Le Infezioni in Medicina, n. 2, 151-162, 2025
doi: 10.53854/liim-3302-1
REVIEWS
Antigen recognition and immune response to monkeypox virus infection: implications for Mpox vaccine design – a narrative review
Desalegn Abebaw1, Yibeltal Akelew1,2, Adane Adugna1, Zigale Hibstu Teffera1, Bantayehu Addis Tegegne3, Abebe Fenta1, Gashaw Azanaw Amare1, Mohammed Jemal4, Temesgen Baylie4, Aytenew Atnaf1
1Department of Medical Laboratory Science, College of Medicine and Health Sciences, Debre Markos University, 269, Debre Markos, Ethiopia;
2Department of Medicine, Centre for Inflammatory Diseases, Monash University, Clayton, VIC 3168, Australia;
3Department of Pharmacy, College of Medicine and Health Sciences, Debre Markos University, 269, Debre Markos, Ethiopia;
4Department of Biomedical Sciences, School of Medicine, Debre Markos University, 269, Debre Markos, Ethiopia.
Article received 2 December 2024 and accepted 1 April 2025
SummaRY
Monkeypox virus (MPXV) is a DNA virus from the Orthopoxvirus genus, sharing significant genomic similarity with the variola virus that causes smallpox. The cessation of smallpox vaccinations has contributed to recent Mpox outbreaks, with reduced immunity levels, particularly in younger populations born after the vaccine was discontinued. The virus triggers innate and adaptive immune responses, with toll-like receptors (TLRs) playing a key role in recognizing viral components and activating proinflammatory cytokines. However, MPXV evades the immune system by producing proteins that inhibit immune signaling pathways. Natural killer (NK) cells and interferons are crucial for early defense, but MPXV impairs their function. Adaptive immunity involves robust antibody and T-cell responses, similar to smallpox vaccination responses. Various mRNA-based candidate vaccines have demonstrated strong immunogenicity, with preclinical studies confirming their ability to trigger potent B-cell and T-cell responses. However, the genetic changes observed in the current outbreak strains necessitate ongoing surveillance of MPXV mutations and their impact on immunogenic proteins. This review aimed to summarize current insights into antigen recognition and immune responses to MPXV, with a focus on key antigenic proteins relevant to vaccine development.
Keywords: Adaptive immune response, Antigen recognition, Innate immune response, Monkeypox virus infection, MPXV.
Corresponding author
Desalegn Abebaw
E-mail: Desalegnabebaw20@gmail.com
INTRODUCTION
Monkeypox virus (MPXV) is a DNA virus belonging to the Orthopoxvirus genus and is responsible for Mpox disease [1, 2]. The virus shares 90% genomic similarity with the variola virus, which causes smallpox, and smallpox vaccines and treatments have proven effective in protecting against MPXV infection [3-5]. It has two infectious forms: the intracellular mature virion (MV) and the extracellular enveloped virion (EV), and both forms contain multiple outer membrane proteins that can trigger an immune response [2, 6]. MVs are released through host cell lysis, while the EVs exit via exocytosis [7].
Several observational studies have shown that the smallpox vaccine is approximately 85% effective in reducing MPXV infection [8]. A substantial waning of herd immunity against smallpox is projected for the 2022 Mpox outbreak [9, 10]. The cessation of smallpox vaccination is the primary reason for viral transmission, and the median age transition of cases from children to young adults is likely due to this [11, 12]. In Nigeria, the majority of Mpox patients are under 40 years of age and lack immunity since they were born after smallpox vaccinations ceased [13]. Compared with those from younger participants, plasma samples from individuals in their early 40s and older in Korea were more reactive to MPXV and smallpox viral proteins [14]. The level of herd immunity required to halt the spread of the virus among Moscow residents was found to be less than expected [4]. Moreover, only 38% of historically vaccinated individuals in China showed a reduced level of humoral response to MPXV-specific antigens [15], and this trend was observed in both HIV-negative and HIV-positive individuals [16].
Natural MPXV infection elicits a robust immune response capable of managing the disease [17] and provoking the innate and adaptive immune systems [2, 18]. Innate immune cells, including natural killer (NK) cells and monocytes, initiate the body’s defenses by producing type I interferons (IFNs) and inflammatory cytokines in response to viral invasion [19, 20]. Combinations of MV and EV surface proteins stimulate antigen-specific CD4+ T-cell responses and neutralizing antibodies, protecting mice from lethal doses of vaccinia virus (VACV) challenge [7].
MPXV proteins such as M1R, E8L, H3L, A29L, A35R, and B6R trigger strong B-cell and T-cell responses [21]. Immunocompromised individuals are at increased risk of contracting an illness and succumbing to it because of an insufficient protective immune response [22, 23]. Inadequate clearance of infected cells and infection of lesion-associated fibroblasts driven by profibrotic macrophages may contribute to merging lesions and a severe, prolonged Mpox course in immunocompromised patients [24].
There is no licensed vaccine directly originating from MPXV, and vaccines from other Poxviridae family viruses have been recommended [21]. Currently, the WHO-approved vaccines for Mpox include replicating (ACAM2000), low-replicating (LC16m8), and nonreplicating (MVA-BN) types [25]. However, replication-competent second-generation smallpox vaccines are restricted by potential risks [26]. JYNNEOS is a third-generation FDA-approved vaccine recommended for adults containing the weakened Modified Vaccinia Ankara-Bavarian Nordic (MVA-BN) virus [27, 28]. It induces neutralizing antibody response against A29, A35, B6, M1, H3, and I1 proteins of MPXV [29]. Compared with first- and second-generation VACV-based protein vaccines, this vaccine has fewer side effects [30]. However, this vaccine is not licensed for the population under 18 years of age [31]. No specific therapy currently exists for MPXV infection, and the virus has developed resistance to several antiviral treatments, highlighting the urgent need for alternative therapies for this deadly disease [32, 33].
MPXV circulates in two distinct clades: Clade I, with a case fatality rate of 10.6%, and Clade II, with a rate of 3.6% [34, 35]. The clade I lineage has Ia and Ib subclades [36], whereas clade II consists of IIa and IIb, the latter being responsible for the ongoing global outbreak [37]. Zoonotic transmission is a key factor in the spread of clades Ia and IIa, whereas clades Ib and IIb primarily spread via continuous transmission between humans [38].
The first human MPXV infections emerged as zoonoses in Central and West Africa approximately 1970, with sporadic cases reported throughout the decade [39]. The Ministry of Public Health of the Democratic Republic of Congo (DRC) reported over 11,806 cases, including more than 2,298 confirmed cases and 459 deaths as of 2024 [40]. By 2022, the virus had spread beyond Africa, reaching Europe, the Americas, Asia, and Oceania, with more than 99,500 cases globally [41-43]. Between January 2022 and November 2024, 127 countries reported 117,663 confirmed cases and 263 deaths to WHO [44].
This study aims to explore the complex dynamics of antigen recognition and immune responses to Mpox, which are crucial for developing effective vaccines and therapeutic strategies.
METHODS
We conducted a thorough electronic search of research databases, including; Scopus, PubMed, PubMed Central (PMC), Web of Science, Google Scholar, and Cochrane Library via a combination of keyword terms for “Mpox” or “Monkeypox”, “Monkeypox virus infection”, “antigen recognition”, “innate immune response” and “adaptive immune response”. Articles written in languages other than English were excluded. The authors prioritized recently published papers, although no specific time frame was set for study inclusion. A manual search was also performed, and all articles were initially screened to identify relevant ones.
Antigen recognition and the innate immune response to MPXV infection
The role of toll-like receptors (TLRs)
TLRs are pattern recognition receptors that distinguish pathogen-associated molecular patterns (PAMPs) from viruses, including Mpox. For example, TLR3 senses viral double-stranded RNA (dsRNA), whereas TLR9 detects unmethylated CpG DNA sequences characteristic of viral genomes [20, 45]. Viral dsRNA also activates protein kinase R (PKR), which phosphorylates eukaryotic translation initiation factor-2a (eIF2α), inhibiting protein translation and triggering antiviral responses [46].
The activation of TLRs stimulates the production of proinflammatory cytokines and IFN I, which are crucial for an effective immune response to MPXV. This response involves cytokines such as IL-6, TNF-α, and IL-12 [19, 45]. The myeloid differentiation factor 88 (MyD88)-dependent pathway predominantly mediates this process for most TLRs, activating the NF-κB and MAPK signaling pathways and intensifying inflammatory responses [45]. TLR3 activation in the lungs enhances inflammation to combat viral replication, whereas TLR9 activation is key for activating dendritic cells and recruiting NK cells to infection sites and is crucial for controlling viral spread [20]. Additionally, TLR2 plays a role in activating NK cells and promoting the differentiation of memory cells into CD8+ T cells. This results in enhanced production of antimicrobial peptides, such as cathelicidin (LL-37), in mast cells [19, 20]. Human tripartite motif protein 5α (TRIM5α) binds to the capsid protein L3 of MPXV and restricts viral replication [47].
Despite its role in boosting the immune response, MPXV produces proteins that disrupt host immune pathways by inhibiting apoptosis, altering chemokine binding, and blocking complement activation, allowing it to evade immune detection more effectively [20, 48]. Binding of the viral F3L protein with its dsRNA disrupts the signaling pathways associated with PKR and decreases IFN production [49, 50]. The virus also produces proteins that interfere with the NF-κB signaling pathway, impairing the host’s ability to generate an effective inflammatory response, leading to extended viral replication and more severe disease outcomes [20]. The MPXV protein p2 interacts with karyopherin α-2 (KPNA2) to promote its nuclear translocation, while competitively inhibiting KPNA2-mediated interferon regulatory factor 3 (IRF3) nuclear translocation and suppressing downstream IFN production [51] (Figure 1). Moreover, the viral C6 protein interacts with the host TRIM5α and is subjected to proteasome-dependent degradation [47].
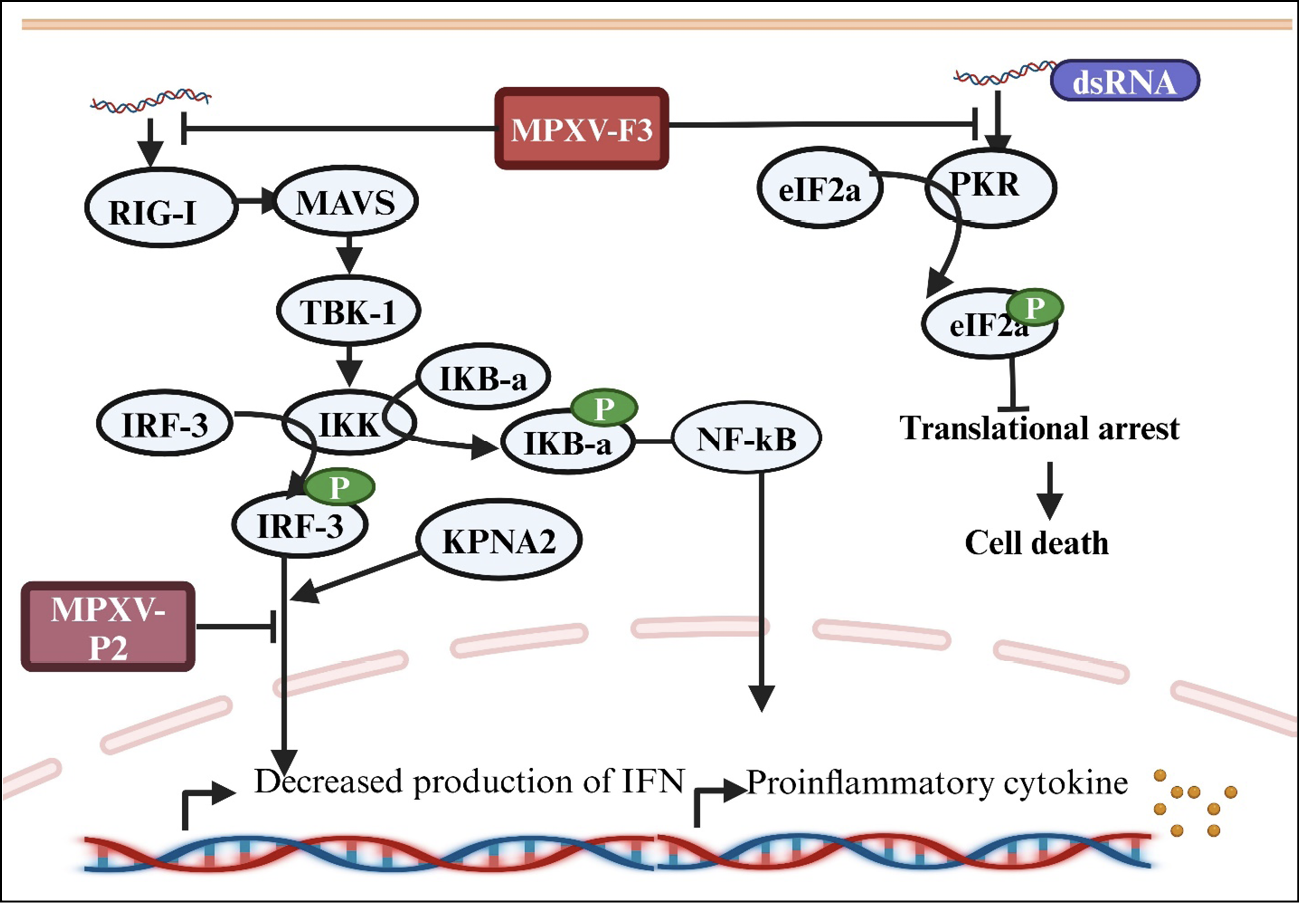
Figure 1 - Innate immune evasion strategies of MPXV: evasion from detection, inhibition of host cell apoptosis, and disruption of host immune signaling pathways. The figure was produced with https://app.biorender.com
Notes: RIG-1, retinoic acid-inducible gene I; MAVS, mitochondrial antiviral signaling; MPXV-F3, monkeypox virus protein F3; MPXV-P2, monkeypox virus protein 2; dsRNA, double-stranded RNA; PKR, protein kinase R; eIF2a, eukaryotic translation initiation factor 2a; TBK-1, TANK-binding kinase 1; IRF-3, interferon regulatory factor 3; IKB-a, inhibitor of kappa B alpha; IKK, inhibitor of nuclear factor-κB kinase; NFkB, nuclear factor kappa B; KPNA2, karyopherin subunit alpha 2
Natural killer (NK) cell response
The function of NK cells is essential for the host’s immune defense against Mpox [52]. During MPXV infection, NK cells are activated via TLR2 signaling and express NKG2D receptors, enabling them to recognize stress-induced ligands on infected cells [20]. The release of perforin and granzymes facilitates the cytotoxic activity of NK cells [19]. Their protective role against lethal MPXV infection was demonstrated in IL-15-treated CAST mice [53].
Studies have shown that MPXV infection significantly increases the number of NK cells. In rhesus monkeys, the total number of NK cells in the blood increased approximately 23-fold by day 7 after infection, whereas the number of NK cells in the lymph nodes increased approximately 46.1-fold by days 8–9. This expansion was marked by the increased proliferation of different NK cell subsets, such as CD56+ and CD16+ cells, as indicated by increased expression of Ki67, a marker of cell proliferation. However, their functionality is greatly impaired, and the expression of chemokine receptors, including CXCR3, CCR6, and CCR7, is significantly downregulated [54].
The downregulation of chemokines likely hampers the ability of NK cells to migrate to inflamed tissues. Additionally, NK cells display reduced degranulation and decreased secretion of key cytokines, such as interferon-gamma (IFN-γ) and tumor necrosis factor-alpha (TNF-α) [19, 54]. MPXV produces the Orthopoxvirus major histocompatibility complex class I (MHC I)-like protein (OMCP), which binds to the NKG2D receptor on NK cells, helping the virus evade the NK cell response. Additionally, this protein reduces MHC I molecule expression in infected cells, further decreasing the likelihood of NK cell activation [20]. Moreover, the virus directly targets NK cells, reducing their number and functionality during infection [50]. Given the essential role of NK cells in controlling viral loads, the use of NK cell stimulants or enhancers and targeting specific receptors such as NKG2D may increase their ability to detect and destroy infected cells [55].
Interferon response
Type I and II IFNs are critical in the host defense against MPXV infection [52]. In vitro experiments indicated that treatment with IFN-β significantly prevented the production and spread of MPXV. IFN-β enhances the expression of the antiviral protein myxovirus resistance protein A (MxA) in infected cells, thereby assisting in the inhibition of MPXV [56]. The protection of IFN-γ against lethal MPXV infection has also been demonstrated in mouse models [57]. During viral infections, CD4+ and CD8+ T cells are the main producers of IFN-γ. As NK cells kill virally infected cells [58], IFN-γ increases the cytotoxic activity of NK cells and supports the differentiation of T cells into Th1 cells, both of which are crucial for strong antiviral defenses [59]. It also aids in the elimination of infected cells by increasing the expression of MHC molecules, which display viral antigens to T cells [19, 48].
However, MPXV evades the IFN I response [52, 60]. The virus produces a protein known as B16 (an orthologue of VACV B19), which functions as a decoy receptor by binding to IFNs. This prevents IFNs from binding to their actual receptors, thereby blocking downstream IFN signaling and antiviral effects [52, 61] (Figure 2). The virus also evades the host immune response by releasing proteins that antagonize the functions of host IFN-γ [62].
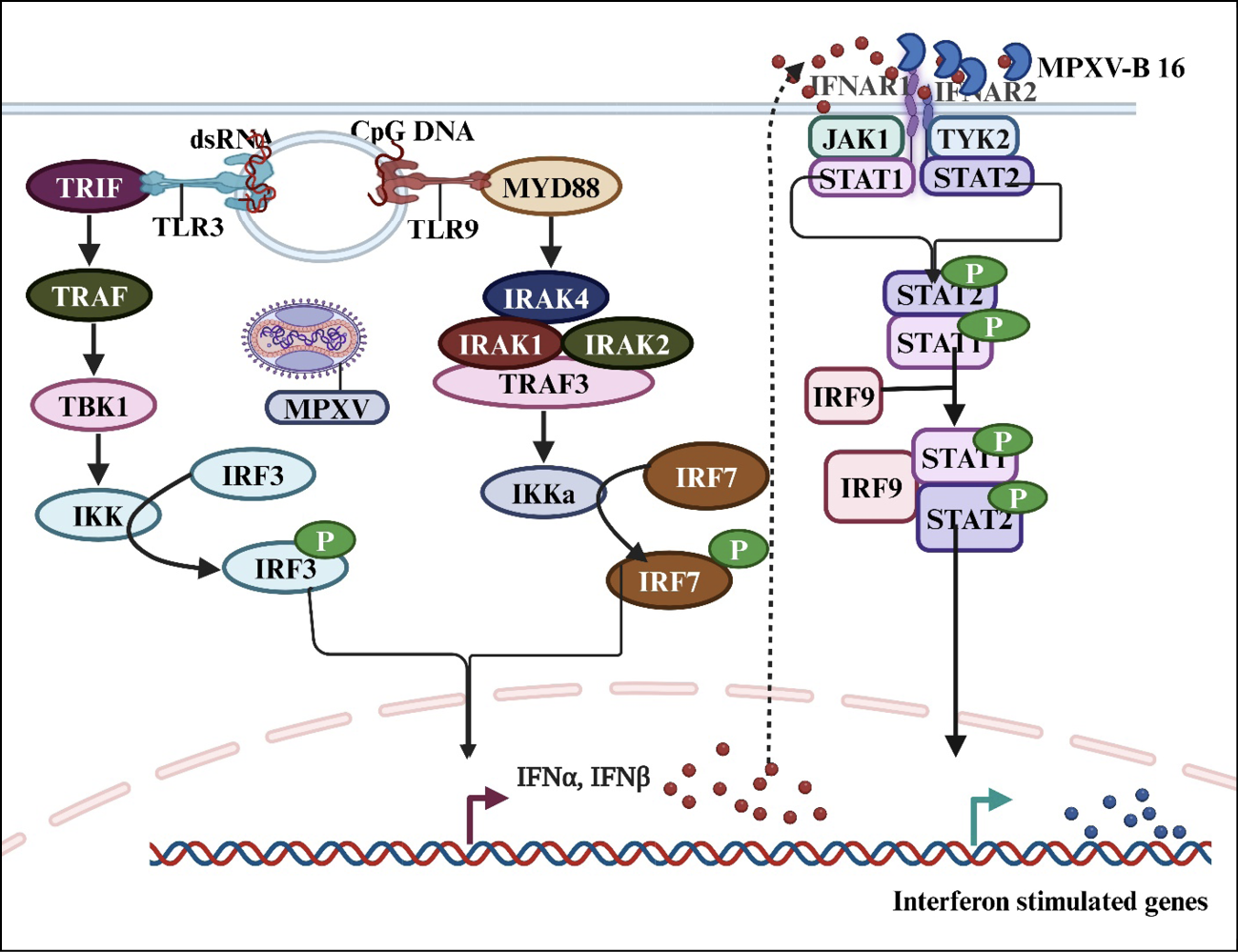
Figure 2 - Activation of TLR signaling during MPXV infection and the mechanisms of viral evasion from the IFN response. The figure was produced using https://app.biorender.com
Notes: dsRNA, double-stranded RNA; CpG DNA, cytosine-phosphate-guanosine DNA; TRIF, toll and interleukin-1 receptor (TIR) domain-containing adaptor inducing interferon β; TLR, toll-like receptor; MyD88, myeloid differentiation factor 88; IRF, interferon regulatory factor; TRAF, tumor necrosis factor receptor-associated factor; TBK1, TANK binding kinase 1; MPXV, monkeypox virus; MPXV-B16, monkeypox virus protein B16; IKK, inhibitor of nuclear factor-κB kinase; IRAK, interleukin-1 receptor-associated kinase; IFNAR, interferon alpha/beta receptor; JAK2, Janus kinase 2; TYK2, tyrosine kinase 2; IFN, interferon; STAT, signal transducer and activator of transcription.
Antigen recognition and adaptive immune response to MPXV infection
B-cell response
MPXV infection triggered antibody responses to various poxvirus antigens similar to those observed in Smallpox-vaccinated individuals, reflecting the antigenic similarity between VACV and MPXV [48, 63, 64]. The MPXV-2022 sequences present an average genetic similarity of approximately 84% to the VACV reference sequence, and VACV proteins recognized by neutralizing antibodies exhibit high sequence similarity with MPXV-2022 orthologs [65]. Preexisting monoclonal antibodies from the vaccinia vaccine demonstrated broad binding to epitopes on the B6 protein of MPXV and orthologs of variola and cowpox viruses [66]. Neutralizing antibodies against the A29, A35, B6, M1, H3, and I1 antigens of MVA-BN also demonstrated cross-reactivity against MPXV in mouse models [29, 67]. A subset of individuals in China vaccinated with the historic VACV Tiantan (VTT) strain demonstrated cross-reactivity to the MPXV surface proteins A35R, B6R, A29L, and M1R [68, 69]. Recombinant versions of these proteins stimulate the production of neutralizing antibodies in mice, significantly suppressing viral replication [6]. Additionally, those vaccinated with VTT strains before 1980 presented with cross-reactive IgG antibodies against MPXV [70, 71].
Despite the antigenic homology among Orthopoxvius antigens, previous studies highlighted that differences in amino acid sequences can affect cross-protection, as observed in A33R orthologs, underscoring the need for an MPXV-specific vaccine [72]. Furthermore, studies on MVA vaccine immunogenicity indicate low levels of neutralizing antibodies against MPXV, reflecting antigenic variability among poxvirus targets [73]. Comparisons between 2022-2023 MPXV strains and smallpox vaccine strains revealed amino acid changes in B-cell epitope regions, raising concerns regarding vaccine effectiveness [74]. Compared with classical strains isolated from 218–2019, the 2022 outbreak strain also exhibited an unexpected level of genetic divergence, with an average of 50 single nucleotide polymorphisms, exceeding predictions based on the estimated Orthopoxvirus substitution rate [75]. In the presence of these changes, the effectiveness of existing vaccines in the context of the current multi-country outbreak still needs to be verified [25]. On the other hand, three monoclonal antibodies (9F8, 3A1, and 2D1) produced against A29L protein of MPXV were found to effectively neutralize Orthopoxviruses [76]. The B16 protein B-cell epitopes are more specific to MPXV, allowing differentiation between MPXV-infected individuals and those vaccinated with MVA-BN [77].
Antibody responses are crucial in protecting against MPXV, with infected individuals generating strong responses to MPXV proteins [19, 78]. During the 2022 MPXV outbreak, antibody profiles, including IgG, IgM, IgA, and neutralizing antibodies, were observed across individuals regardless of prior vaccination status [79]. High IgG and IgA levels are correlated with quicker viral clearance. However, HIV-positive individuals show a more rapid antibody decline and lower neutralizing antibody levels over time compared to those without HIV [80, 81]. The rapid decline in the antibody titer could result from HIV-induced disruption of the coordination between humoral and cellular responses [80]. Moreover, IgG and IgM antibodies were detected 3–5 days later in HIV-positive MPXV patients than in HIV-negative MPXV patients [82]. High seropositivity rates for A29L and H3L were noted in men experiencing acute infections [83]. Individuals who recovered from MPXV infection presented stronger antibody and B-cell responses to H3L and A35R than those vaccinated with vaccinia-based vaccines did, indicating that these protein markers are associated with natural MPXV infection [84]. Additionally, compared with the MVA-BN vaccine, MPXV infection elicited stronger antibody responses to A29L, A35R, A33R, B18R, and A30L [85, 86].
Complement proteins enhance neutralizing antibody production against MPXV, and this enhancement was more effective in those with a history of smallpox vaccination [87]. Clade I MPXV encodes a complement control protein (CCP), which prevents the classical and alternative pathway of complement activation. The removal of CCP from these strains reduces Mpox disease morbidity and mortality without significantly altering the viral load in prairie dogs [88]. However, the loss of CCP expression has been reported to restrict the adaptive immune response to MPXV infection in rhesus monkeys [89].
T-cell response
The immune response to MPXV infection involves a complex interplay of T-cell activation and immune evasion mechanisms [50, 86]. Orthopoxvirus-specific T cells are crucial for eliminating MPXV and can persist over time [80]. In a study of 17 patients with confirmed Mpox, a rapid and robust T-cell response was observed, marked by increased inflammatory mediators, regardless of HIV status [90]. MPXV infection activates CD4+ and CD8+ T cells, which contribute to a Th1-type response essential for viral clearance [80, 91, 92]. These T cells produce a range of inflammatory cytokines, including IL-1β, IL-8, IL-6, TNF, MCP-1, and IFN-γ [93]. Notably, protective T-cell responses were observed in HIV-positive patients, with significantly greater IFN-γ and IL-2 responses in MPXV-infected individuals than in those vaccinated with smallpox [94]. Additionally, histopathological analyses of Mpox skin lesions revealed a balanced presence of CD4+ and CD8+ T cells in dermal inflammatory infiltrates [95].
Research has demonstrated that IFNγ-producing Th1 effector memory cells safeguard the skin against pox virus infections [96]. Most epitopes from VACV vaccines are conserved in MPXV and can trigger memory T-cell responses [97, 98]. The F8L protein and its analog F9L are pox virus-specific proteins with a common CD8+ T-cell epitope for all pox viruses and a unique epitope conserved in MPXV and VACV [99]. Over four decades after VACV exposure, older individuals presented long-lived memory CD8+ T-cells that targeted conserved VACV/MPXV epitopes. Additionally, strong effector memory MPXV-specific CD4 and CD8 responses were observed in mild Mpox cases [100]. Analysis of MPXV-specific T-cell responses in recovered Mpox patients revealed a significant presence of Th1 and Th2 memory cells in response to MPXV and VACV antigens compared with healthy donors [101]. Historic smallpox vaccination protects against MPXV infection via T-cell responses, and significant preexisting CD8+ T-cell reactivities were found toward both conserved and variant epitopes between VTT and MPXV [17, 71]. Notably, studies on nonhuman primate MPXV infections have revealed the development of immune memory in gamma-delta (γδ) T-cells, which may play a role in protecting against secondary infections [102].
MPXV-specific CD4+ and CD8+ T-cells recognize VACV-infected monocytes and produce inflammatory cytokines such as IFNγ and TNFα but mostly do not respond to MPXV-infected cells [50]. MPXV-infected cells were demonstrated to inhibit antigen-specific T-cell activation via the T-cell receptor (TCR) [50, 64]. Recent studies have shown that the M2 protein of MPXV binds to the B7 molecule on antigen-presenting cells, disrupting the B7-CD28 interaction and inhibiting T-cell costimulation during activation [103] (Figure 3). This blocks inflammatory cytokine production and likely aids in the spread of MPXV within the host [50, 104].
A dominant Th2 response rather than a Th1 response during MPXV infection increases disease severity, as the virus evades the immune system by promoting a Th2 response that suppresses the Th1 response necessary for effective virus elimination [105, 106].
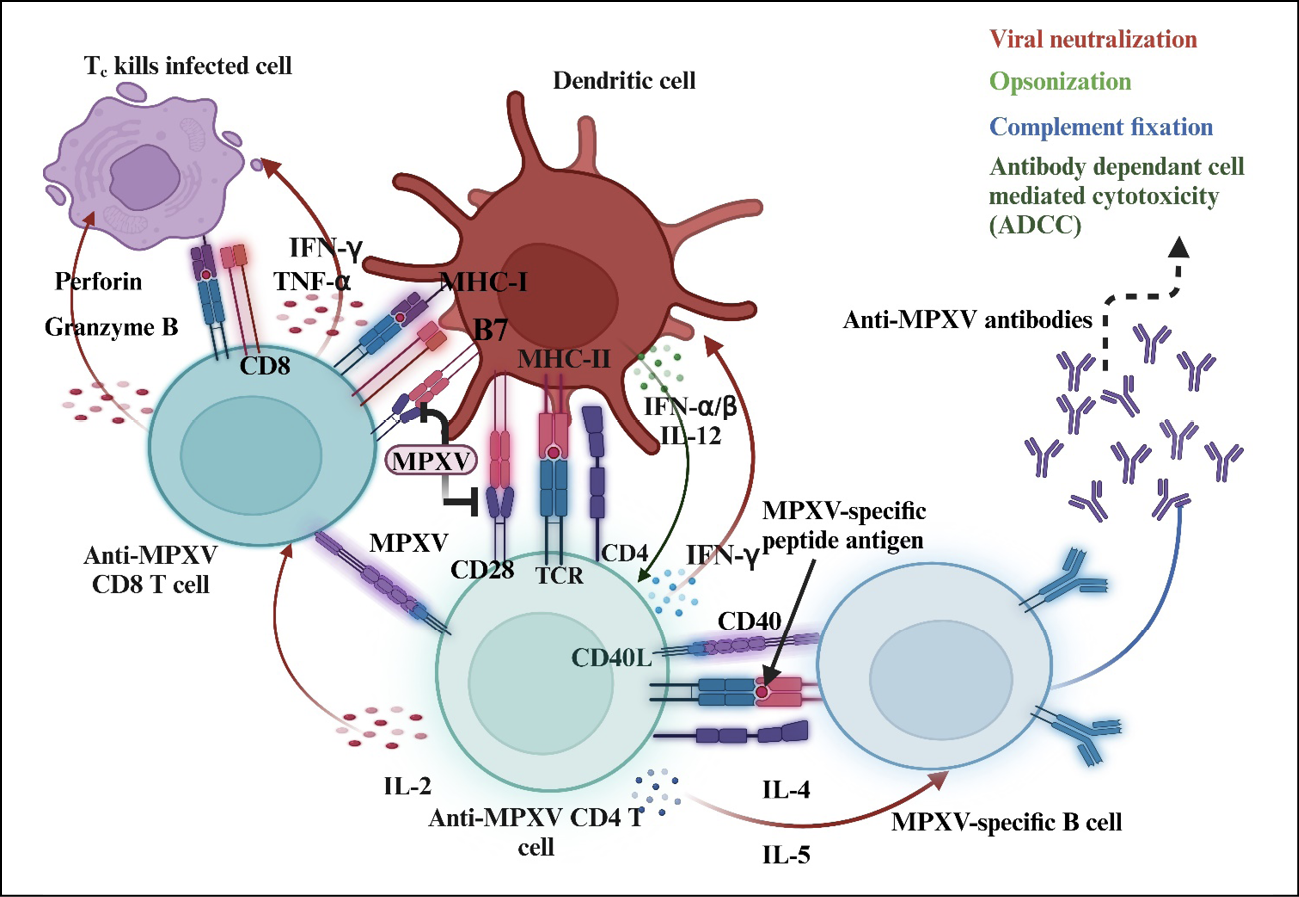
Figure 3 - Adaptive immune response against MPXV and inhibition of T-cell costimulation. The figure was produced using https://app.biorender.com
Notes: MPXV, monkeypox virus; IL, interleukin; TNF-α, tumor necrosis factor alpha; IFN-γ, interferon-gamma; IFN-α/β, interferon-alpha/beta; TCR, T-cell receptor; MHC, major histocompatibility complex.
Immune response heterogeneity in different groups
Although data on immune response variability specific to MPXV across age, sex, and different groups are lacking, sex-based differences were observed in MVA-BN smallpox vaccine recipients, with males exhibiting an average of 27% higher anti-MVA titers [107]. However, prior studies on the immune response to the Dryvax® vaccine revealed that although no significant differences were observed based on race or ethnicity, females presented significantly higher neutralizing antibody titers than males did [108]. On the other hand, the Dryvax® vaccine-induced T-cell response indicated that males presented a strong T-cell response, and statistically significant differences in the secretion of IL-2, IL-1β, and IL-10 were observed. Moreover, Caucasians had higher levels of IFNγ-producing CD8 T cells, IL-2, and TNF-α than African Americans and Hispanics [109].
Current vaccine candidates
Multiple vaccine candidates for MPVX are available, with various platforms focusing on potent immunogens such as M1R, E8L, H3L, A29L, A35R, and B6R proteins to trigger strong B-cell and T-cell responses [110]. Two mRNA vaccine candidates, BNT166a (quadrivalent, encoding MPXV antigens A35, B6, M1, and H3) and BNT166c (trivalent, lacking H3), were evaluated pre-clinically and elicited strong T-cell and antibody responses. Both vaccines confer complete protection against vaccinia, as well as clade I and clade IIb MPXVs in animal models, and are currently undergoing clinical evaluation (NCT05988203) [111]. Two multiantigen mRNA vaccine candidates encoding either four MPXV antigens (M1, A29, B6, and A35; designated Rmix4) or six antigens (M1, H3, A29, E8, B6, and A35; designated Rmix6) were developed. Both multiantigen mRNA vaccines induced strong cross-neutralizing responses against VACV, with Rmix6 generating significantly stronger cellular immunity than Rmix4 in mice [112]. Three mRNA vaccines encoding the MPXV proteins A35R and M1R were developed, including A35R extracellular domain-M1R fusions (VGPox 1 and VGPox 2) and a formulation with encapsulated full-length A35R and M1R mRNAs (VGPox 3). These vaccines induce early anti-A35R antibodies and protect against lethal VACV challenge in mice [113]. Recently, Mucker et al. tested a novel Mpox mRNA-1769 vaccine encoding MPXV surface proteins in a lethal nonhuman primate MPXV model and compared it with MVA strain 572, which is closely related to the standard-of-care MVA-BN vaccine. The authors reported comparable protection against lethality but superior efficacy in preventing disease [43]. Moreover, the mRNA-1769 vaccine protects mice from intranasal and intraperitoneal MPXV infections, while bioluminescence imaging demonstrated that vaccination markedly reduces VACV replication and spread at inoculation sites [114]. A phase I/II clinical trial is currently assessing the safety and efficacy of this novel mRNA-1769 in humans under a clinical registration number (NCT05995275).
Researchers developed a polyvalent mRNA candidate vaccine (MPXVac-097) against Mpox and evaluated the immune response in mice. Five MPXV viral antigens, A29L, E8L, M1R, A35R, and B6R, were tandemly connected via 2A peptides and optimized through codon modification. This vaccine induces a broad neutralizing antibody response, MPXV-specific T-cell activation, and protection against VACV challenge. The administration of this vaccine did not result in considerable pathological alterations in mice [115].
LIMITATIONS OF THE STUDY
This work is presented as a narrative review. Consequently, its scope is limited and does not offer a comprehensive review of the subject matter. The authors did not conduct a systematic literature review or directly compare studies. Therefore, the included material and conclusions drawn are not exhaustive and may reflect the author’s perspective.
CONCLUSIONS AND FUTURE PERSPECTIVES
The recent rise of Mpox as a global concern underscores the need for deeper understanding and preparedness in combating this virus. While the smallpox vaccine offers significant cross-protection against MPXV, waning immunity due to the cessation of smallpox vaccination has contributed to the spread of the virus, affecting previously protected demographics. Immune responses to MPXV are complex and involve innate and adaptive mechanisms, with crucial roles played by NK cells, B cells, and T cells, alongside specific viral proteins that facilitate immune evasion. Although third-generation vaccines such as JYNNEOS provide safer options for at-risk populations, age restrictions and a lack of direct antiviral treatments leave vulnerable groups at greater risk. Several MPXV-specific mRNA-based vaccines have shown promising clinical efficacy in protecting against MPXV; some are currently under clinical evaluation. However, the genetic divergence observed in the current outbreak strain may hinder vaccine efficacy. Therefore, ongoing surveillance of MPXV mutations, targeted vaccine development, and vaccine efficacy verification in the context of the current outbreak strain are recommended.
Authors’ contributions
All the authors directly participated in the preparation of this manuscript. Conceptualization: D. A., Y. A., A.A., M.J., and A.A.; Literature search: D.A., Y.A., A.A., Z.H.T., A.F, G.A.A., M.J., T.B., and A.A.; Original draft preparation: D.A., Y.A., A.A., Z.H.T., B.A.T., A.F., G.A.A., M.J., T.B., and A.A.; Critical review: D.A., Y.A., A.A., Z.H.T., B.A.T., A.F., G.A.A., M.J., T.B., and A.A.; Supervision: D.A., Y.A., M.J., A.A., and A.A. All the authors have read and approved the final manuscript.
Conflict of interest
All the authors declare that they have no competing interests.
Funding
Nothing to declare.
REFERENCES
[1] Alakunle E, Moens U, Nchinda G, Okeke MI. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses. 2020; 12(11): 1257.
[2] Yi XM, Lei YL, Li M, Zhong L, Li S. The monkeypox virus-host interplays. Cell Insight. 2024; 3(5): 100185.
[3] Hatmal MM, Al-Hatamleh MAI, Olaimat AN, et al. Comprehensive literature review of monkeypox. Emerg Microbes Infect. 2022; 11(1): 2600-2631.
[4] Gushchin VA, Ogarkova DA, Dolzhikova IV, et al. Estimation of anti-orthopoxvirus immunity in Moscow residents and potential risks of spreading Monkeypox virus. Front Immunol. 2022; 13: 1023164.
[5] Reina J, Iglesias C. Vaccines against monkeypox. Med Clin (Barc). 2023; 160(7): 305-309.
[6] Tang D, Liu X, Lu J, et al. Recombinant proteins A29L, M1R, A35R, and B6R vaccination protects mice from mpox virus challenge. Front Immunol. 2023; 14: 1203410.
[7] Zhang N, Cheng X, Zhu Y, et al. Multi-valent mRNA vaccines against monkeypox enveloped or mature viron surface antigens demonstrate robust immune response and neutralizing activity. Sci China Life Sci. 2023; 66(10): 2329-2341.
[8] Reynolds MG, McCollum AM, Nguete B, Shongo Lushima R, Petersen BW. Improving the Care and Treatment of Monkeypox Patients in Low-Resource Settings, Applying Evidence from Contemporary Biomedical and Smallpox Biodefense Research. Viruses. 2017; 9(12): 380.
[9] Riccardo V, Pablo GC. Neutralization Determinants on Poxviruses. Viruses. 2023; 15(12): 2396.
[10] Jayswal S, Kakadiya J. A narrative review of pox: smallpox vs monkeypox. Egypt J Intern Med. 2022; 34(1): 90.
[11] Bunge EM, Hoet B. The changing epidemiology of human monkeypox-A potential threat? A systematic review. PLoS Negl Trop Dis. 2022; 16(2): e0010141.
[12] Saghazadeh A, Rezaei N. Poxviruses and the immune system: Implications for monkeypox virus. Int Immunopharmacol. 2022; 113(Pt A): 109364.
[13] Petersen E, Kantele A, Koopmans M, et al. Human Monkeypox: Epidemiologic and Clinical Characteristics, Diagnosis, and Prevention. Infect Dis Clin North Am. 2019; 33(4): 1027-1043.
[14] Kim Y, Kim G, Min G, et al. Age-related antibody response to Orthopoxviruses and implications for public health measures: Insights from a South Korean study. J Infect Public Health. 2024; 17(6): 956-960.
[15] Zhang K, Xu Y, Zhang H, Zheng H, Chen X, Lin D. Low levels of humoral response to monkeypox virus-specific antigens in healthy individuals. J Infect. 2023; 86(6): e156-e157.
[16] Li D, Wang H, Sun L, et al. Levels of antibodies against the monkeypox virus compared by HIV status and historical smallpox vaccinations: a serological study. Emerg Microbes Infect. 2024; 13(1): 2356153.
[17] Sammartino JC, Cassaniti I. Characterization of immune response against monkeypox virus in cohorts of infected patients, historic and newly vaccinated subjects. J Med Virol. 2023; 95(5): e28778.
[18] Pashazadeh Azari P, Rezaei Zadeh Rukerd M, Charostad J, et al. Monkeypox (Mpox) vs. Innate immune responses: Insights into evasion mechanisms and potential therapeutic strategies. Cytokine. 2024; 183: 156751.
[19] Li H, Huang QZ, Zhang H, et al. The land-scape of immune response to monkeypox virus. EBioMedicine. 2023; 87: 104424.
[20] Parnian R, Heydarifard F, Mousavi FS, Heydarifard Z, Zandi M. Innate Immune Response to Monkeypox Virus Infection: Mechanisms and Immune Escape. J Innate Immun. 2024; 16(1): 413-424.
[21] Pritam M. Exploring the whole proteome of monkeypox virus to design B cell epitope-based oral vaccines using immunoinformatics approaches. Int J Biol Macromol. 2023; 252: 126498.
[22] Ahmed SK, Mohamed MG. Monkeypox (mpox) in immunosuppressed patients. F1000Res. 2023; 12: 127.
[23] Saghazadeh A, Rezaei N. Insights on Mpox virus infection immunopathogenesis. Rev Med Virol. 2023; 33(2): e2426.
[24] Matschke J, Hartmann K, Pfefferle S, et al. Inefficient tissue immune response against MPXV in an immunocompromised mpox patient. J Med Virol. 2024; 96(7): e29811.
[25] Saadh MJ, Ghadimkhani T, Soltani N, et al. Progress and prospects on vaccine development against monkeypox infection. Microb Pathog. 2023; 180: 106156.
[26] Poland GA, Kennedy RB, Tosh PK. Prevention of monkeypox with vaccines: a rapid review. Lancet Infect Dis. 2022; 22(12): e349-e58.
[27] Sah R, Paul D, Mohanty A, Shah A. Monkeypox (Mpox) vaccines and their side effects: the other side of the coin. Int J Surg. 2023; 109(2): 215-217.
[28] Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for Preexposure Vaccination of Persons at Risk for Occupational Exposure to Orthopoxviruses: Recommendations of the Advisory Committee on Immunization Practices - United States, 2022. MMWR Morb Mortal Wkly Rep. 2022; 71(22): 734-742.
[29] Gao F, He C, Liu M, et al. Cross-reactive immune responses to monkeypox virus induced by MVA vaccination in mice. Virol J. 2023; 20(1): 126.
[30] Rastogi A, Kumar M. Current Status of Vaccine Development for Monkeypox Virus. Adv Exp Med Biol. 2024; 1451: 289-300.
[31] WHO 2024. WHO adds MVA-BN vaccine to prequalification list as first vaccine against mpox. Available at: https://wwwwhoint/news/item/13-09-2024-who-prequalifies-the-first-vaccine-against-mpox.
[32] Shah M, Jaan S, Shehroz M, et al. Deciphering the Immunogenicity of Monkeypox Proteins for Designing the Potential mRNA Vaccine. ACS Omega. 2023; 8(45): 43341-43355.
[33] Begum JPS, Ngangom L. Emergence of monkeypox: a worldwide public health crisis. Hum Cell. 2023; 36(3): 877-893.
[34] Ejaz H, Junaid K, Younas S, et al. Emergence and dissemination of monkeypox, an intimidating global public health problem. J Infect Public Health. 2022; 15(10): 1156-1165.
[35] Vakaniaki EH, Kacita C, Kinganda-Lusamaki E. Sustained human outbreak of a new MPXV clade I lineage in eastern Democratic Republic of the Congo. Nat Med. 2024; 30(10): 2791-2795.
[36] Kinganda-Lusamaki E, Amuri-Aziza A, Fernandez-Nuñez N, et al. Clade I mpox virus genomic diversity in the Democratic Republic of the Congo, 2018-2024: Predominance of zoonotic transmission. Cell. 2025; 188(1): 4-14.e6.
[37] Happi C, Adetifa I, Mbala P, et al. Urgent need for a non-discriminatory and non-stigmatizing nomenclature for monkeypox virus. PLoS Biol. 2022; 20(8): e3001769.
[38] Beiras CG, Malembi E, Escrig-Sarreta R, et al. Concurrent outbreaks of mpox in Africa-an update. Lancet. 2025; 405(10472): 86-96.
[39] Van Dijck C, Hoff NA, Mbala-Kingebeni P, et al. Emergence of mpox in the post-smallpox era-a narrative review on mpox epidemiology. Clin Microbiol Infect. 2023; 29(12): 1487-1492.
[40] Acosta-España JD, Bonilla-Aldana DK, Luna C, Rodriguez-Morales AJ. The Resurgence of Mpox: A New Global Health Crisis. Infez Med. 2024; 32(3): 267-271.
[41] Antinori S, Casalini G, Giacomelli A, Rodriguez-Morales AJ. Update on Mpox: a brief narrative review. Infez Med. 2023; 31(3): 269-276.
[42] Guarducci G, Porchia BR, Lorenzini C, Nante N. Overview of case definitions and contact tracing indications in the 2022 monkeypox outbreak. Infez Med. 2022; 31(1): 13-19.
[43] Mucker EM, Freyn AW, Bixler SL, et al. Comparison of protection against mpox following mRNA or modified vaccinia Ankara vaccination in nonhuman primates. Cell. 2024; 187(20): 5540-5553.e10.
[44] WHO 2024. Multi-country outbreak of mpox, External situation report. Available at: https://wwwwhoint/publications/m/item/multi-country-outbreak-of-mpox--external-situation-report-44---23-december-2024.
[45] Kircheis R, Planz O. The Role of Toll-like Receptors (TLRs) and Their Related Signaling Pathways in Viral Infection and Inflammation. Int J Mol Sci. 2023; 24(7): 6701.
[46] Nichols DB, De Martini W, Cottrell J. Poxviruses Utilize Multiple Strategies to Inhibit Apoptosis. Viruses. 2017; 9(8): 215.
[47] Zhao Y, Lu Y, Richardson S, Sreekumar M, Albarnaz JD. TRIM5α restricts poxviruses and is antagonized by CypA and the viral protein C6. Nature. 2023; 620(7975): 873-880.
[48] Lum FM, Torres-Ruesta A. Monkeypox: disease epidemiology, host immunity and clinical interventions. Nat Rev Immunol. 2022; 22(10): 597-613.
[49] Suleman M, Ahmad T, Shah K, et al. Exploring the natural products chemical space to abrogate the F3L-dsRNA interface of monkeypox virus to enhance the immune responses using molecular screening and free energy calculations. Front Pharmacol. 2023; 14: 1328308.
[50] Zandi M, Shafaati M, Hosseini F. Mechanisms of immune evasion of monkeypox virus. Front Microbiol. 2023; 14: 1106247.
[51] Jiao P, Ma J, Zhao Y, et al. The nuclear localization signal of monkeypox virus protein P2 orthologue is critical for inhibition of IRF3-mediated innate immunity. Emerg Microbes Infect. 2024; 13(1): 2372344.
[52] Al-Musa A, Chou J, LaBere B. The resurgence of a neglected orthopoxvirus: Immunologic and clinical aspects of monkeypox virus infections over the past six decades. Clin Immunol. 2022; 243: 109108.
[53] Earl PL, Americo JL, Moss B. Natural killer cells expanded in vivo or ex vivo with IL-15 overcomes the inherent susceptibility of CAST mice to lethal infection with orthopoxviruses. PLoS Pathog. 2020; 16(4): e1008505.
[54] Song H, Josleyn N, Janosko K, et al. Monkeypox virus infection of rhesus macaques induces massive expansion of natural killer cells but suppresses natural killer cell functions. PLoS One. 2013; 8(10): e77804.
[55] Mehta K, Joshi M, Bhatt J, Kagathra N, Omar M. Enhancing the use of natural killer cells as a promising therapy against monkeypox: Mechanisms and implications. Health Sci Rep. 2023; 6(11): e1732.
[56] Johnston SC, Lin KL, Connor JH, Ruthel G, Goff A, Hensley LE. In vitro inhibition of monkeypox virus production and spread by Interferon-β. Virol J. 2012; 9: 5.
[57] Earl PL, Americo JL, Moss B. Lethal monkeypox virus infection of CAST/EiJ mice is associated with a deficient gamma interferon response. J Virol. 2012; 86(17): 9105-9112.
[58] Adugna A. Antigen Recognition and Immune Response to Acute and Chronic Hepatitis B Virus Infection. J Inflamm Res. 2023; 16: 2159-2166.
[59] Lu J, Xing H, Wang C, et al. Mpox (formerly monkeypox): pathogenesis, prevention, and treatment. Signal Transduct Target Ther. 2023; 8(1): 458.
[60] Arndt WD, Cotsmire S, Trainor K, et al. Evasion of the Innate Immune Type I Interferon System by Monkeypox Virus. J Virol. 2015; 89(20): 10489-10499.
[61] Fernández de Marco Mdel M, Alejo A, Hudson P, Damon IK, Alcami A. The highly virulent variola and monkeypox viruses express secreted inhibitors of type I interferon. Faseb j. 2010; 24(5): 1479-1488.
[62] Lucena-Neto FD, Falcão LFM, Vieira-Junior AS, Moraes ECS, David JPF, Silva CC. Monkeypox Virus Immune Evasion and Eye Manifestation: Beyond Eyelid Implications. Viruses. 2023; 15(12): 2301.
[63] Otter AD, Jones S, Hicks B. Monkeypox virus-infected individuals mount comparable humoral immune responses as Smallpox-vaccinated individuals. Nat Commun. 2023; 14(1): 5948.
[64] Ophinni Y, Frediansyah A, Sirinam S, et al. Monkeypox: Immune response, vaccination and preventive efforts. Narra J. 2022; 2(3): e90.
[65] Ahmed SF, Sohail MS. Vaccinia-Virus-Based Vaccines Are Expected to Elicit Highly Cross-Reactive Immunity to the 2022 Monkeypox Virus. Viruses. 2022; 14(9): 1960.
[66] Zhao R, Wu L, Sun J, et al. Two noncompeting human neutralizing antibodies targeting MPXV B6 show protective effects against orthopoxvirus infections. Nat Commun. 2024; 15(1): 4660.
[67] Asquith W, Hueston L, Dwyer D, et al. Characterizing the acute antibody response of monkeypox and MVA-BN vaccine following an Australian outbreak. J Med Virol. 2024; 96(1): e29407.
[68] Xia A, Wang X, He J, et al. Cross-reactive antibody response to Monkeypox virus surface proteins in a small proportion of individuals with and without Chinese smallpox vaccination history. BMC Biol. 2023; 21(1): 205.
[69] Li E, Guo X, Hong D, et al. Duration of humoral immunity from smallpox vaccination and its cross-reaction with Mpox virus. Signal Transduct Target Ther. 2023; 8(1): 350.
[70] Zhen Z, Zhang L, Li Q, et al. Cross-reactive antibodies against monkeypox virus exist in the population immunized with vaccinia Tian Tan strain in China. Infect Genet Evol. 2023; 113: 105477.
[71] Li M, Guo Y, Deng Y, et al. Long-lasting humoral and cellular memory immunity to vaccinia virus Tiantan provides pre-existing immunity against mpox virus in Chinese population. Cell Rep. 2024; 43(1): 113609.
[72] Golden JW, Hooper JW. Heterogeneity in the A33 protein impacts the cross-protective efficacy of a candidate smallpox DNA vaccine. Virology. 2008; 377(1): 19-29.
[73] Zaeck LM, Lamers MM. Low levels of monkeypox virus-neutralizing antibodies after MVA-BN vaccination in healthy individuals. Nat Med. 2023; 29(1): 270-278.
[74] Zhang F, Chai Z, Wang X, et al. Monkeypox: Can we count on the current smallpox immunization? Virology. 2024; 592: 109994.
[75] Isidro. J, Borges. V, Pinto. M, et al. Multi-country outbreak of Monkeypox virus: genetic divergence and first signs of microevolution. Available at: https://bitly/3Lz174F. 2022.
[76] Li M, Ren Z, Wang Y, et al. Three neutralizing mAbs induced by MPXV A29L protein recognizing different epitopes act synergistically against orthopoxvirus. Emerg Microbes Infect. 2023; 12(2): 2223669.
[77] Kupritz J, Pahwa S, Pallikkuth S. Serosurvey of Immunity to Monkeypox (Mpox) Virus Antigens in People Living with HIV in South Florida. Pathogens. 2023; 12(11): 1355.
[78] Yan H, Peng Y, Zhang J, et al. Rapid and highly potent humoral responses to mpox nanovaccine candidates adjuvanted by thermostable scaffolds. Vaccine. 2024; 42(8): 2072-2080.
[79] Colavita F, Matusali G. Profiling the acute phase antibody response against mpox virus in patients infected during the 2022 outbreak. J Med Virol. 2023; 95(6): e28851.
[80] Moraes-Cardoso I, Benet S, Carabelli J, et al. Immune responses associated with mpox viral clearance in men with and without HIV in Spain: a multisite, observational, prospective cohort study. Lancet Microbe. 2024; 5(8): 100859.
[81] Guo L, Song R, Zhang Q, et al. Profiling of viral load, antibody and inflammatory response of people with monkeypox during hospitalization: a prospective longitudinal cohort study in China. EBioMedicine. 2024; 106: 105254.
[82] Yang H, Xie X, Zeng M, et al. Clinical characteristics, viral dynamics, and antibody response of monkeypox virus infections among men with and without HIV infection in Guangzhou, China. Front Cell Infect Microbiol. 2024; 14: 1412753.
[83] Yang Y, Niu S, Shen C. Longitudinal viral shedding and antibody response characteristics of men with acute infection of monkeypox virus: a prospective cohort study. Nat Commun. 2024; 15(1): 4488.
[84] Yefet R, Friedel N, Tamir H, et al. Monkeypox infection elicits strong antibody and B cell response against A35R and H3L antigens. iScience. 2023; 26(2): 105957.
[85] Cohn H, Bloom N, Cai G, et al. Mpox vaccine and infection-driven human immune signatures. medRxiv. [Preprint]. 2023, 2023.03.07.23286701.
[86] Cohn H, Bloom N, Cai GY, et al. Mpox vaccine and infection-driven human immune signatures: an immunological analysis of an observational study. Lancet Infect Dis. 2023; 23(11): 1302-1312.
[87] Hubert M, Guivel-Benhassine F, Bruel T, et al. Complement-dependent mpox-virus-neutralizing antibodies in infected and vaccinated individuals. Cell Host Microbe. 2023; 31(6): 937-48.e4.
[88] Hudson PN, Self J, Weiss S, et al. Elucidating the role of the complement control protein in monkeypox pathogenicity. PLoS One. 2012; 7(4): e35086.
[89] Estep RD, Messaoudi I, O’Connor MA, et al. Deletion of the monkeypox virus inhibitor of complement enzymes locus impacts the adaptive immune response to monkeypox virus in a nonhuman primate model of infection. J Virol. 2011; 85(18): 9527-9542.
[90] Kennedy RB. Monkeypox infection creates immune signatures of disease progression. Lancet Infect Dis. 2023; 23(3): 265-266.
[91] Grüner E, Grossegesse M, Stern D, et al. Mpox-specific immune responses elicited by vaccination or infection in people living with HIV. J Infect Dis. 2024; 230(5): 1110-1119.
[92] Agrati C, Cossarizza A, Mazzotta V, et al. Immunological signature in human cases of monkeypox infection in 2022 outbreak: an observational study. Lancet Infect Dis. 2023; 23(3): 320-330.
[93] Mitjà O, Ogoina D, Titanji BK, et al. Monkeypox. Lancet. 2023; 401(10370): 60-74.
[94] Stefanie S, Koldehoff M, Schenk-Westkamp P, Horn PA, Esser S, Lindemann M. T Cell Responses against Orthopoxviruses in HIV-Positive Patients. Vaccines (Basel). 2024; 12(2): 131.
[95] Venturi C, Guadagno A, Varesano S, et al. Histopathologic and transmission electron microscopic findings in monkeypox cutaneous lesions. Infez Med. 2024; 32(1): 76-82.
[96] Harbour JC, Abdelbary M, Schell JB, et al. T helper 1 effector memory CD4(+) T cells protect the skin from poxvirus infection. Cell Rep. 2023; 42(5): 112407.
[97] Wellington D, Dong T. T cells are ready for the fight against monkeypox. Cell Host Microbe. 2022; 30(12): 1653-1654.
[98] Grifoni A, Zhang Y, Tarke A, et al. Defining antigen targets to dissect vaccinia virus and monkeypox virus-specific T cell responses in humans. Cell Host Microbe. 2022; 30(12): 1662-1670.e4.
[99] Song H, Sidney J, Wiseman RW, et al. Characterizing monkeypox virus specific CD8+ T cell epitopes in rhesus macaques. Virology. 2013; 447(1-2): 181-186.
[100] Adamo S, Gao Y, Sekine T, et al. Memory profiles distinguish cross-reactive and virus-specific T cell immunity to mpox. Cell Host Microbe. 2023; 31(6): 928-936.e4.
[101] Petruccioli E, Sbarra S, Vita S, Salmi A, Cuzzi G, De Marco P. Characterization of the Monkeypox Virus [MPX]-Specific Immune Response in MPX-Cured Individuals Using Whole Blood to Monitor Memory Response. Vaccines (Basel). 2024; 12(9): 964.
[102] Comeau K, Paradis P, Schiffrin EL. Human and murine memory γδ T cells: Evidence for acquired immune memory in bacterial and viral infections and autoimmunity. Cell Immunol. 2020; 357: 104217.
[103] Yang S, Wang Y, Yu F, et al. Structural and functional insights into the modulation of T cell costimulation by monkeypox virus protein M2. Nat Commun. 2023; 14(1): 5186.
[104] Hammarlund E, Dasgupta A, Pinilla C, Norori P, Früh K, Slifka MK. Monkeypox virus evades antiviral CD4+ and CD8+ T cell responses by suppressing cognate T cell activation. Proc Natl Acad Sci U S A. 2008; 105(38): 14567-14572.
[105] Johnston SC, Johnson JC, Stonier SW, et al. Cytokine modulation correlates with severity of monkeypox disease in humans. J Clin Virol. 2015; 63: 42-45.
[106] Tolomeo M, Cascio A. STAT4 and STAT6, their role in cellular and humoral immunity and in diverse human diseases. Int Rev Immunol. 2024; 43(6): 394-418.
[107] Troy JD, Hill HR, Ewell MG, Frey SE. Sex difference in immune response to vaccination: A participant-level meta-analysis of randomized trials of IMVAMUNE smallpox vaccine. Vaccine. 2015; 33(41): 5425-5431.
[108] Kennedy RB, Ovsyannikova IG, Pankratz VS, et al. Gender effects on humoral immune responses to smallpox vaccine. Vaccine. 2009; 27(25-26): 3319-3323.
[109] Haralambieva IH, Ovsyannikova IG, Kennedy RB, Larrabee BR, Shane Pankratz V, Poland GA. Race and sex-based differences in cytokine immune responses to smallpox vaccine in healthy individuals. Hum Immunol. 2013; 74(10): 1263-1266.
[110] Wang Y, Yang K, Zhou H. Immunogenic proteins and potential delivery platforms for mpox virus vaccine development: A rapid review. Int J Biol Macromol. 2023; 245: 125515.
[111] Zuiani A, Dulberger CL, De Silva NS, et al. A multivalent mRNA monkeypox virus vaccine (BNT166) protects mice and macaques from orthopoxvirus disease. Cell. 2024; 187(6): 1363-73.e12.
[112] Zeng J, Li Y, Jiang L, et al. Mpox multi-antigen mRNA vaccine candidates by a simplified manufacturing strategy afford efficient protection against lethal orthopoxvirus challenge. Emerg Microbes Infect. 2023; 12(1): 2204151.
[113] Hou F, Zhang Y, Liu X. mRNA vaccines encoding fusion proteins of monkeypox virus antigens protect mice from vaccinia virus challenge. Nat Commun. 2023; 14(1): 5925.
[114] Cotter CA, Ignacio MA, Americo JL, Earl PL, Mucker EM. Mpox mRNA-1769 vaccine inhibits orthopoxvirus replication at intranasal, intrarectal, and cutaneous sites of inoculation. NPJ Vaccines. 2024; 9(1): 256.
[115] Fang Z, Monteiro VS. Polyvalent mRNA vaccination elicited potent immune response to monkeypox virus surface antigens. Cell Res. 2023; 33(5): 407-410.