicate that bacterial infections are common as well [2]. The incidence of brucellosis, in particular, has been increasing in Asia [3, 4]. It is essential to differentiate between mycobacterial and bacterial causes as their treatments and prognosis are starkly different. Microbiological diagnosis often requires invasive procedures, and consequently, many patients are started on empirical therapies in resource-limited settings [5]. Incorrect empirical choice of drugs inevitably leads to poor outcomes and/or promotes resistance. In such a situation, the availability of validated scoring systems that can guide clinical decision-making will be invaluable. In a previous study from Karnataka, South India, a clinical scoring system was developed to differentiate mycobacterial from bacterial spondylitis [4]. The scoring system was originally developed in 85 patients with microbiologically confirmed spondylodiscitis (44 tuberculosis, 41 bacterial). Univariate analysis was used to identify clinical, laboratory, and histopathological parameters that significantly differed between tubercular and bacterial cases. Variables with high discriminatory ability were selected for inclusion in the scoring model (Table 1). Each variable was assigned a certain weightage of 1 point, an empirical choice aimed at maintaining simplicity and bedside applicability. At a cutoff score of ≥5, the model demonstrated an area under the receiver operating characteristic (ROC) curve of 0.93 (95% CI: 0.86–1.00), with a sensitivity of 75% and specificity of 91% for differentiating bacterial from tubercular spondylodiscitis. The authors concluded that the scoring system must be validated in another cohort. Our study’s primary objective is to validate this previously published scoring system, and the secondary objective is to compare the clinical profile, radiological profile, treatment and outcomes of patients with mycobacterial and bacterial spondylodiscitis.
Table 1 - A scoring system developed in the Karnataka cohort to differentiate mycobacterial and bacterial spondylodiscitis.

METHODOLOGY
This retrospective validation cohort study was conducted at a tertiary care hospital in Thiruvananthapuram, Kerala, India. This centre is located south of Manipal, Karnataka, where the scoring system had been previously developed. The records of all patients diagnosed with infectious spondylodiscitis admitted between January 2017 and August 2024 were screened. Those with a community-acquired infection with a microbiological confirmation were finally included. Patients with healthcare-associated infections were not included as they are less likely to be confused clinically with tubercular spondylodiscitis. Hospital-acquired spondylodiscitis was defined as a spinal infection in which symptoms developed ≥48 hours after hospital admission or were temporally and anatomically related to an inpatient spinal procedure (e.g., spinal surgery, epidural instrumentation) performed during the same admission. Patients with incomplete clinical or radiological investigations were also excluded.
The sample size of the validation study was calculated based on the primary objective of validating the clinical scoring system. Using a prevalence estimate of bacterial infections as the aetiology of infectious spondylodiscitis at 51% from a previous study in Kerala, with a precision of 15% and a 95% confidence level, a chance of missing data of 10%, the required sample size was determined to be 70 cases of spondylodiscitis [6]. The sensitivity (75%) and specificity (91%) of the original scoring system were used for the calculation [4].
The study defined mycobacterial infection as infectious spondylodiscitis, which was confirmed by either a positive cartridge-based molecular test or culture positivity for TB from tissue or pus samples obtained from the affected site. Bacterial infections were confirmed through monobacterial culture positivity in blood or tissue (or pus) samples for bacterial organisms (identified by the VITEK® 2 system) in patients with spondylodiscitis.
The clinical scoring system was evaluated for its ability to diagnose confirmed bacterial infections as a group. Data relevant to the scoring system, microbiological diagnoses, test details, and patient outcomes were collected and recorded in a predefined case record form. Magnetic Resonance Imaging (MRI) images of the included patients were independently reviewed by an experienced radiologist, blinded to the aetiological diagnosis. A predefined set of objective MRI findings was systematically documented for each case. The evaluation included the presence of paravertebral soft tissue enhancement, epidural abscesses, spinal deformity (kyphosis or scoliosis), posterior element involvement (pedicle or spinous process), and neurological complications such as nerve root or spinal cord compression. A reduction in vertebral body height greater than 25% was defined as a significant loss of height, while disc destruction was considered significant when more than 50% of the disc was involved.
Data Analysis
Continuous variables were summarised using mean ± standard deviation or median (interquartile range), as appropriate. Categorical variables were presented as frequencies and percentages. Demography, clinical features, MRI features and outcomes were compared between mycobacterial and bacterial infections using the independent samples t-test or Mann-Whitney U test for continuous variables and the chi-square or Fisher’s exact test for categorical variables. The diagnostic accuracy of the clinical scoring system was assessed using microbiological diagnosis as the gold standard. A receiver operating characteristic (ROC) curve was generated to evaluate the performance of the scoring system (including its components), and the area under the curve (AUC) was calculated to measure its overall diagnostic ability to identify the group of bacterial spondylodiscitis. Sensitivity and specificity were computed at different score thresholds. Sensitivity, specificity, positive and negative likelihood ratios of the individual components of the scoring system were also calculated. Statistical significance was evaluated at a 95% confidence level, and all analyses were performed using appropriate statistical software (IBM SPSS Statistics for Windows, Version 26.0, IBM Corp). A p-value of <0.05 was considered significant.
RESULTS
During the period considered for recruitment, there were 151 cases of suspected infectious spondylodiscitis. Out of these, 83 had a microbiologically proven infectious spondylodiscitis. Ten cases were excluded because they were hospital-acquired infections. A total of 73 patients with spondylodiscitis were included, comprising 41/73 (56.2%) culture-confirmed bacterial cases (Staphylococcus aureus - 27/73 [37.0%], Escherichia coli - 8/73 [11.0%], Klebsiella pneumoniae - 8/73 [11.0%], Streptococcus spp - 3/73 [4.1%], Salmonella spp – 1/73 [1.4%]) and 32/73 (43.8%) spondylodiscitis cases due to Mycobacterium tuberculosis. The mean age of patients was 54.8±19.11 years, and 45/73 (61.6%) were male. A total of 26/73 (35.6%) patients had diabetes mellitus (DM). The median duration of illness at presentation was 55 (IQR: 47–70) days for all participants. Thoracic involvement was present in 33/73 (45.2%) patients, and lumbar involvement was present in 43/73 (58.9%). At presentation, the mean total leukocyte count was 12.6±6.1/mm³, and the median CRP was 53 (IQR: 22.25–89.8) mg/dL. A total of 61/73 (83.6%) patients completed treatment and were declared cured, while 12/73 (16.4%) were lost to follow-up.
When the scoring system was applied to the study cohort, a cutoff>5 predicted pyogenic spondylodiscitis with a sensitivity of 63.4% and specificity of 75% (AUC 0.78; 95% CI: 0.67-0.88; p<0.001) (Figure 1). Increasing the threshold to ≥6 improved specificity to 94%, but sensitivity declined to 32%. Lowering the cutoff to ≥4 raised sensitivity to 85%, though specificity decreased to 53%. The individual performance of the components of the scoring system has been summarised in Figure 2 and Table 2. CRP (AUC 0.734, p=0.001) and duration of illness (AUC 0.706, p=0.003) were the strongest individual predictors of pyogenic spondylodiscitis.

Figure 1 - Receiver operating characteristic curve to predict bacterial spondylodiscitis.

Figure 2 - Individual ROC curves of the parameters used in the scoring system. Abbreviations: DOI- Duration of illness, concur bact- concurrent bacterial infection, PTB refers to pulmonary tuberculosis; DM denotes diabetes mellitus; Lumbar- lumbar vertebrae involvement, TLC stands for total leukocyte count; CRP is C-reactive protein; ROC indicates receiver operating characteristic.
Table 2 - Diagnostic accuracy of the individual parameters used in the scoring system to predict a diagnosis of bacterial spondylodiscitis.
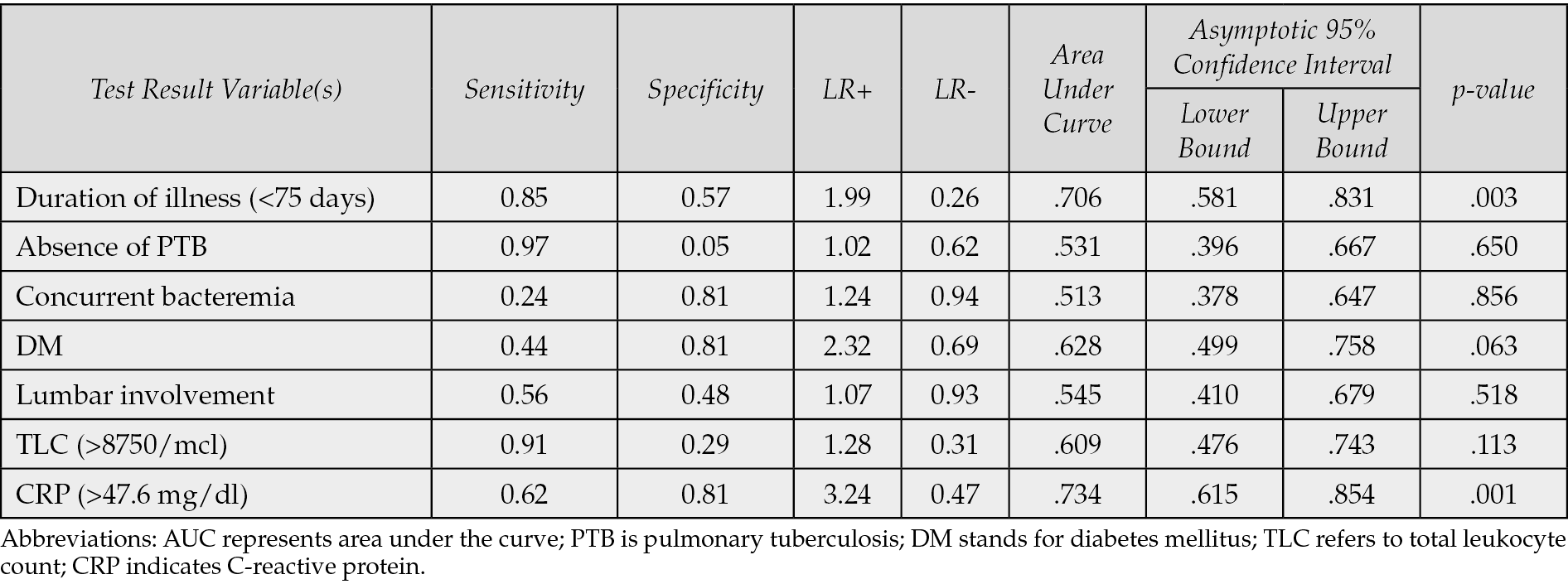
Significant differences between bacterial and tubercular spondylodiscitis were observed in diabetes mellitus (DM), age, duration of symptoms, TLC, and CRP levels (Table 3). Differences in sex distribution, pulmonary TB, concurrent bacteremia, and vertebral involvement were not statistically significant (Table 3). Among the 54 patients with MRI available, loss of vertebral height was the only feature that differed significantly between groups, being more common in mycobacterial spondylodiscitis (47.6% vs 9.1%; p=0.001). Disc destruction, paravertebral soft tissue involvement and epidural abscesses were also more common in mycobacterial infections, though not statistically significant. Posterior element involvement, spinal deformity, and neurological complications were seen across both groups, with no significant differences.
Table 3 - Comparison of Clinical, Laboratory, and Treatment Parameters in mycobacterial versus bacterial spondylodiscitis.

All patients with mycobacterial spondylodiscitis were treated with a combination of isoniazid, rifampicin, pyrazinamide and ethambutol. The patients with bacterial spondylodiscitis were treated with varying durations of intravenous followed by oral antibiotics. The duration of therapy until the last follow-up was significantly longer in the tubercular group (p<0.001). The treatment outcomes were not significantly different, and there were good outcomes in both groups (p=0.268).
DISCUSSION
In this retrospective cohort of 73 patients used to validate a previously developed scoring system, 56.2% had culture-confirmed bacterial spondylodiscitis, and 43.8% had a mycobacterial infection. Significant differences between the two groups were observed in age, duration of symptoms, TLC, CRP levels, and the prevalence of DM, as was observed in the derivation cohort. Mycobacterial spondylodiscitis was associated with a longer illness duration, lower inflammatory markers, and a higher frequency of vertebral height loss on MRI. The clinical scoring system demonstrated good diagnostic performance, with an area under the curve (AUC) of 0.78 (95% CI: 0.67-0.88). No previously published scoring system has been developed specifically to differentiate between bacterial and tubercular spondylodiscitis at initial presentation. Existing literature has focused primarily on imaging patterns or clinical series but lacks standardised, validated diagnostic tools for early stratification. Our scoring system aims to address this gap by combining key clinical, laboratory, and radiological features into a simple, point-based model. Regarding the optimal cutoff, while a threshold of ≥5 demonstrated the best balance between sensitivity (75%) and specificity (91%) in the derivation cohort, clinicians may prefer to prioritise sensitivity in acute settings. Given that bacterial spondylodiscitis can progress rapidly and carries a higher risk of bacteremia and systemic complications, an empiric treatment approach is often justified [7]. In such scenarios, using a lower cutoff (e.g., ≥4) may improve sensitivity and help identify high-risk patients who may benefit from prompt initiation of antibiotics, even in the absence of confirmatory microbiological results. This approach aligns with clinical reasoning, where empiric bacterial coverage is typically preferred over empiric anti-tubercular therapy due to the latter’s prolonged duration, toxicity, and monitoring requirements.
Although the original scoring system assigns somewhat equal weightage to each parameter, we recognise that individual variables differ in their diagnostic performance. To explore this further, we conducted a detailed analysis of the diagnostic accuracy of each component. Among the components, CRP >47.6 mg/dL and duration of illness <75 days emerged as the strongest individual predictors of bacterial spondylodiscitis, with AUCs of 0.734 and 0.706, respectively. Notably, CRP had a positive likelihood ratio of 3.24, indicating a moderate increase in the probability of bacterial infection when elevated. In contrast, other variables, such as the absence of pulmonary tuberculosis and lumbar involvement, showed lower AUCs and minimal diagnostic impact, suggesting limited standalone utility.
Although non-tubercular mycobacteria have been known to cause spondylodiscitis, all the cases of mycobacterial spondylodiscitis in this series were due to Mycobacterium tuberculosis [8, 9]. In our study, among the microbiologically confirmed cases of bacterial spondylodiscitis, Staphylococcus spp. was the most frequently isolated pathogen (65.8% of bacterial cases), followed by Escherichia coli and Klebsiella pneumoniae. This finding aligns with previous studies, where Staphylococcus aureus has consistently emerged as the predominant causative organism in hematogenous vertebral infections [10-13]. In some regions (including South Asia), gram-negative enteric bacilli, including E. coli and K. pneumoniae, contribute significantly to spondylodiscitis’s aetiological spectrum [11, 14]. Interestingly, although Brucella spp. was a significant pathogen in the derivation cohort of the scoring system (23/85, 27%), it was not identified in any patient within the current validation cohort [4]. This discrepancy likely reflects regional epidemiological differences, as our cohort was derived from a tertiary care centre serving a predominantly urban population with limited exposure to brucellosis risk factors. The derivation cohort served patients from rural backgrounds with a high risk of acquiring brucellosis. Despite this shift in aetiology, the scoring system maintained its discriminative performance, underscoring its robustness and applicability across diverse clinical settings.
Patients with tubercular spondylodiscitis were significantly younger and had a more prolonged duration of symptoms before diagnosis, consistent with the indolent and insidious nature of most mycobacterial infections. Notably, the duration of illness demonstrated a high AUC on ROC analysis. In contrast, similar to previous studies, bacterial cases tended to present acutely, with significantly higher inflammatory markers, including TLC and CRP [11]. Incidentally, a longer duration of illness at presentation in bacterial infections has been associated with poor outcomes in previous studies [12, 13]. DM was also significantly more prevalent among patients with bacterial infections, consistent with its known role as a risk factor [14]. Although male predominance was more frequently observed in bacterial infections, this difference was not statistically significant [12, 14].
In our study, thoracic involvement was seen in 47% of TB cases and was not significantly different from bacterial cases. In contrast, in the derivation cohort for the scoring system, thoracic vertebral involvement was observed in 59% of TB patients, which was significantly more common than in bacterial infections, consistent with the well-established thoracic predilection in spinal TB [4, 11]. Notably, the proportion of patients with concurrent pulmonary TB was also lower in the validation cohort (6% vs. 20%). This may have reduced the likelihood of thoracic seeding and contributed to the lack of statistical significance [4, 15]. It should be noted that the thoracic vertebrae predilection in mycobacterial infections is also associated with the presence of oxygen-rich red marrow that provides a favourable environment for mycobacterial survival [15]. In contrast, lumbar vertebrae are more prone to mechanical stress and microtrauma, predisposing them to bacterial seeding [15]. In the original derivation cohort, isolated lower lumbar vertebrae involvement was more common in bacterial infections. However, this was not observed in this study.
In our cohort, loss of vertebral height emerged as the only MRI feature significantly common in mycobacterial infections. This finding aligns with earlier reports, where vertebral collapse has been identified as a strong predictor of TB spondylitis [7, 16, 17]. The increased frequency of collapse in TB is likely due to anterior vertebral body involvement, which predisposes to kyphotic deformity, a recognised late complication of spinal TB [18]. In addition to vertebral collapse, involvement of multiple vertebral bodies, and anterior element destruction was identified to be more common in TB than in pyogenic spondylodiscitis in a recent study [7]. Although other findings like disc destruction, paravertebral soft tissue involvement, and epidural abscesses were also more frequent in TB, these did not reach statistical significance in our subgroup, likely because the study was underpowered for these comparisons. Nonetheless, disc destruction and large paravertebral abscesses have been commonly seen in TB cases [19, 20]. While posterior element involvement and spinal deformity were seen in both groups in our analysis, prior studies have identified these features as independent predictors of TB spondylodiscitis [17].
This study has several limitations. The data for the validation cohort, like those of the derivation cohort, were collected retrospectively from clinical records, introducing potential biases such as incomplete documentation and variability in diagnostic work-up. The absence of Brucella spp. in the validation cohort also precluded evaluating the scoring system’s performance in distinguishing brucellosis, a key aetiology in the original derivation cohort. As such, the applicability and diagnostic accuracy of the score in brucellosis-endemic settings remains uncertain and will require further external validation in regions where Brucella is a common cause of spondylodiscitis. MRI images were unavailable for all patients, and some patients were lost to follow-up before the outcomes could be assessed. The small sample size and single-centre design are also significant limitations.
Despite epidemiological differences between the derivation and validation cohorts, the scoring system accurately distinguished mycobacterial from bacterial spondylodiscitis. In resource-limited settings where timely microbiological confirmation may not be feasible, this tool could assist clinicians in initiating targeted empirical therapy, potentially improving patient outcomes and reducing inappropriate antibiotic use. Future studies should focus on external prospective multicentric validation, incorporating radiological data, and evaluating its impact on clinical decision-making and outcomes.
Conflict of interest
None declare.
Funding
None to declare.
References
[1] Rawall S, Hiatt LA, Rajaram SM, et al. Management of Pyogenic Spondylodiscitis in Adults. J Am Acad Orthop Surg. 2025. https://doi.org/10.5435/JAAOS-D-24-01088.
[2] Aithala JP, Attar A, Imthiaz AKA, et al. Is there a change in trend towards pyogenous spondylodiscitis compared to tubercular spondylodiscitis in India-A study of percutaneous biopsy evaluation in spondylodiscitis. Indian J Tuberc. 2020; 67: 509-514. https://doi.org/10.1016/j.ijtb.2020.07.002.
[3] Mallappa A, Kuralayanapalya Puttahonnappa S, Shome R, et al. Systematic review, Meta-analysis, and Pan-genome analytics predict the surging of Brucella melitensis by China and India-specific strains, elucidating the demand for enhanced preparedness. J Infect Public Health. 2025; 18: 102693. https://doi.org/10.1016/j.jiph.2025.102693.
[4] Gupta N, Bhat SN, Reddysetti S, et al. Clinical profile, diagnosis, treatment, and outcome of patients with tubercular versus nontubercular causes of spine involvement: A retrospective cohort study from India. Int J Mycobacteriol. 2022; 11: 75-82. https://doi.org/10.4103/ijmy.ijmy_243_21.
[5] Mohamad G, Amritanand R, David KS, et al. Treatment Strategy and Outcomes in Patients with Hematogenous Culture-Negative Pyogenic Vertebral Osteomyelitis. Asian Spine J. 2019; 13: 61-67. https://doi.org/10.31616/asj.2018.0093.
[6] Sha II, Edwin A, Shah N, et al. Infective Spondylodiscitis: A Comparative Analysis of Magnetic Resonance Imaging Findings with Etiology. J Orthop TraumatolRehab. 2020; 12: 129. https://doi.org/10.4103/jotr.jotr_27_20.
[7] Tanaviriyachai T, Pornsopanakorn P, Choovongkomol K, et al. A Comparative Factor Analysis and New Magnetic Resonance Imaging Scoring System for Differentiating Pyogenic Versus Tuberculous Spondylodiscitis. Neurospine. 2024; 21: 690-700. https://doi.org/10.14245/ns.2448120.060.
[8] Smimmo A, Perna A, Fantoni M, et al. Non tuberculous mycobacteria related spondylodiscitis: a case report and systematic literature review. Infez Med. 2020; 28: 425-435.
[9] Carrega G, Arena S, Bartolacci V, et al. Non-tubercular vertebral osteomyelitis: diagnosis and therapy of 45 patients from a single Italian centre. Infez Med. 2003; 11: 133-138.
[10] Boody BS, Tarazona DA, Vaccaro AR. Evaluation and Management of Pyogenic and Tubercular Spine Infections. Curr Rev Musculoskelet Med. 2018; 11: 643-652. https://doi.org/10.1007/s12178-018-9523-y.
[11] Okay G, Akkoyunlu Y, Bolukcu S, et al. Analysis of infectious spondylodiscitis: 7-years data. Pak J Med Sci. 2018;34: 1445-1451. https://doi.org/10.12669/pjms.346.15717.
[12] Gupta A, Kowalski TJ, Osmon DR, et al. Long-term outcome of pyogenic vertebral osteomyelitis: a cohort study of 260 patients. Open Forum Infect Dis. 2014; 1: ofu107. https://doi.org/10.1093/ofid/ofu107.
[13] McHenry MC, Easley KA, Locker GA. Vertebral osteomyelitis: long-term outcome for 253 patients from 7 Cleveland-area hospitals. Clin Infect Dis. 2002; 34: 1342-1350. https://doi.org/10.1086/340102.
[14] Mylona E, Samarkos M, Kakalou E, et al. Pyogenic vertebral osteomyelitis: a systematic review of clinical characteristics. Semin Arthritis Rheum. 2009; 39: 10-17. https://doi.org/10.1016/j.semarthrit.2008.03.002.
[15] Jain AK. Tuberculosis of the spine: a fresh look at an old disease. J Bone Joint Surg Br. 2010; 92: 905-913. https://doi.org/10.1302/0301-620X.92B7.24668.
[16] Kanna RM, Babu N, Kannan M, et al. Diagnostic accuracy of whole spine magnetic resonance imaging in spinal tuberculosis validated through tissue studies. Eur Spine J 2019; 28: 3003-3010. https://doi.org/10.1007/s00586-019-06031-z.
[17] Gupta N, Kadavigere R, Malla S, et al. Differentiating tubercular from pyogenic causes of spine involvement on Magnetic Resonance Imaging. Infez Med. 2022; 31: 62-69. https://doi.org/10.53854/liim-3101-9.
[18] Leonard MK, Blumberg HM. Musculoskeletal Tuberculosis. Microbiol Spectr. 2017; 5. https://doi.org/10.1128/microbiolspec.TNMI7-0046-2017.
[19] Andronikou S, Jadwat S, Douis H. Patterns of disease on MRI in 53 children with tuberculous spondylitis and the role of gadolinium. Pediatr Radiol. 2002; 32: 798-805. https://doi.org/10.1007/s00247-002-0766-8.
[20] Rajasekaran S, Soundararajan DCR, Shetty AP, et al. Spinal Tuberculosis: Current Concepts. Global Spine J. 2018; 8: 96S-108S. https://doi.org/10.1177/2192568218769053.