Le Infezioni in Medicina, n. 2, 203-211, 2025
doi: 10.53854/liim-3302-6
ORIGINAL ARTICLES
High Mortality and Antimicrobial Resistance: Challenges of Bloodstream Infections in Liver Transplant Recipients
Lucia Brescini1, Gabriele Gelo Signorino1, Roberto Montalti2, Francesco Pallotta1, Gianluca Morroni3, Daniele Nicolini4, Federico Mocchegiani4, Marco Vivarelli4, Gianluca Svegliati-Baroni5, Marcello Tavio6, Elisabetta Cerutti7, Andrea Giacometti1, Francesco Barchiesi1,8
1Infectious Disease Clinic, Department of Biomedical Sciences & Public Health, Polytechnic University of Marche, Ancona, Italy;
2Department of Clinical Medicine & Surgery, Division of Minimally Invasive & Robotic Hepato-Pancreato-Biliary Surgery, & Transplantation Service, Federico II University Hospital, Naples, Italy;
3Microbiology Unit, Department of Biomedical Sciences & Public Health, Polytechnic University of Marche, Ancona, Italy;
4Hepatobiliary surgery and Transplants AOU delle Marche, Ancona, Italy;
5Liver Injury and Transplant Unit, Polytechnic University of Marche, Ancona, Italy;
6 Infectious Disease, AOU delle Marche, Ancona, Italy;
7Division of Anaesthesia and Resuscitation AOU delle Marche, Ancona, Italy;
8Infectious Disease, Azienda Sanitaria Pesaro-Urbino, Pesaro, Italy.
Article received 19 February 2025 and accepted 18 April 2025
Corresponding author
Lucia Brescini
E-mail: l.brescini@staff.univpm.it
SummaRY
Purpose: Bloodstream infections (BSIs) are one of the most frequent complications among liver transplant recipients and are associated with a markedly increased risk of death. Our study aims to define the incidence, epidemiology and clinical characteristics of BSIs after liver transplantation (LT) and to investigate risk factors related to 30-day mortality.
Methods: We considered all patients who underwent LT at the Università Politecnica delle Marche of Ancona for up to 10 years and selected those who experienced at least one episode of BSI.
Results: Out of 601 LT recipients, 96 had at least one episode of BSI. Most BSIs occurred within the first month post-transplantation. Central vascular catheter infections were the most frequent source, followed by surgical site infections, pneumonia, and urinary tract infections. Overall, we isolated 102 microorganisms: 66% were Gram-negative bacteria, 30% Gram-positive bacteria and 4% fungi. High rates of antimicrobial resistance were observed among both Gram-negative (34%) and Gram-positive bacteria (84%).
Thirty-day mortality was 26%, with septic shock (HR 226.980 [CI 95% 5.083-10135.347], p=0.005), the absence of invasive procedures within 72 hours before BSI onset (HR 88.567 [CI 95% 2.484-357.754], p=0.014), white blood cell count ≥ 2,500/mmc (HR 34.948 [CI 95% 1.568-778.750], p=0.025), creatinine >1.02 mg/dl (HR 13.982 [CI 95% 1.487-131.485], p=0.021] and hypoalbuminemia (HR 24.775 [CI 95% 1.371-447.744], p=0.030) being significant risk factors.
Conclusions: This study provides detailed insights into post-transplant BSIs, highlighting the alarming rates of antimicrobial resistance, thereby suggesting a major effort to rationalize the use of antimicrobial therapy.
Keywords: Liver transplant, post-surgical complications, Bloodstream infections, immunosuppression, MDR pathogens.
INTRODUCTION
Infectious complications are a major cause of morbidity and mortality following solid organ transplantation (SOT). Liver transplantation, a lifesaving procedure for the treatment of many end-stage liver diseases (ESLD) and acute liver failure [1], has one of the highest rates of post-operative infections among all SOTs, due to the complexity of the surgical procedure, which involves breaching the hepatobiliary system [2].
Bloodstream infections (BSIs) are a significant cause of mortality among LT recipients [3], with the highest incidence in the first post-operative month: during this period, devices, such as central vascular lines or intra-abdominal/biliary catheters, can become sources of bacteremia. Patients are also at risk of developing urinary, pulmonary or surgical site infections [4].
Historically, Gram-positive bacteria (GPB) were considered the main cause of BSIs, due to their association with indwelling vascular devices; however, over the last decade, there has been a shift towards Gram-negative bacteria (GNB) as the predominant pathogens, with an increase in multi-drug-resistant (MDR) strains [5, 6]. This trend poses a major global concern due to limited treatment options and higher rates of poor outcomes [7].
The objective of our study was to investigate the incidence, epidemiology, and clinical characteristics of BSIs in a large cohort of LT recipients followed at our center since 2005.
PATIENTS AND METHODS
Study design
This retrospective, observational study was conducted at the Università Politecnica delle Marche, Ancona, Italy, from August 2005 to January 2021.
The study population included all patients who underwent LT during this period, selecting those who experienced at least one BSI episode.
Demographic, laboratory, and clinical variables were collected, along with operative details. Infections were identified through active surveillance in the LT ward (closely monitoring patients, ensuring the detection of any signs of complications early) and reviews of outpatient medical records. BSIs were categorized based on onset time post-LT: very early (≤30 days), early (31-180 days), intermediate (181-365 days) and late (>365 days) [1]. Mortality rate was calculated at 30 days post- BSI. The present research has been performed in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments. The Institutional Review Board of the Azienda Ospedaliero Universitaria delle Marche granted retrospective access to the data without need for individual informed consent.
Definitions and microbiology
The criteria for defining and classifying BSIs were based on those proposed by the Centers for Disease Control and Prevention [9]. Specifically, a BSI was defined by either one or more positive blood cultures growing a recognized pathogenic microorganism, or two or more positive cultures (from separate blood draws collected within a 48-hour period and from different sites) growing a common commensal organism, accompanied by at least one of the following signs/symptoms: fever, chills, or hypotension.
Multiple cultures positive for the same pathogen were considered a single BSI episode if they were separated by less than 30 days and the patient did not recover with negative blood culture in the interim. A polymicrobial BSI was defined as the isolation of two or more bacteria from a single blood culture specimen, while a mixed BSI was defined as the isolation of both bacterial and fungal species. Each microorganism was considered a separate isolate in the analysis.
The source of the BSI was determined based on clinical criteria and the isolation of the same organisms in the blood specimen and from a clinically significant site of infection [10]. The following were considered: pneumonia, surgical site infections (SSIs, including surgical wound and deep abdominal organ/space infections), central venous catheter-related infections and urinary tract infections (UTIs). When no focal infection could be demonstrated or when the germ isolated from blood cultures was not recovered from other suspected sites of infection, the source was categorized as unknown.
Susceptibility testing of the strains to antibacterial agents was performed using various methods, including Vitek-2 and Sensititre microdilution method, with results interpreted according to either CLSI (up to 2011) or EUCAST (from 2012 onwards) breakpoints [11, 12].
All susceptibility patterns were reviewed and classified according to ESCMID guidelines [13]. Specifically, multidrug resistance (MDR) was defined as acquired non-susceptibility to at least one agent in three or more antimicrobial categories, extensive drug resistance (XDR) as non-susceptibility to at least one agent in all but two or fewer antimicrobial categories (i.e., bacterial isolates remaining susceptible to only one or two categories), and pandrug resistance (PDR) as non-susceptibility to all agents in all antimicrobial categories.
Prophylaxis and immunosuppression
The standard surgical prophylaxis during the study period consisted of ampicillin/sulbactam with cefotaxime. Vancomycin or teicoplanin was administered to patients suspected of having methicillin-resistant Staphylococcus aureus (MRSA) based on preoperative screening. Prophylaxis was typically discontinued on the third day after LT unless a suspected or documented infection occurred. Selective digestive decontamination was not performed.
Antifungal prophylaxis (in selected cases) included fluconazole, liposomal amphotericin B or an echinocandin, according to ESCMID recommendations [14]. CMV prophylaxis (valganciclovir) was used in patients at high risk for CMV infection, such as seronegative recipients of seropositive donor organ and those receiving immunosuppressive therapy with anti-lymphocyte immunoglobulin.
For most patients, standard immunosuppression consisted of tacrolimus for the first 15 days after LT, followed by cyclosporine or everolimus. When combination therapy was necessary, mycophenolate mofetil was the most frequently used agent. A minority of patients received cyclosporine as the primary immunosuppressive agent.
Statistical analysis
Quantitative data were expressed as median and 1st and 3rd quartile and compared using the Mann-Whitney U test or Student’s t-test. Qualitative variables were expressed as absolute frequencies and percentages and compared using the Chi-Square test. The diagnostic accuracy of selected risk factors was evaluated using Receiver Operating Curve (ROC) analysis.
Factors associated with 30-day mortality after BSI were analyzed using a stepwise binary logistic regression model, where variables found to be significant at the univariate level (p value <0.05) with at least 5 events were introduced. Statistical analysis was performed using SPSS, version 20 (Statistical Package for Social Sciences Inc., Chicago, IL).
RESULTS
During the study period, 601 patients underwent liver transplantation (LT). Among these, 96 (16%) experienced at least one episode of bloodstream infection (BSI) post-transplant. Many BSIs (45%) occurred within the first month after LT (Figure 1A). The median age of the patients was 55.5 years, with males accounting for 76% of the population. The most common indications for LT were ESLD due to viral hepatitis (HCV 40% and HBV 28%), followed by alcoholic cirrhosis.
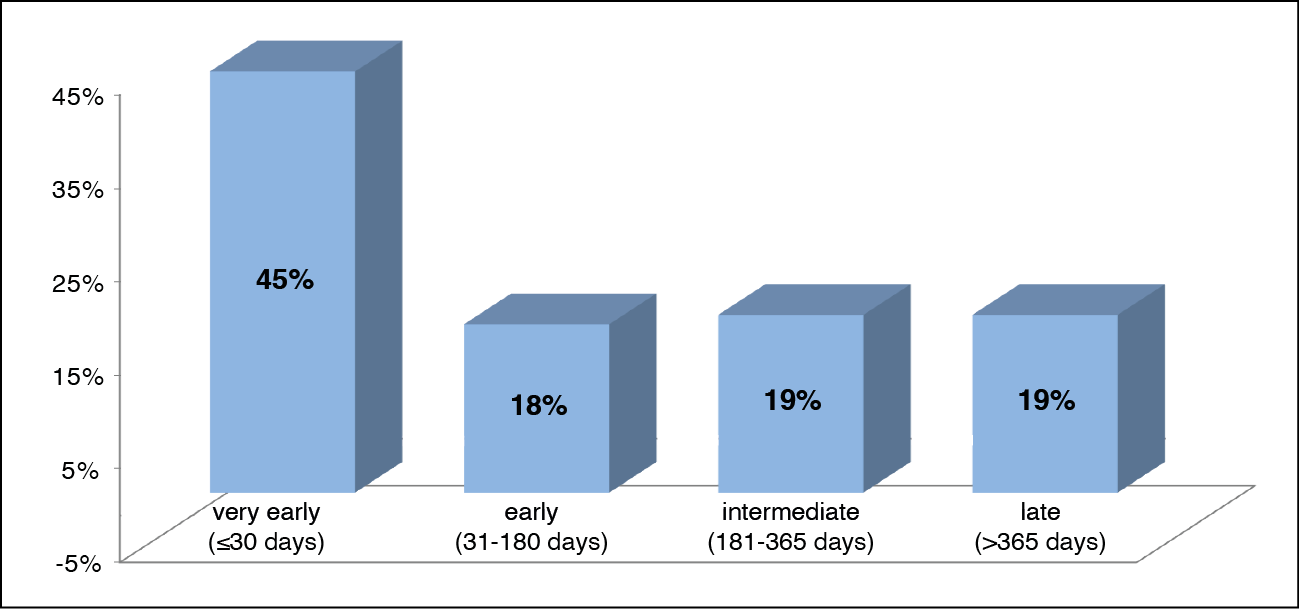
Figure 1A - BSIs distribution in post-surgical periods.
The most frequent sources of BSIs were central vascular catheter infections (23; 24%), followed by SSI (16; 17%), pneumonia (15; 16%) and UTI (6; 6%); only one case of BSI was associated with meningoencephalitis. In 35 cases (37%), the primary focus remained unknown.
Figure 1B illustrates the distribution of infectious sources based on post-surgical periods. Notably, the proportion of BSIs with unknown origin increased as the interval from LT lengthened (from 19% to 67%).
GNB accounted for 70% (67/96) of culture-positive cases, followed by GPB (29/96, 30%) and fungi (2/96, 2%). Twelve episodes (12/96, 13%) were polymicrobial while only one BSI was mixed. The frequency of the isolated species is shown in Figure 2. Enterobacterales members were the majority of GNB, with Escherichia coli (24/25%) and Klebsiella pneumoniae (12/12%) being the most frequent. Among GPB, Enterococcus faecium (13/13%), Staphylococcus epidermidis (6/6%) and Enterococcus faecalis (4/4%) were the most common.

Figure 1B - Infectious source distribution based on post-surgical periods.

Figure 2 - Frequency of causative organisms.
Note: Other gram-neg.: A. baumannii, S. marcescens, A. hydrophila, A. radioresistens, C. freundii, C. koseri, E. aerogenes, K. oxytoca, Prevotella spp., P. mirabilis. Other gram-pos.: E. avium, E. casseliflavus, E. gallinarum, S. pasterianus. Fungi: C. albicans, C. neoformans.
Among the 67 GNB isolates, 23 (34%) were antibiotic resistant strains, including 17 MDR, 6 XDR and one PDR. The MDR isolates included 2 carbapenemase-producing K. pneumoniae, 7 extended-spectrum beta-lactamase-producing (ESBL) bacteria, one Enterobacter cloacae, 2 Klebsiella aerogenes, one Serratia marcescens, 3 Pseudomonas aeruginosa and one Klebsiella oxytoca.
Among the 29 GPB isolates, 19 (65 %) were antibiotic resistant strains, including 17 MDR and 2 XDR. The MDR isolates included 4 methicillin-resistant S. epidermidis (MRSE), 2 Enterococcus faecalis, 9 Enterococcus faecium (with one vancomycin-resistant Enterococcus), one Enterococcus gallinarum and one Enterococcus raffinosus. The XDR isolates included one E. faecium Van-A.
The crude mortality rate within 30 days after BSI was 26% (25/96). Differences between deceased and surviving patients are depicted in Tables 1 to 3.
Table 1 - Demographic and clinical characteristics of the study cohort.

Table 2 - Intra- and post-operative characteristics of the study cohort.

Table 3 - Clinical, microbiological and laboratory characteristics at the time of BSI

A significantly higher proportion of female LT recipients died within one month after BSI (p=0.014); higher mortality was observed in patients with a waiting list time of less than 20 days (p=0.021). Additionally, hospitalization in the month before LT was more common among patients with negative outcomes (p=0.027) as they more frequently underwent combined kidney-liver transplantation or liver retransplantation (p=0.004).
Regarding chronic comorbidities, there were no significant differences between the two groups. However, the need for renal replacement therapy (including hemo- or peritoneal dialysis, haemo (dia)filtration and plasmapheresis) in the perioperative periods was significantly associated with higher mortality (p=0.003).
An earlier BSI onset (≤ 26 days from LT), the presence of intravascular central catheter, the occurrence of acute renal failure and septic shock were all significantly associated with higher mortality (p ranging from <0.001 to 0.044). Undergoing invasive procedures before 72 hours of BSI onset was a protective factor (p 0.044). Furthermore, a significantly larger percentage of deceased versus living LT recipients presented pneumonia as the primary source of infection (36% vs 9 %, p=0.003) and were infected with an XDR strain (24% vs 3%, p=0.004).
Among laboratory variables, serum albumin levels ≤ 2.9 g/dl, red blood cell count ≤ 2,980,000/mmc, platelet count ≤ 38,000/mmc, white blood cell count ≥ 2,500/mmc, creatinine >1.02 and hemoglobin ≤ 9 g/dl were all significantly associated with higher mortality (p ranging from <0.001 to 0.019).
Multivariate analysis identified the following independent risk factors for mortality: septic shock (HR 226.980 [CI 95% 5.083-10135.347], p=0.005), the absence of invasive procedures within 72 hours before BSI onset (HR 88.567 [CI 95% 2.484-357.754], p=0.014), white blood cell count ≥ 2500/mmc (HR 34.948 [CI 95% 1.568-778.750], p=0.025), creatinine >1.02 (HR 13.982 [CI 95% 1.487-131.485], p=0.021] and hypoalbuminemia (HR 24.775 [CI 95% 1.371-447.744], p=0.030), (Tables 1 to 3).
DISCUSSION
In this study, we found that BSIs are a common complication in LT recipients: 16% of our patients developed at least one BSI episode after transplantation. These findings align with those reported in previous studies [15-17].
Although BSIs can occur at any time post- transplant, their incidence is highest in the first post-operative month, likely due to factors such as the clinical severity of the underlying illness at the time of transplant, breaches in the muco-cutaneous barrier resulting from surgery, the use of various invasive devices, technical complications of surgery and immunosuppression [4, 6, 18]. However, our data showed that BSIs are not rare even later after the transplant.
In our center, central vascular line-associated infections were the most common source of bacteremia, primarily during the early post-operative period. This result is clearly associated with the frequent use of intravascular devices in the early post-operative care of transplant recipients.
Interestingly, no primary focus of infection could be documented in 37% of the episodes. This high rate of unknown sources may be explained by early aggressive antibiotic therapy and the intrinsic difficulty in documenting intra-abdominal and biliary sources in many cases [3].
GNB accounted for 70% of all isolates causing BSIs in our series. A shift toward GNB as the predominant cause of BSI after LT has been reported in the last decade [6].
Enterobacterales members represented the vast majority of GNB, with the most common species being E. coli (22%) [1].
Notably, we observed a high incidence rate of resistant strains (35% MDR e 8% XDR) within both Gram-negative and Gram-positive bacteria. This confirms the alarming emergence and spread of MDR pathogens over the last two decades, causing significant morbidity and mortality among transplant recipients. Drug-resistant strains can emerge due to exposure to broad-spectrum antibiotics in the pre-transplant phase and the frequent use of broad-spectrum empiric antibiotic therapy, given the vulnerability of transplant recipients [18]. The crude mortality rate in our series was 26%, consistent with data from other clinical studies documenting mortality rates ranging from 10% to 52% [16, 17].
Although as many as 15 variables (including the presence of XDR isolates, the source of infection and RRT) were linked to a higher probability of death, only five variables remained significantly associated with negative outcome in a multivariate analysis: septic shock, absence of invasive procedures within 72 hours before BSI onset, hypoalbuminemia, white blood cell count ≥ 2500/mmc and creatinine >1.02.
The association between septic shock and mortality echoed the findings of others studies [19-21] on SOT recipients with BSIs. Septic shock is the most severe complication in this context, which is not surprising as it reflects the severity of the illness. Unfortunately, improving the management of septic shock remains a significant challenge.
Hypoalbuminemia has already been described as an independently significant predictor of infections with a high risk of mortality in liver transplant recipients [19]. It is difficult to attribute the association between decreased serum albumin levels and mortality to a single pathological mechanism. One possible explanation is that, in the immediate post-transplant period, there may be a transient reduction in the hepatic production of serum proteins, such as albumin, complement, and acute phase proteins. This could result in a temporary suppression of the non-specific defense system [22]. Additionally, in a pre-clinical study, P. aeruginosa quorum sensing was inhibited by physiological levels of serum albumin, suggesting the importance of this protein in modulating its virulence [23]. Increased creatinine levels in patients with sepsis, including those who have undergone liver transplantation, have been correlated with disease severity in the literature [24].
Unlike other studies that identify leukopenia as the most common risk factor for mortality in BSI [21, 25], our study found that patients who died within 30 days more frequently had elevated WBC counts.
Curiously, since the biliary tract and the surgical site are among the most frequent sources of BSI, patients who did not receive early invasive procedures had a higher risk of mortality, probably due to poorer source control; this data underscores the importance of correct diagnosis and source control when indicated.
In conclusion, we documented that BSIs occurred at a high rate after LT, especially in the first month following surgery, with central lines being the most frequent source of infection in our center. Although GNB were the most common causative agents, a high proportion of MDR strains was found within both Gram-negative and Gram-positive bacteria. We documented a high 30-day mortality rate and identified five specific risk factors: septic shock, absence of invasive procedures within 72 hours before BSI onset, hypoalbuminemia, white blood cell count ≥ 2500/mmc, creatinine >1.02.
Being a retrospective study, our study is subject to selection bias and limitations in data collection. Furthermore, the study was conducted at a single center, which may limit the generalizability of the results to other populations or clinical settings. Although the sample of 601 patients is considerable, the number of bloodstream infection episodes (96) may not be sufficient to detect all possible significant variables.
Our study provides detailed information on post-transplant BSI occurrence. In particular, the high rates of antimicrobial resistance among blood culture isolates are concerning for these fragile patients, suggesting a major effort to rationalize the use of antimicrobial therapy.
Conflict of interest
All authors declare no conflict of interest. No financial or non-financial interests are directly or indirectly related to the work.
Funding
None.
REFERENCES
[1] Kim SI. Bacterial infection after liver transplantation. World J Gastroenterol. 2014; 20(20): 6211-6220.
[2] Linares L, García-Goez JF, Cervera C, et al. Early bacteremia after solid organ transplantation. Transplant Proc. 2009; 41: 2262-2264.
[3] Bert F, Larroque B, Paugam-Burtz C, et al. Microbial epidemiology and outcome of bloodstream infections in liver transplant recipients: an analysis of 259 episodes. Liver Transpl. 2010; 16(3): 393-401.
[4] Sganga G, Spanu T, Bianco G, et al. Bacterial bloodstream infections in liver transplantation: etiologic agents and antimicrobial susceptibility profiles. Transplant Proc. 2012; 44(7): 1973-1976.
[5] Singh N, Gayowski T, Wagener MM, et al. Bloodstream infections in liver transplant recipients receiving tacrolimus. Clin Transplant. 1997; 11(4): 275-281.
[6] Singh N, Wagener MM, Obman A, et al. Bacteremias in liver transplant recipients: shift toward gram-negative bacteria as predominant pathogens. Liver Transpl. 2004; 10(7): 844-849.
[7] Shi SH, Kong HS, Xu J, et al. Multidrug resistant gram-negative bacilli as predominant bacteremic pathogens in liver transplant recipients. Transpl Infect Dis. 2009; 11(5): 405-412.
[8] Singh N, Wagener MM, Marino IR, et al. Trends in invasive fungal infections in liver transplant recipients: correlation with evolution in transplantation practices. Transplantation. 2002; 73: 63-67.
[9] Centers for Disease Control and Prevention (CDC). Bloodstream Infection Event (Central Line-Associated Bloodstream Infection and non-central line-associated Bloodstream Infection) Available from: https://www.cdc.gov/nhsn/pdfs/pscmanual/4psc_clabscurrent.pdf
[10] Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 2008; 36(5): 309-332.
[11] Clinical and Laboratory Standards Institute: Performance standards for antimicrobial susceptibility testing. CLSI M100-S20U. Wayne, PA. Clinical and Laboratory Standards Institute. 2010.
[12] European Committee on antimicrobial susceptibility testing (EUCAST). Breakpoints tables for interpretation of MICs and zone diameters. 2016; Available from: http://www.eucast.org/clinical_breakpoints.
[13] Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012; 18 (3): 268-281.
[14] Gavalda J, Meije Y, Fortun J, et al. Invasive fungal infections in solid organ transplant recipients. Clin Microbiol Infect. 2014; 20 Suppl. 7: 27-48.
[15] Santos CA, Hotchkiss RS, Chapman WC, et al. Epidemiology of Bloodstream Infections in a Multicenter Retrospective Cohort of Liver Transplant Recipients. Transplant Direct. 2016; 2(3): e67.
[16] Kim SI, Kim YJ, Jun YH, et al. Epidemiology and risk factors for bacteremia in 144 consecutive living-donor liver transplant recipients. Yonsei Med J. 2009; 50(1): 112-121.
[17] Lida T, Kaido T, Yagi S, et al. Posttransplant bacteremia in adult living donor liver transplant recipients. Liver Transpl. 2010; 16(12): 1379-1385.
[18] Santoro-Lopes G, Ferraz de Gouvêa E. Multidrug-resistant bacterial infections after liver transplantation: An ever-growing challenge. World J Gastroenterol. 2014; 20(20): 6201-6210.
[19] Singh N, Paterson DL, Gayowski T, et al. Predicting bacteremia and bacteremic mortality in liver transplant recipients. Liver Transpl. 2000; 6: 54-61.
[20] Wan QQ, Ye QF, Ming YZ, et al. The risk factors for mortality in deceased donor liver transplant recipients with bloodstream infections. Transplant Proc. 2013; 45(1): 305-307.
[21] Oriol I, Sabé N, Melilli E, et al. Factors influencing mortality in solid organ transplant recipients with bloodstream infection. Clin Microbiol Infect. 2015; 21(12): 1104.e9-14.
[22] Hashimoto M, Sugawara Y, Tamura S, et al. Bloodstream infection after living donor liver transplantation. Scand J Infect Dis. 2008; 40(6-7): 509-516.
[23] Smith AC, Rice A, Sutton B, et al. Albumin inhibits Pseudomonas aeruginosa quorum sensing and alters polymicrobial interactions. Infect Immun. 2017; 85(9): e00116-17.
[24] Qiao B, Wu J, Wan Q, et al. Factors influencing mortality in abdominal solid organ transplant recipients with multidrug-resistant gram-negative bacteremia. BMC Infect Dis. 2017; 17(1): 171.
[25] Møller DL, Sørensen SS, Perch M, et al. Bacterial and fungal bloodstream infections in solid organ transplant recipients: results from a Danish cohort with nationwide follow-up. Clin Microbiol Infect. 2022; 28(3): 391-397.