Le Infezioni in Medicina, n. 2, 226-232, 2025
doi: 10.53854/liim-3302-9
CASE REPORTS
Aspergillus fumigatus endocarditis in an immunocompetent host aided by metagenomic next-generation sequencing assay: case report and literature review
Carlos E. Plazola1,2, Aliya Rehman3, Maria Morel1,2, Christine Vu2, Jose G. Castro1,2, Folusakin Ayoade1,2
1Division of Infectious Diseases, University of Miami Miller School of Medicine, Miami, USA;
2Jackson Health System, Miami, USA;
3University of Maryland, Baltimore, USA.
Article received 5 February 2025 and accepted 5 May 2025
Corresponding author
Folusakin Ayoade
E-mail: fxa375@med.miami.edu
SummaRY
Aspergillus fumigatus endocarditis is a rare but severe infection associated with high mortality, typically affecting immunocompromised individuals. Diagnosing fungal endocarditis can be challenging due to the often negative blood cultures and nonspecific symptoms. We present a case of Aspergillus fumigatus infective endocarditis in a 59-year-old immunocompetent man with no typical risk factors, diagnosed with the assistance of metagenomic microbial plasma cell-free DNA next-generation sequencing assay (Karius test). The patient presented with ocular symptoms and was found to have a heart murmur and a hypodense liver lesion, leading to suspicion of infective endocarditis. Blood cultures failed to reveal a pathogen, but elevated fungal biomarkers and the Karius test supported Aspergillus fumigatus as the causal agent. The patient received antifungal therapy with voriconazole and liposomal amphotericin B followed by surgical intervention for mitral valve replacement. The case exemplifies the difficulty in diagnosing Aspergillus endocarditis, as blood cultures are often negative and histological confirmation may be delayed. Molecular diagnostics, such as metagenomic microbial plasma cell-free DNA next-generation sequencing assay, significantly enhance pathogen detection in culture-negative cases.
However, although treatment with antifungal therapy and surgery can improve outcomes, the high mortality associated with Aspergillus endocarditis remains a critical concern, highlighting the need for further research and advancements in both diagnostic and therapeutic approaches.
Keywords: Aspergillus, fungal, endocarditis, DNA, molecular.
INTRODUCTION
Fungal endocarditis is a rare but serious condition with high morbidity and mortality rates. It accounts for less than 10% (approximately 1 to 3%) of all infective endocarditis cases, with Aspergillus spp. accounting for up to 30% of all cases (second only to Candida spp.) [1, 2]. This condition primarily affects immunocompromised patients and individuals with risk factors such as intravenous drug use, malignancy, and prosthetic valves. Diagnosis can be challenging as blood cultures are often negative, necessitating confirmation through histological and tissue culture methods. This highlights the increasing importance of molecular techniques like broad-range bacterial PCR and metagenomic sequencing for accurate diagnosis. Managing fungal endocarditis remains difficult due to delayed detection and the limited effectiveness of antifungal therapy alone, contributing to its high fatality rates [3]. Here, we report a case of Aspergillus fumigatus infective endocarditis in an immunocompetent patient without typical risk factors, diagnosed with the assistance of metagenomic microbial plasma cell-free DNA next-generation sequencing assay.
CASE DESCRIPTION
This is a 59-year-old man with a past medical history of hypertension and type 2 diabetes mellitus. He presented to the emergency room complaining of headache and painless loss of vision in the right eye and floaters in the left eye for one-day duration. He denied fever, chills, night sweats, or weight loss. Other reviews of systems were negative. Social history revealed that the patient was born and raised in the Miami, South Florida area of the United States where he lived with his wife. He worked as a waste disposal truck driver. He had three dogs but denied other animal exposure. He did admit to occasionally drinking a few beers weekly but otherwise denied tobacco use, recreational drug use, or recent travel. A physical exam revealed a 2/6 systolic murmur and ophthalmologic exam showed panuveitis, as well as inflammation and vitritis in the subretinal, retinal, and choroidal areas. Other parts of the physical examination were unremarkable. Vitreous bacterial cultures were taken from the right eye, and anterior chamber paracentesis of the left eye was performed to obtain samples for additional viral and parasitic testing. The patient subsequently received intravitreal vancomycin, ceftazidime, and voriconazole in both eyes. Initial laboratory results showed mild leukocytosis of 13,600 cells/mcL with an 81.4% neutrophil predominance. Kidney and liver function testing were also within normal limits. HIV and hepatitis C testing were negative. Computed tomography (CT) scan of the abdomen and pelvis with intravenous contrast showed a hypodense lesion in the left liver lobe measuring 8 mm. The combination of ocular findings, heart murmur, and liver lesion were suggestive of infective endocarditis. He was empirically started on intravenous cefepime and vancomycin pending further infectious workup. The Infectious Diseases service was consulted and recommended switching cefepime to ceftazidime for better eye penetration. Blood cultures and further imaging were also ordered. Transthoracic echocardiogram showed a 0.9 cm mobile density on the anterior mitral valve tip, which was concerning for vegetation. Magnetic resonance imaging (MRI) of the brain with contrast showed findings consistent with
1) small bilateral cerebellar infarctions,
2) meningeal enhancements in the right sylvian, anterior interhemispheric fissures, and right frontal lobe, suggestive of meningitis,
3) right frontal subarachnoid hemorrhage and possible hemorrhagic meningitis.
On hospital day 2, the patient developed abdominal pain, and a CT of the abdomen and pelvis with intravenous contrast showed complete occlusion of the celiac artery with soft tissue attenuation surrounding the vessel measuring 3.3 x 2.6 cm, extending into the confluence of the splenic and hepatic artery as well as the left gastric artery, likely representing thrombosis with surrounding inflammatory changes. CT of the head with intravenous contrast showed a circumscribed hyper-density in the right sylvian fissure, concerning for a small aneurysm in the right M2 segment. A transesophageal echocardiogram (TEE) was also obtained which showed a mitral valve vegetation attached to A2, measuring 1 cm with moderate mitral valve regurgitation but no evidence of perforation or abscess formation. The Ophthalmology team meanwhile continued with bilateral intravitreal injections of ceftazidime, vancomycin, and voriconazole. On day 4 of the hospital course, intravenous ceftazidime was switched to meropenem, as eye findings were worsening. By day 5, blood cultures had not shown any significant microbial growth. Bartonella and Coxiella serum PCR and serologic testing were negative. Brucella IgG was positive but IgM was negative. Histoplasma urine antigen and cryptococcal serum antigen were both negative. On day 6, (1-3)-β-d-glucan (Fungitell) in the blood returned elevated at >500 pg/mL and serum Aspergillus galactomannan antigen returned at 1.40 (normal <0.5). At this time, metagenomic microbial plasma cell-free DNA next-generation sequencing assay from blood was ordered as fungal markers were elevated and blood cultures had shown no growth. On day 7, intravenous voriconazole was started. On day 8, digital subtraction angiography (DSA) showed a 2.8 x 2.4 mm M2/3 aneurysm at the bifurcation of the angular M2 segment of the middle cerebral artery, concerning for mycotic aneurysm. On day 9, metagenomic microbial plasma cell-free DNA next-generation sequencing assay returned positive for Aspergillus fumigatus. Intravenous liposomal amphotericin B was then added to intravenous voriconazole for combination therapy due to concern for active metastasizing infection, while the Ophthalmology team continued administering intravitreal voriconazole to both eyes. While on intravenous voriconazole 350 mg twice daily, a serum trough returned at 3.3 ug/mL. During this time, Cardiothoracic surgery recommended no surgical intervention due to the inability to be placed on anticoagulants required for surgery, due to recent subarachnoid hemorrhage and increased bleeding risk. On day 28, the patient was considered less at risk for hemorrhagic conversion and cleared for surgery; he underwent mitral valve replacement with a bioprosthetic mitral valve. The patient developed acute kidney injury around the time of surgery, and so intravenous liposomal amphotericin B was discontinued. On day 31, mitral valve pathology revealed acute and chronic endocarditis with numerous septate, branching hyphae morphologically consistent with Aspergillus (Figure 1 and Figure 2). On day 37, the mitral valve culture grew Aspergillus fumigatus and this was sent out for antifungal susceptibility testing. On day 38, the patient underwent clipping of the middle cerebral artery aneurysm. A week later, antifungal susceptibilities returned with the following minimum inhibitory concentrations: amphotericin 2, voriconazole 0.25, posaconazole ≤0.03 and isavuconazole 0.5. On day 71, the patient underwent open repair of mycotic superior mesenteric artery aneurysm. On day 82, the patient developed prolonged QTc, and voriconazole and was subsequently switched to isavuconazole. Due to isavuconazole’s long half-life, micafungin was also started for bridging purposes until a level could be obtained. A week later, isavuconazole trough returned 0.9 ug/mL and the dose was increased from 372 mg orally daily to twice daily. On day 114, the isavuconazole trough returned at 3.5 ug/mL, and micafungin was stopped. On day 165, the patient was discharged to a long-term acute care hospital with plans to continue prolonged isavuconazole treatment. Since then, the patient has been lost to follow up. The chronology of the clinical course is outlined in Figure 1.

Figure 1 - The clinical course chronology. CTAP, computed tomography of the abdomen and pelvis; IV, intravenous; MRI, magnetic resonance imaging; SAH, subarachnoid hemorrhage; TTE, transthoracic echocardiogram; TEE, transesophageal echocardiogram; BDG, (1-3)-β-d-glucan; GM, Aspergillus galactomannan antigen; DSA, digital subtraction angiography; AKI, acute kidney injury; AST: antimicrobial susceptibility testing.
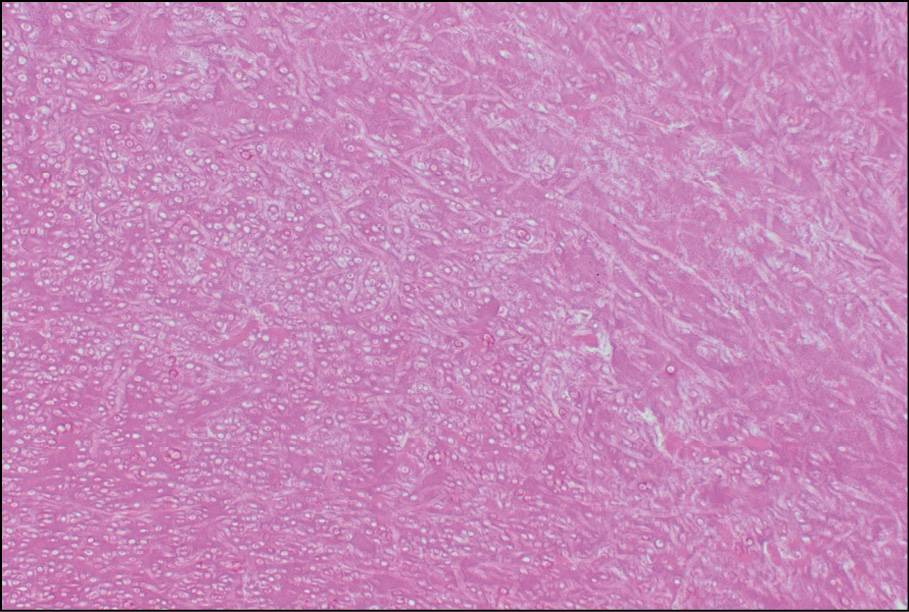
Figure 2 - Hematoxylin and eosin section slide of mitral valve tissue showing acute branching septate hyphae consistent with Aspergillus fumigatus.
SYSTEMATIC LITERATURE REVIEW
Methodology
We conducted a systematic PubMed review of cases published from 2000 to 2025 using the terms Aspergillus fumigatus and endocarditis. Inclusion was limited to patients aged ≥18 years with negative blood cultures. Seventeen cases met criteria. Data were extracted on immune status, prosthetic valve presence, affected valve, embolic events, fungal marker use, surgery, diagnostic method, treatment modification, and outcomes.
Results
The findings of the literature review are presented in Table 1. “Immunocompromise” status was reported in 10 cases (59%) [4-10], and prosthetic valves in 3 (18%) [11-13]. The mitral and tricuspid valves were most commonly involved (47% and 35%, respectively).
Table 1 - Retrospective review of 17 cases of culture-negative endocarditis caused by Aspergillus fumigatus.

Embolic phenomena were seen in 11 cases (65%), commonly affecting the brain, lungs, or abdominal organs [6-10, 13-17], findings echoed in our case. This supports the idea that Aspergillus fumigatus endocarditis should be suspected when unexplained emboli occur with negative blood cultures.
Non-invasive fungal markers especially (1-3)-βD-glucan and galactomannan were measured and elevated in 7 cases (41%) [4, 5, 7, 8, 10, 15, 16]. In our case, both markers were markedly elevated early in the clinical course and prompted further testing with metagenomic next-generation sequencing, which ultimately confirmed the diagnosis. This contrasts with the reviewed literature, where these markers were not consistently used, suggesting their diagnostic potential may be underutilized.
Pathogen detection was most commonly achieved via tissue culture (13/17, 76%), while molecular diagnostics (polymerase chain reaction or mNGS) were used in 3 cases (18%) [4, 7, 11].
DISCUSSION
Aspergillus fumigatus endocarditis is an uncommon but serious condition with high mortality. While traditionally associated with immunocompromise, indwelling devices, intravenous drug use, or prosthetic valves, our case and others show it can also occur in immunocompetent individuals. Environmental exposures, such as handling organic waste, may also increase risk [18, 19].
Diagnosis is challenging due to nonspecific symptoms and frequent embolic phenomena [1]. Blood cultures are often negative, and histology or tissue cultures, while diagnostic, are invasive and not always feasible. As a result, non-invasive fungal markers like serum (1-3)-β-D-glucan and galactomannan, along with molecular tools such as mNGS, have become increasingly recommended in culture-negative endocarditis. Our patient’s diagnosis was supported by early elevation of both serum markers and confirmed by mNGS (Karius test) [20-22].
In our case, mNGS provided the first definitive identification of Aspergillus fumigatus, enabling early, targeted therapy. This combined approach, using both fungal markers and molecular testing, was uncommon in the reviewed literature but played a pivotal role in our case. Wider adoption may improve outcomes by helping clinicians recognize and treat culture-negative Aspergillus fumigatus endocarditis earlier.
Treatment remains challenging. Despite optimal care, including surgery and antifungal therapy, mortality may approach 80% [23]. In our review, 10 patients underwent valve surgery, of whom 8 survived. Overall mortality was 41% (7/17), with worse outcomes in patients lacking surgery or facing delays in diagnosis [8, 9, 17].
Antifungal therapy was modified or initiated based on pathogen identification in 88% of the cases reviewed (15/17) [4-8, 10-16]. Though current guidelines are cautious about combination therapy, retrospective data suggest potential benefit in reducing mortality [6, 24]. This was reflected in our case, where combination therapy was used in the setting of disseminated infection and was adjusted based on antifungal susceptibility testing.
Aspergillus fumigatus endocarditis remains a rare but daunting clinical challenge. Symptoms are often vague, and blood cultures are typically negative, delaying diagnosis and treatment. Our case, alongside those reviewed, highlights the importance of early use of molecular diagnostics, especially mNGS [4, 25], and non-invasive fungal markers. Together, these tools can enable faster identification and initiation of appropriate therapy.
Outcomes vary widely, with highest mortality seen in those without surgery or with late diagnosis. Some cases are only identified post-mortem, underscoring the need for high clinical suspicion and early use of advanced diagnostics in culture-negative endocarditis. Continued progress in diagnostic and therapeutic strategies will be critical to improving outcomes in this life-threatening condition.
Conflicts of interest
None.
Funding
This work was not by specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
REFERENCES
[1] Ellis ME, Al-Abdely H, Sandridge A et al. Fungal endocarditis: evidence in the world literature, 1965-1995. Clin Infect Dis. 2001; 32(1): 50-62.
[2] Antinori S, Ferraris L, Orlando G et al. Fungal endocarditis observed over an 8-year period and a review of the literature. Mycopathologia. 2014; 178(1-2): 37-51.
[3] Pasha AK, Lee JZ, Low SW et al. Fungal Endocarditis: Update on Diagnosis and Management. Am J Med. 2016; 129(10): 1037-1043.
[4] Li X, Li D, Han D. Rapid diagnosis of Aspergillus fumigatus endocarditis using mNGS assay: A case report and review of the literature. Diagn Microbiol Infect Dis. 2024; 108(3): 116171.
[5] Kalokhe AS, Rouphael N, El Chami MF et al. Aspergillus endocarditis: a review of the literature. Int J Infect Dis. 2010; 14(12): e1040-7.
[6] Aldosari MA, Alghamdi MH, Alhamdan AA et al. Native valve fungal endocarditis caused by Aspergillus fumigatus: management dilemma. Oxf Med Case Reports. 2020; 2020(3): omz147.
[7] Hiroshima Y, Kurumisawa S, Aizawa K, Fukushima N, Kawahito K. Successful treatment of Aspergillus mural endocarditis in malignant lymphoma using a combined antifungal and surgical approach: a case report. Surg Case Rep. 2020; 6(1): 128.
[8] Chevalier K, Barde F, Benhamida S et al. Invasive aspergillosis and endocarditis. Rev Med Interne. 2021; 42(10): 678-685.
[9] García CG, García-Fernández MA, Sarnago Cebada F. Aspergillus endocarditis. Echocardiography. 2005; 22(7): 623-624.
[10] Pavlina AA, Peacock JW, Ranginwala SA et al. Aspergillus mural endocarditis presenting with multiple cerebral abscesses. J Cardiothorac Surg. 2018; 13(1): 107.
[11] Museedi AS, Nashawi M, Ghali A et al. Prosthetic mitral valve Aspergillus fumigatus endocarditis. IDCases. 2020; 21: e00891.
[12] Abuzaid AA, Zaki M, Tarif H. Atypical early Aspergillus endocarditis post prosthetic mitral valve repair: a case report. Heart Views. 2015; 16(1): 30-33.
[13] Lennard K, Bannan A, Grant P et al. Potential benefit of combination antifungal therapy in Aspergillus endocarditis. BMJ Case Rep. 2020; 13(6): e234008.
[14] Aggarwal A, Hogan K, Paez A. Aspergillus fumigatus endocarditis in a splenectomized patient with no risk factors. IDCases. 2020; 19: e00694.
[15] Marinelli T, Kidd S, Shaw D et al. Aspergillus fumigatus endocarditis in an indigenous Australian male without classic risk factors. Med Mycol Case Rep. 2018; 22: 61-64.
[16] Zhao Y, Wu X, Wang X et al. Severe fever with thrombocytopenia syndrome complicated with aspergillus endocarditis and multiple organ infarctions after glucocorticoid treatment in an immunocompetent man: a case report. BMC Infect Dis. 2025; 25(1): 116.
[17] Vohra S, Taylor R, Aronowitz P. The tell-tale heart: Aspergillus fumigatus endocarditis in an immunocompetent patient. Hosp Pract (1995). 2013; 41(1): 117-121.
[18] Pierrotti LC, Baddour LM. Fungal endocarditis, 1995-2000. Chest. 2002; 122(1): 302-10.
[19] Sabino R, Veríssimo C, Viegas C et al. The role of occupational Aspergillus exposure in the development of diseases. Med Mycol. 2019; 57(Supplement. 2): S196-S205.
[20] Hatlen TJ, Filler SG, Bayer A et al. Aspergillus endocarditis diagnosed by fungemia plus serum antigen testing. Med Mycol Case Rep. 2018; 23: 1-3.
[21] Imbert S, Gauthier L, Joly I et al. Aspergillus PCR in serum for the diagnosis, follow-up and prognosis of invasive aspergillosis in neutropenic and nonneutropenic patients. Clin Microbiol Infect. 2016; 22(6): 562.e1-8.
[22] Zeng X, Liang Y, Wang H et al. Detection of pathogens from venous or arterial blood of patients with left-sided infective endocarditis by metagenomic next-generation sequencing: A prospective study. Clin Chim Acta. 2024; 552: 117698.
[23] Ikediobi U, Sutton RE. Aspergillus fumigatus native valve infective endocarditis in an otherwise healthy adult. JMM Case Rep. 2016; 3(1): e005018.
[24] Meshaal MS, Labib D, Said K et al. Aspergillus endocarditis: Diagnostic criteria and predictors of outcome, A retrospective cohort study. PLoS One. 2018; 13(8): e0201459. Erratum in: PLoS One. 2018; 13(8): e0203103.
[25] Mitchell SL, Simner PJ. Next-Generation Sequencing in Clinical Microbiology: Are We There Yet? Clin Lab Med. 2019; 39(3): 405-418.