Le Infezioni in Medicina, n. 1, 15-28, 2025
doi: 10.53854/liim-3301-3
REVIEWS
Maternal and foetal outcomes in women with gestational Dengue: A systematic review
Yanet Vélez Jaramillo1, Marco Antonio Reveiz Montes2,3, Johana Patricia Galván-Barrios4, Yelson Alejandro Picón-Jaimes5
1Sub Secretary of Health Protection and Service Provision - District Public Health Secretariat, Cali, Colombia;
2University of Costa Rica, San José, Costa Rica;
3Cali Clinic, Santiago de Cali, Colombia;
4Biomedical Scientometrics and Evidence-Based Research Unit -Department of Health Sciences - Universidad de la Costa, Barranquilla, Colombia;
5Blanquerna-Faculty of Health Sciences- Ramon Llull University, Barcelona, Spain.
Article received 3 October 2024 and accepted 9 January 2025
Corresponding author
Yelson Alejandro Picón-Jaimes
E-mail: ypicon@unab.edu.co
SummaRY
Introduction: Dengue is a mosquito-borne viral disease. It has been associated with high maternal and foetal morbidity and mortality. Therefore, this study aimed to describe the outcomes of Dengue infection in pregnant women in terms of maternal bleeding, miscarriage, preterm delivery, severe Dengue, Dengue shock and maternal mortality, as well as foetal outcomes in terms of foetal distress, low birth weight and neonatal mortality.
Methods: This systematic review followed PRISMA guidelines and was PROSPERO-registered (CRD42024578212). It examined publications from 2019-2024 across major databases, including Medline, Scopus, Web of Science, Scielo, and CENTRAL. The selection process utilised Rayyan AI® for duplicate removal, followed by a two-reviewer screening system. The two reviewers initially filtered the papers by title and, then, by abstract; finally, they read the full text and chose the articles to synthesise. The same two reviewers performed data extraction independently using a Microsoft Excel® matrix. For the risk of bias assessment and Quality evaluation, the ROBINS-E tool and the STROBE guidelines were employed.
Results: Maternal Dengue was associated with an increased risk of preterm delivery and complications such as obstetric bleeding, especially postpartum haemorrhage, which was a complication that reached proportions of up to 25% in some studies.
Severe Dengue and Dengue shock were also complications present in these women; thrombocytopenia was the most common sign, and complications such as pre-eclampsia and multi-organ dysfunction appeared, leading to fatal outcomes such as maternal and foetal mortality.
Conclusions: Dengue infection during gestation carries significant maternal health risks, including complications such as bleeding, miscarriage and preterm delivery. In addition, it is associated with foetal distress and low birth weight, as well as increased foetal and neonatal mortality, highlighting the need for vigilance and appropriate medical care.
Keywords: Dengue, severe dengue, pregnancy, maternal mortality, foetal mortality.
INTRODUCTION
Dengue is a viral disease transmitted by mosquitoes and is prevalent in tropical and subtropical regions, with nearly half of the global population at risk [1-3]. The World Health Organization 2024 estimated that between 100 and 400 million infections occurred annually, resulting in an economic burden exceeding 8.9 billion dollars annually [3-6].
The dengue virus (DENV), a positive-strand RNA virus from the Flaviviridae family, has a nucleocapsid of about 50 nanometres and a genome length of 11 kb, encoding three structural and seven non-structural proteins, including NS1, which is linked to severe complications [7-10]. Dengue can be asymptomatic, with only 20% of cases showing symptoms, primarily fever, intense headaches, joint and muscle pain, skin rashes, and nausea [11-13].
The disease progresses through three phases: febrile, critical (where severe dengue or dengue shock may occur), and resolution [14-15]. Severe dengue predominantly affects children under 15 years old and individuals with pre-existing antibodies to any of the four DENV serotypes, a phenomenon known as ‘antibody-dependent enhancement’. This situation occurs because these antibodies can facilitate the entry of the virus into cells, leading to a more severe immune response. When someone previously infected with one serotype encounters a different serotype, the antibodies produced during the first infection may not effectively neutralise the new virus. Instead, they can bind to it and enhance its uptake by immune cells, resulting in increased viral replication and a heightened risk of severe illness. This mechanism contributes to the severity of Dengue in susceptible populations, particularly in children, who may have less robust immune responses [14-15].
Pregnant women face heightened risks, including significant bleeding and severe forms of the disease [16]. They are more susceptible to dengue-induced shock, and physiological changes during pregnancy, such as hypotension and increased heart rate, can obscure dengue symptoms, complicating diagnosis [15-17]. Increased capillary permeability may lead to acute pulmonary oedema if hyperhydration is not properly managed during the critical phase. Thus, distinguishing between dengue symptoms and normal pregnancy changes is essential [17-19].
This systematic review aims to detail the symptoms of dengue infection in pregnant women, focusing on maternal and foetal outcomes.
MATERIALS AND METHODS
This systematic review adhered to the PRISMA guidelines and was registered in PROSPERO under the identifier CRD42024578212.
Eligibility criteria
Clinical trials (randomized and non-randomized) and observational studies (cross-sectional, cohort, or case-control) were included. Systematic, exploratory, and narrative reviews, as well as clinical cases or case series, were excluded.
Sources of information
The review covered studies published between 2019 and 2024, utilizing databases such as Medline (PubMed), Scopus, Web of Science, and Scielo, along with the Cochrane Central Register of Controlled Trials (CENTRAL), following Cochrane guidelines [20]. The search formulas are summarized in Table 1.
Table 1 - Search formulas in the different databases and clinical trial registries.

Study selection process
Search results were processed using Rayyan AI®, where the AI removed duplicates. Two independent reviewers assessed the titles’ relevance in the initial screening and further evaluated articles meeting the initial criteria through their abstract. The reviewers read the selected manuscripts fully after the second screening to confirm eligibility and explore references for additional relevant studies.
Data extraction process
Data were synthesized in a Microsoft Excel® matrix, capturing variables like authors, publication year, geographic location, study design, follow-up duration, patient count, and participant age, alongside maternal and foetal outcomes. Two reviewers independently extracted data, resolving discrepancies through discussion. In cases where differences in opinion arose between the authors, both parties revisited the article and engaged in thorough discussions to reach a consensus. This collaborative approach ensured that all perspectives were considered, ultimately leading to a resolution of the discrepancies. The corresponding authors of the papers were also contacted for any missing information to enhance the completeness and accuracy of the data collected.
Assessment of risk of bias
The risk of bias was evaluated using the ROBINS-E tool from Cochrane® [20]. Two reviewers assessed studies independently, with disagreements resolved by consensus.
Synthesis methods
A narrative synthesis summarized the extracted information, detailing outcomes aligned with the review’s objectives and presenting key findings from each manuscript.
Assessment of publication bias
The authors checked compliance with STROBE guidelines to evaluate the certainty of the evidence. Discrepancies in assessments were resolved by consensus, with scores classified as high (≥70%), moderate (40%-69%), or low (<40%) quality.
RESULTS
Selection of studies
Searches were performed in various databases, obtaining 334 records in Medline, 553 in Scopus, 366 in Web Of Science, 50 in Scielo and 10 in CENTRAL, and two manuscripts by reverse search. One thousand three hundred and fifteen manuscripts were imported into Rayyan AI®, eliminating duplicates and leaving 750 texts. After a review by title, 450 were discarded, leaving 300 for reading abstracts. Of these, 250 did not meet the inclusion criteria, and 50 were read in complete text. Of these last 50 manuscripts, 34 were eliminated: 24 for addressing other febrile diseases and 10 for treating fever in pregnancy without considering relevant outcomes. Finally, 16 papers were included for the final analysis. The diagram summarizing the screening process according to the PRISMA methodology is shown in Figure 1.

Figure 1 - Diagram according to PRISMA methodology that summarizes the screening process of the manuscripts considered for this systematic review.
Characteristics of the studies
Regarding the typology of the manuscripts, all were observational. The studies by Kallur et al., Sondo et al., Thiyagalingam et al., Escobar et al., and Brar et al. were cross-sectional [21-25]. The studies by Nujum et al., Naik et al., Gupta et al. and Mewada et al. were prospective cohort studies [26-29]. Meanwhile, the studies by Singkibutr et al., Tougma et al., Mubashir et al. and Sagili et al. were retrospective cohort studies [30-33].
The study by Paixão et al. was a population-based cohort study; Mulyana et al. was a prospective longitudinal study with follow-up to six weeks postpartum, and Ortiz-Mesina et al. was a case-control study [34-36].
The total number of patients included in the 16 studies was 18,359 with Dengue and 16,740,584 without Dengue. The participants’ ages ranged between 19 and 43. Table 2 summarizes the main characteristics of the studies.
Table 2 - Characteristics of the studies included in the review.
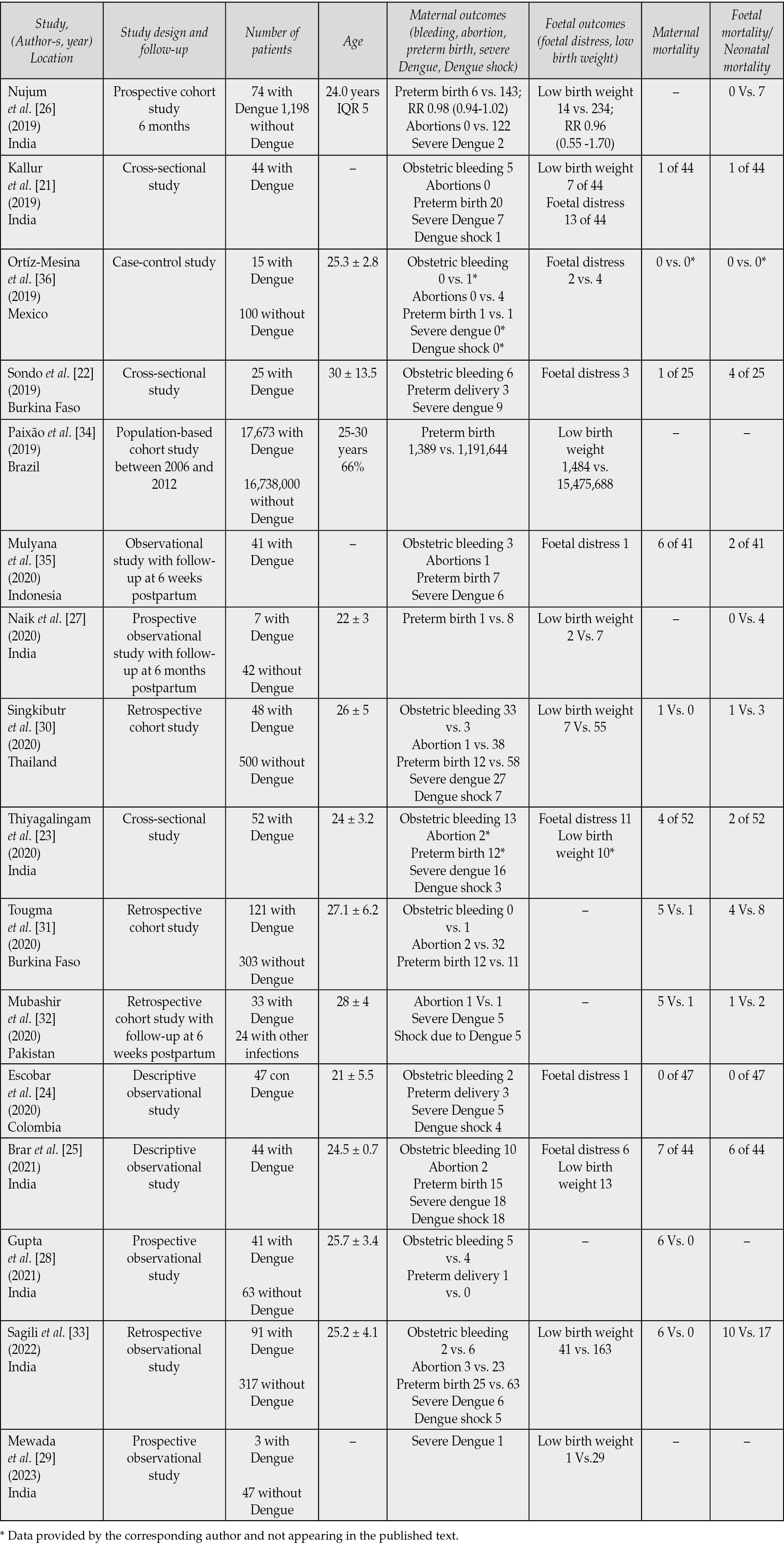
Risk of bias of individual studies
The risk of bias was analysed with the ROBINS-E tool and is presented in Figure 2. This tool consists of seven domains that identify the risk of different types of bias, such as confusion, selection, and classification. In this case, six observational studies were classified as “with some concerns about the risk of bias” in their total score.
Figure 2 - Analysis of risk of bias in observational studies according
to the ROBINS-E tool of the Cochrane collaboration.
Results of individual studies
1. Obstetric bleeding
In the study by Kallur et al., five cases of bleeding in the form of postpartum haemorrhage were identified, something similar to those reported by Thiyagalingam et al., where postpartum haemorrhage affected 13 of the patients, which represented 25% of the population reviewed [21,23]. Two of these patients required massive transfusion of blood products. According to Brar et al., out of a total of 40 women who gave birth, 10 (25%) experienced postpartum haemorrhage [25]. Of these, three women needed uterine tamponade with a Bakri balloon, one woman required drainage of vulvar hematoma, and the rest were treated with pharmacological methods. Other authors, such as Escobar et al. and Sagili et al., also reported two cases of postpartum haemorrhage in each of their studies, with the difference that Sagili et al. also reported six postpartum bleedings in patients without Dengue; but the difference was not significant (2.5% vs. 2.0%; p = 0.76) [24, 33].
Sondo et al. described five cases of intrapartum obstetric haemorrhage and one postpartum, indicating that 25% of the patients had thrombocytopenia, with platelet counts of less than 50,000/mm³, for which they received platelet transfusion [22]. On the other hand, Mulyana et al. observed two cases (4.9%) of postpartum haemorrhage and one case of placental abruption haemorrhage that ended in emergency caesarean section [35]. Additionally, in the study by Singkibutr et al., 36 cases of postpartum haemorrhage were reported, corresponding to 6.6% of the analysed cohort, of which 33 cases occurred in women with dengue and three in women without the infection [30].
2. Spontaneous abortions
The study by Ortiz-Mesina et al. did not identify cases of abortions in patients with Dengue; the multivariate analysis adjusted for obstetric risk factors found that neither Dengue infection nor obstetric history was associated with abortion (p = 0.129) [36].
Singkibutr et al. found 39 spontaneous abortions, corresponding to 7.1% of the cohort, of which one (2.1%) occurred in a woman with Dengue. In contrast, the other 38 (7.6%) occurred in women without Dengue, although there were no differences between the groups (p = 0.23) [30]. In the study by Tougma et al., two cases of spontaneous abortion were identified, corresponding to 1.6% of the total number of patients with Dengue and 32 cases (10.5%) in those without the infection [31].
Also, Mubashir et al. recorded one abortion in the group of patients with Dengue and one abortion in patients without Dengue [32]. Brar et al. reported two spontaneous abortions, corresponding to 4.5% of the total number of pregnancies with Dengue and Sagili et al. reported three spontaneous abortions among patients with Dengue and 23 in the control group (3.3 vs 7.2), but the difference was not significant p = 0.15 [25, 33].
3. Preterm birth
Preterm birth and associated complications were investigated in several studies regarding maternal Dengue. Nujum et al. reported an RR of 8.08 (95% CI: 4.34 to 15.09) for preterm birth, premature rupture of membranes, and meconium appearance [26]. Paixão et al. noted a slight increase in the risk of premature birth associated with maternal Dengue, rising from 7.3% to 7.9% (RR: 1.1, 95% CI: 1.0 to 1.2) [34]. Severe Dengue significantly increased this risk, escalating rates from 7.3% to 15.2% (OR: 2.4, 95% CI: 1.3 to 4.4), particularly within the first ten days of illness (RR: 2.0, 95% CI: 1.6 to 2.6).
Naik et al. found nine premature birth cases, with only one linked to Dengue, showing no significant difference (p > 0.05) [27]. Contrastingly, Singkibutr et al. reported a significant prevalence of premature births in Dengue-infected pregnant women (25% vs. 11.6%, p = 0.008), with logistic regression indicating an OR of 2.1 (95% CI: 1.0 to 4.4; p = 0.049) for premature birth due to Dengue [30].
Tougma et al. identified 12 preterm births (9.9%) among Dengue patients compared to 11 cases (3.6%) in non-infected patients, with a statistically significant difference (p = 0.016) [31]. Sondo et al. documented three preterm births (12%) leading to fatalities, while Escobar et al. reported three premature births, two requiring hospitalization and presenting neonatal complications [22, 24].
Brar et al. found that 15 premature babies (37.5%) were born to Dengue-positive mothers, with three cases in severely ill women, including one delivery via caesarean section due to placental abruption [25]. Finally, Sagili et al. noted a higher proportion of premature births among Dengue-infected women (32.1%) compared to controls (22.7%) [33]. However, this difference was not statistically significant (p = 0.08), and it presented an RR of 1.33 (95% CI: 0.89 to 1.97).
4. Severe Dengue
Singkibutr et al. found 27 cases of severe Dengue, representing 56.3% of the patients with Dengue. Brar et al. reported 18 cases, of which eight had acute kidney injury, and four with liver failure [25, 30].
Thiyagalingam et al. identified 16 women with severe Dengue, with thrombocytopenia as the most common warning sign [23]. A platelet count < 20,000/mm³ was reported in seven women, with the majority requiring platelet transfusions. In addition, 46.2% of the pregnant women had abnormal liver function tests. Severe preeclampsia was observed in eight women, with HELLP syndrome in six of them.
Sondo et al. reported that nine women (36%) had severe Dengue, with elevated AST levels [22]. Kallur et al. documented seven cases of severe Dengue with platelets < 100,000/mm³ and elevated liver enzymes; additionally, three cases presented acute pulmonary oedema, encephalitis and acute kidney injury [21].
Other studies reported low levels of severe Dengue: Sagili et al. described six cases, Mubashir et al. and Escobar et al. reported five cases each, Nujum et al. documented two cases, and Mewada et al. a single case of severe Dengue [24, 26, 29, 32, 33].
5. Dengue-induced shock
In the study by Brar et al., 18 women had a shock in the context of Dengue infection, of which seven died, all with platelet levels below 100,000/mm3 and elevated liver enzyme levels [25]. On the other hand, Singkibutr et al. found seven cases of Dengue-induced shock, corresponding to 14.6% of patients with Dengue [30]. Also, Mubashir et al. reported five cases of Dengue-induced shock in patients who eventually died [32].
Sagili et al. also reported 5 cases of Dengue shock who died. Similarly, in the study by Escobar et al., four patients had acute respiratory distress syndrome in the context of capillary leak [24, 33]. Additionally, in the study by Thiyagalingam et al., three women experienced multiple organ dysfunction syndrome secondary to dengue [23]. Finally, the study by Kallur et al. reported a single case of Dengue-induced shock among 44 patients who had the infection [21].
6. Foetal distress
Acute foetal distress was a condition reported by Nujum et al. in pregnant women with Dengue, with an RR of 8.08 95% CI (4.34 to 15.09), compared to those who did not have the infection [26]. For their part, Kallur et al. mention that the rate of caesarean sections in their work was 84% (n = 37), with foetal distress being one of the leading causes, with 35% (n = 13) of the cases [21]. Consequently, Brar et al. reported that 35% of pregnancies were terminated by caesarean sections, of which six (13.6%) were due to foetal distress [25].
Additionally, Thiyagalingam et al. indicated a caesarean section rate of 44.2% (n = 23), and the most common indication for caesarean section was foetal distress (n = 11) [23]. On the other hand, Mulyana et al. reported five patients (12.2%) who underwent emergency caesarean section due to various complications, one of them due to foetal distress, another due to placental abruption, and two with decreased platelet count and one with respiratory complications due to pneumonia [35].
In the work of Ortiz-Mesina et al., 13% of cases of foetal distress were identified in pregnant women with Dengue vs. 4% in those without the infection [36]. The specific risk of presenting foetal distress due to exposure or not to the infection was analysed, and no significant association could be found between viral exposure and the outcome (p >0.05).
7. Low birth weight
In the study by Paixão et al., maternal Dengue was associated with a slight increase in the risk of low birth weight (7.2% vs. 8.4%), with a RR 1.2; 95% CI (1.1 to 1.2) [34]. This risk increased even more in mothers with severe Dengue, as evidenced by an RR of 2.1 with 95% CI (1.1 to 4.0) [34].
The other studies did not find significant differences between the weights of children of mothers with and without Dengue. For example, Nujum et al. described 14 neonates with low birth weight in mothers with Dengue vs. 234 in mothers without Dengue; RR 0.96 with 95% CI (0.55 to 1.70) [26]. Naik et al. also identified nine patients with low birth weight, of which two were children of patients with Dengue; OR of 1.9 with 95% CI (0.1 to 16.8) [27].
In the study by Sagili et al., 41 neonates with low birth weight were documented among mothers with Dengue and 163 in those without the disease; the risk of low birth weight associated with Dengue had a RR of 1.13, with 95% CI (0.87 to 1.45) [33]. Finally, Singkibutr et al. reported that low birth weight had no statistically significant differences between children of mothers with and without Dengue (p = 0.454) [30].
8. Maternal mortality
Seven women (15.9%) died in the work reported by Brar et al. [25]. All the women who died had severe Dengue with a platelet count of less than 100,000/mm³ and liver transaminases exceeding twice the standard limit [25]. Among the seven cases of maternal deaths, five women presented with gestations beyond the age of foetal viability, and all these women had live births without neonatal complications; also, four of these women had required transfusions of blood products [25].
In the study by Gupta et al., there were six maternal deaths in pregnant women with Dengue, with a higher probability of mortality than in patients without Dengue with an OR of 19 and 95% CI (1.01 to 357.10) [28]. Sagili et al. found six maternal deaths among the group of patients with Dengue, five of them due to Dengue shock syndrome and one due to severe Dengue [33]. Likewise, four of the 52 women in the study by Thiyagalingam et al. died from complications associated with Dengue, three of them with severe Dengue and one with Dengue with warning signs [23]. Two were in the third trimester of pregnancy, and two were in the first days postpartum [23]. All four patients had a platelet count below 30,000/mm3; three had preeclampsia, and one had placental abruption [23].
In the study by Mubashir et al., five maternal deaths were reported in the group of patients with Dengue and one death among those without Dengue, a difference that was not significant (p = 0.98) [32]. Similarly, Tougma et al. reported five deaths (4.1%) in the group with Dengue infection and one death (0.3%) in the group without the disease, a difference that was significant p = 0.008. Furthermore, the probability of maternal death was higher among patients with viral infection, with an OR of 2.09 with a 95% CI (1.08 to 4.05) [31].
9. Foetal mortality/neonatal mortality
In the study by Sagili et al., eight stillbirths and two neonatal deaths were found in patients with Dengue, while in patients without Dengue, there were seven neonatal deaths and ten stillbirths [33]. The risk of foetal death was higher among patients with the disease, with an RR of 2.67 with a 95% CI (1.09 to 6.57).
Intrauterine foetal deaths in the study by Tougma et al. were higher in the group of patients with Dengue; in fact, there were four cases (3.3%), and in patients without the disease, there were eight cases (2.6%), this difference was not significant (p > 0.05) [31]. Likewise, Sondo et al. reported four cases of neonatal mortality, of which three were due to prematurity and one to severe infection; all cases occurred in children of patients with Dengue [22].
In the study by Mulyana et al., two cases (4.87%) of intrauterine foetal death were documented among patients with Dengue, which was similar to that reported by Thiyagalingam et al., where two stillbirths were recorded. The mothers of these stillbirths had severe forms of Dengue and had complications such as preeclampsia and placental abruption [23, 35].
In the work by Singkibutr et al., no differences were found in neonatal or foetal mortality between the products of pregnancy of women with and without Dengue (p = 0.30); however, there was one neonatal death among patients with Dengue and three in the group of patients without Dengue [30]. Finally, Naik et al. identified four deaths that occurred in children of women without Dengue, one case of foetal death and three deaths recorded during follow-up after birth. This situation was not associated with Dengue (p > 0.05) [27].
Publication bias and certainty of evidence
We checked the percentage of compliance in writing manuscripts according to the STROBE template. It was estimated that in order to consider the studies sufficient quality, they had to meet a minimum of 70% of the recommendations. See Figure 3.
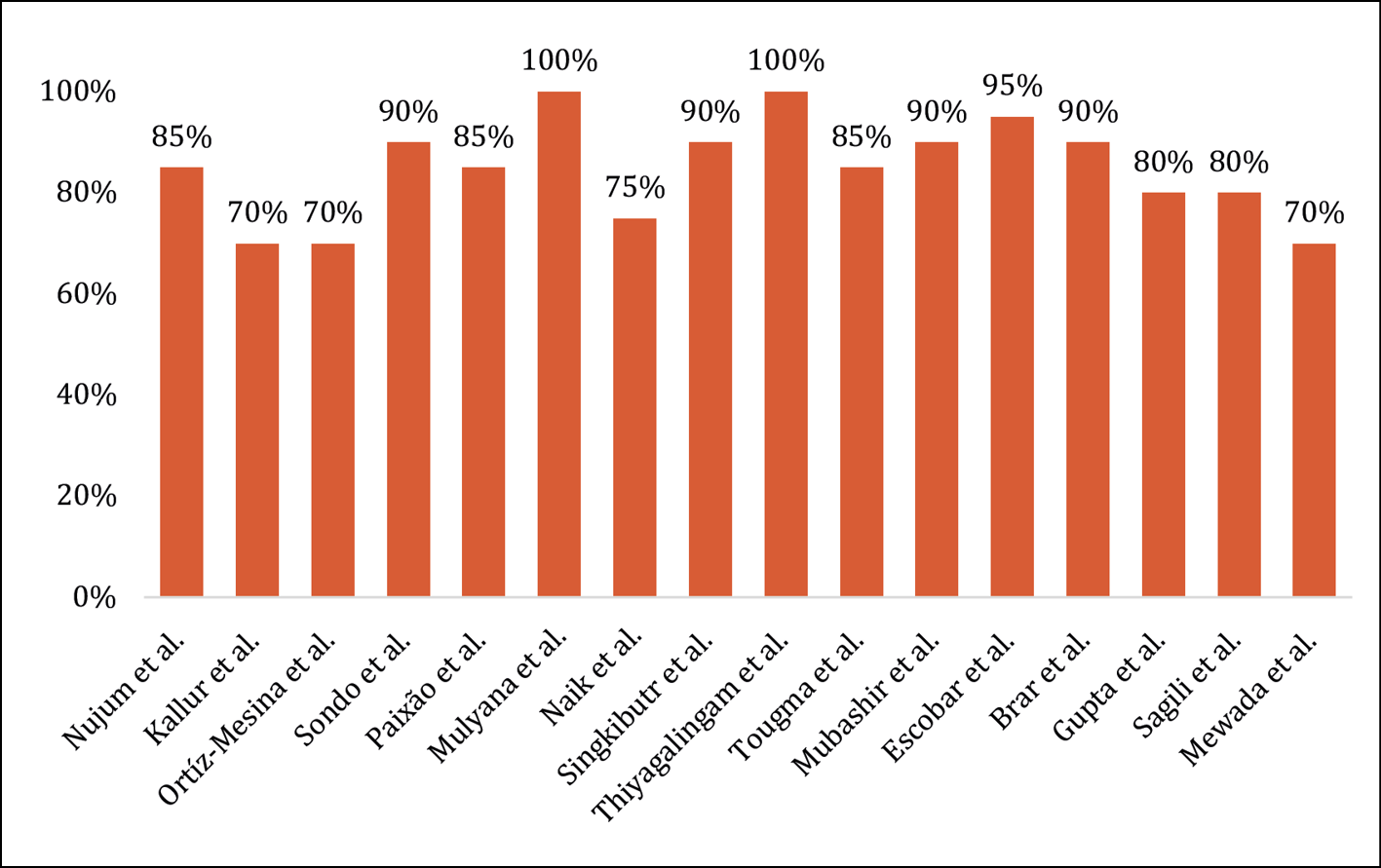
Figure 3 - Percentage of total compliance with the STROBE tool.
DISCUSSION
Bleeding rates in the studies ranged from 6% to 25%, with most of these events being postpartum haemorrhages, spontaneous vaginal bleeding, or placental abruption [21–25]. Studies such as that by Thiyagalingam et al. indicated that some patients required massive transfusions of blood products and additional treatments, such as those described by Brar et al., which included intrauterine balloon tamponades and hematoma drainage [23, 25]. On the other hand, Sagili et al. and Gupta et al. confirmed no significant differences in bleeding rates between women with and without dengue infection [28, 33]. A 2022 meta-analysis also corroborated the lack of significant association between Dengue virus infection during pregnancy and obstetric bleeding, with an OR of 2.79, with a 95% CI (0.86 to 9.07; I2 = 61%) [37]. However, other studies, such as that of Machain-Williams et al., reported obstetric haemorrhage in pregnant women with Dengue and severe Dengue [38]. Likewise, the work of Sharma et al. showed haemorrhagic manifestations in seven pregnant women with Dengue, of which three experienced postpartum haemorrhage, highlighting one case of Dengue-induced shock [39]. The variation in results across these studies may stem from differences in study design, sample size, and the specific populations examined, which can influence the observed outcomes and the generalisability of the findings.
Regarding spontaneous abortion in women with Dengue, several studies, such as those of Ortíz-Mesina et al., Sagili et al., and Singkibutr et al., found no relationship between Dengue infection and the risk of spontaneous abortions, with p values greater than 0.05 [30, 33, 36]. In line with this, the meta-analysis by Rathore et al. also reported that there was no significant association between Dengue infection in pregnancy and spontaneous abortions, with an OR of 0.64, with 95% CI (0.20 to 1.98) and a heterogeneity of I² = 57% [37]. On the other hand, the 2017 meta-analysis by Xiong et al. showed a pooled OR of 1.77 with 95% CI (0.99 to 3.15) and low heterogeneity (I² = 17.5%), suggesting that there was also no significant association between maternal Dengue infection and spontaneous abortion [40]. However, Paixão et al. meta-analysis reported an OR of 3.51 with 95% CI (1.15 to 10.77) and a heterogeneity of I² = 0%, indicating a statistically significant association with Dengue [41]. A study by Sinha and Datta found that the incidence of spontaneous abortions in women with and without Dengue was 33.3%, reaching 71.4% in those with less than 12 weeks of gestation [42]. Risk factors such as gestational age of less than 12 weeks (p = 0.003) and decreased platelet count in the context of severe Dengue (p = 0.03) were identified [42]. The differences in findings regarding the relationship between Dengue infection and the risk of spontaneous abortions can be attributed to the timing of the infection during pregnancy, as early infections may have different implications than those occurring later. Variations in methodologies for assessing spontaneous abortion rates and differing definitions of what constitutes an abortion further contribute to the inconsistencies in reported associations.
The third outcome analysed was the relationship between preterm delivery and dengue infection. The study by Nujum et al. revealed that preterm delivery and premature rupture of membranes were associated with Dengue, with an RR of 8.08, p < 0.05 [26]. Paixão et al. found that maternal Dengue increased the risk of preterm delivery, especially in severe cases (OR of 2.4, p < 0.05) [34]. The meta-analysis by Paixão et al. showed an overall crude OR of 1.71 for the association between Dengue and premature birth, with a 95% CI (1.06 to 2.76) and heterogeneity of I² = 56.1%, this confidence interval being borderline. In other meta-analyses, such as those by Xiong et al. and Rathore et al., no significant association was found (p >0.05) [37, 40, 41].
For the fourth outcome, severe Dengue in pregnant women was analysed. Romero-Machado et al. found that severe Dengue affected 45.5% of pregnant women compared to 17.5% of non-pregnant women [43]. In the study by Machain-Williams et al., of 82 patients with confirmed infections, 15.9% presented severe Dengue [38]. In contrast, Annan et al. studied 94,832 women in Mexico [44]. They found that only 1.8% of pregnant women presented severe Dengue, similar to what was reported in our systematic review, where 96 cases of severe Dengue were reported among 18,359 women, representing 0.52%. In addition, it was highlighted that severe Dengue can be confused with HELLP syndrome, which is characterized by thrombocytopenia and elevated liver enzymes [45-47]. Machain-Williams et al. reported that 92.3% of patients with severe Dengue presented thrombocytopenia and elevations in AST and ALT [38]. The overlap of symptoms between severe Dengue and HELLP syndrome can complicate the diagnosis, especially in pregnancies close to term, as pointed out by Patnaik et al. and Harde et al., since both conditions can present alterations in liver function and haemorrhagic symptoms, which makes their differentiation difficult [48, 49]. Healthcare providers should be vigilant in differentiating between severe Dengue and HELLP syndrome, especially in late pregnancy, to ensure timely and appropriate management. Regular liver function and platelet count assessments may be recommended to identify complications early.
The fifth outcome analysed was Dengue-induced shock, with 43 cases among 18,359 patients, representing a proportion of 0.23% (95% CI: 0.17% to 0.32%). In comparison, Agarwal et al. reported a proportion of 14.5%, associating this shock with more significant maternal morbidity and mortality [50]. Likewise, the meta-analysis by Rathore et al. found a prevalence of 14.9% among 418 participants, with moderate heterogeneity (I² = 73%, p < 0.0001) [37]. Dengue shock is influenced by physiological factors of pregnancy, such as increased blood volume and heart rate, which may make pregnant women more susceptible to hypotension and circulatory collapse in case of plasma loss [37, 51, 52]. In addition, changes in peripheral vascular resistance, driven by oestrogens and nitric oxide, contribute to this susceptibility [50, 53]. Capillary permeability induced by Dengue virulence factors, especially the NS1 protein, has a more severe effect on pregnant women, resulting in more severe disease [54, 55]. The pregnant woman’s immune system also adapts to protect the foetus, which may decrease the effectiveness of the immune response against Dengue [56, 57]. This state of immunological tolerance allows the development of the foetus without being attacked by the maternal immune system but also reduces the response against external pathogens [10, 19, 58]. These modifications, together with cardiovascular changes, increase the severity of DENV disease and the risk of serious complications, including shock [59, 60]. The discrepancies in reported rates of Dengue-induced shock may be attributed to differences in study populations, methodologies, and definitions of shock, which can lead to variations in how cases are identified and reported.
Foetal stress was identified as the sixth outcome in the review, showing a strong association with gestational Dengue, with an RR of 8.08 (95% CI: 4.34 to 15.09) [26]. The caesarean section rate was high, reaching up to 84%, with foetal stress being the cause in 35% of cases [21]. According to López-Barroso et al., Dengue was related to foetal stress in 10% of cases of infected patients, being a significant cause of dystocic delivery [61]. In the study by Machain-Williams et al., foetal stress was reported in five women with severe Dengue, representing 6% of the total treated, all requiring emergency caesarean sections [38]. Romero-Angarita et al. documented that caesarean sections were performed in four patients due to acute foetal stress [62]. The seventh outcome analysed was low birth weight; Paixão et al. showed that maternal Dengue, especially severe, increases the risk of low birth weight, with a RR of 2.1 (p <0.05) [34]. However, other studies, such as those by Nujum et al. and Naik et al., found no significant differences in the birth weight of new-borns of mothers with and without Dengue [26, 27]. The meta-analysis by Paixão et al. reported that, although low birth weight was a common adverse outcome, the crude OR was 1.41 (95% CI: 0.90 to 2.21), which was not statistically significant [41]. Likewise, the meta-analysis by Xiong et al. showed no association between Dengue and low birth weight, with a RR of 0.99 (95% CI: 0.87 to 1.12) [40].
Regarding maternal mortality, one study found that 15.9% of pregnant women with severe Dengue died [25]. All of the deceased had low platelet counts and high transaminase levels [25]. Other studies reported several maternal deaths associated with Dengue, with a significantly higher probability of mortality compared to women without the disease. Complications leading to these deaths included Dengue shock syndrome and preeclampsia [23, 28, 31-33]. The findings of the review indicated that maternal mortality was higher in women with Dengue, underscoring the severity of the disease during pregnancy. The study by Rathore et al. identified that DENV infection was associated with an increased risk of maternal mortality, with an OR of 4.14 and a 95% CI (1.17 to 14.73) [37]. In a cohort study in Brazil by Feitoza et al., two maternal deaths were recorded in the dengue-exposed cohort, one of which was directly related to the disease; in contrast, there were no deaths in the unexposed cohort (p = 0.040) [63]. Despite discrepancies between studies, most maintain that mortality is higher in pregnant women with viral infection, especially at the end of pregnancy [64–66]. Maternal mortality associated with Dengue is a significant problem, especially in endemic regions [64-66]. Pregnant women with severe Dengue are at high risk of complications and mortality, and the management of these patients is complicated by the lack of effective antiviral treatments, highlighting the need for epidemiological surveillance and adequate medical care in suspected or confirmed cases of the disease [10, 17, 19].
The last outcome considered was foetal and neonatal mortality. It was identified that, although there were differences in the results that seemed to link Dengue with this mortality, not all were significant [27, 30, 31]. In the meta-analysis of Xiong et al., it was reported that the association between maternal Dengue infection and foetal death was not significant, with an RR of 3.42 and a 95% CI (0.76 to 15.49) [40]. However, Rathore et al. found a significant association between gestational Dengue and neonatal mortality, with an OR of 3.03 and a 95% CI (1.17 to 7.83) [37].
According to the study by Feitoza et al., neonatal mortality was higher in the cohort exposed to Dengue, with a three-fold higher risk (RR = 3.4;
p <0.05) [63]. This risk was increased in deaths occurring during the early neonatal period, with an RR of 6.8 (p <0.05).
Despite the synthesis work in this systematic review, essential limitations are highlighted. The heterogeneity of the studies makes it difficult to compare results due to variations in populations, diagnostic methods and inclusion criteria. In addition, the data quality can be inconsistent, affecting the validity of the conclusions, and there is underreporting of severe cases that influence the outcomes. The lack of specific studies on outcomes such as maternal bleeding, abortion and neonatal mortality contributes to the scarcity of comprehensive information. There is no consensus on how to classify cases such as foetal distress or gestational haemorrhage, complicating the measurement of outcomes. Some studies are based on the World Health Organization (WHO) classification, while others are not, preventing adequate identification of severe Dengue or Dengue-induced shock cases. Despite these limitations, the review provides a comprehensive synthesis of outcomes of dengue infection in pregnant women, which is crucial for clinical practice, as identifying patterns in outcomes can guide future research and help establish priorities in medical care.
CONCLUSIONS
Dengue infection during pregnancy significantly endangers maternal and foetal health, leading to complications such as obstetric bleeding, spontaneous abortions, and preterm births. Further research is needed to explore the long-term effects on children born to mothers who experienced Dengue during pregnancy, which is vital for developing effective care strategies for these families.
Public health initiatives should enhance epidemiological surveillance and improve access to medical care to address the risks associated with Dengue in pregnant women. This could involve implementing targeted educational programmes for healthcare providers in endemic regions to ensure they can effectively recognise and manage Dengue-related complications. Additionally, raising community awareness about Dengue symptoms and risks can empower pregnant women to seek timely medical attention.
By equipping healthcare systems with the necessary training and resources, we can significantly reduce the complications and mortality linked to Dengue during pregnancy, ultimately protecting the health of mothers and their children.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors
REFERENCES
[1] Khan MB, Yang ZS, Lin CY, et al. Dengue overview: An updated systemic review. J Infect Public Health. 2023; 16 (10): 1625-1642.
[2] Sirisena P, Mahilkar S, Sharma C, et al. Concurrent dengue infections: Epidemiology & clinical implications. Indian J Med Res. 2021; 154(5): 669-679.
[3] Organización Mundial de la Salud. OMS. 2024 [citado 22 de julio de 2024]. Dengue y dengue grave. Available in: https://www.who.int/es/news-room/fact-sheets/detail/dengue-and-severe-dengue.
[4] Kallás EG, Cintra M, Moreira JA, et al. Live, Attenuated, Tetravalent Butantan-Dengue Vaccine in Children and Adults. N Engl J Med. 2024; 390(5): 397-408.
[5] Paz-Bailey G, Adams LE, Deen J, et al. Dengue. Lancet Lond Engl. 2024; 403(10427): 667-682.
[6] Harapan H, Michie A, Sasmono R, et al. Dengue: A Minireview. Viruses. 2020; 12(8): e829.
[7] Sinha S, Singh K, Ravi-Kumar YS, et al. Dengue virus pathogenesis and host molecular machineries. J Biomed Sci. 2024; 31 (1): e43.
[8] Roy SK, Bhattacharjee S. Dengue virus: epidemiology, biology, and disease aetiology. Can J Microbiol. 2021; 67(10): 687-702.
[9] Osawa T, Aoki M, Ehara H, et al. Structures of dengue virus RNA replicase complexes. Mol Cell. 2023; 83(15): 2781-2791.
[10] Benfrid S, Park K, Dellarole M, et al. Dengue virus NS1 protein conveys pro-inflammatory signals by docking onto high-density lipoproteins. EMBO Rep. 2022; 23(7): e53600.
[11] Halstead S. Recent advances in understanding dengue. F1000Research. 2019; 8 (1): 1-12.
[12] Valdivia-Conroy B, Vasquez-Calderón JM, Silva-Caso W, et al. Rendimiento diagnóstico de la prueba rápida para la detección del antígeno NS1 y anticuerpos IgM e IgG contra el virus del dengue. Rev Peru Med Exp Salud Pública. 2023; 39 (4): 434-441.
[13] De Santis O, Bouscaren N, Flahault A. Asymptomatic dengue infection rate: A systematic literature review. Heliyon. 2023; 9 (9): e20069.
[14] Narayan R, Tripathi S. Intrinsic ADE: The Dark Side of Antibody Dependent Enhancement During Dengue Infection. Front Cell Infect Microbiol. 2020; 10 (1): e580096.
[15] Chong V, Tan J, Arasoo V. Dengue in Pregnancy: A Southeast Asian Perspective. Trop Med Infect Dis. 2023; 8 (2): e86.
[16] Duarte G, Braga A, Kreitchmann R, et al. Prevention, diagnosis, and treatment protocol of dengue during pregnancy and the postpartum period. Rev Bras Ginecol Obstet. 2024; 46: e-rbgo73.
[17] Ahuja S, Muntode P. A Narrative Review of Maternal and Perinatal Outcomes of Dengue in Pregnancy. Cureus. 2023; 15 (11): e48640.
[18] Tissera H, Wijewickrama A, Weeraman J, et al. Clinical management of dengue infection in pregnancy. Ceylon Med J. 2020; 65 (4): 757-758.
[19] Mulik V, Dad N, Buhmaid S. Dengue in pregnancy: Review article. Eur J Obstet Gynecol Reprod Biol. 2021; 261: 205-210.
[20] Centro Cochrane Iberoamericano. Cochrane. 2012 [Cited on July 23, 2024]. Manual Cochrane de revisiones sistemáticas de intervenciones. Available in: https://training.cochrane.org/es/manual-cochrane-de-revisiones-sistem%C3%A1ticas-de-intervenciones.
[21] Kallur SD, Surapaneni T, Boorugu HK, et al. Need for guidelines for the combined management of pregnancy and dengue: a retrospective study from an Indian tertiary care maternity hospital. Trop Doct. 2019; 49(1): 7-9.
[22] Sondo KA, Ouattara A, Diendéré EA, et al. Dengue infection during pregnancy in Burkina Faso: a cross-sectional study. BMC Infect Dis. 2019; 19(1): 997.
[23] Thiyagalingam S, Rengaraj S, Rajamanickkam S. Clinical Characteristics and Obstetric Outcome of Symptomatic Dengue Infection in Pregnancy from a Tertiary Care Center in South India. J Infect Dis Epidemiol. 2020; 6(3): 133-138.
[24] Escobar MF, Mora BL, Cedano JA, et al. Comprehensive treatment in severe dengue during preterm and term labor: could tocolysis be useful? J Matern Fetal Neonatal Med. 2020; 33(14): 2445-2450.
[25] Brar R, Sikka P, Suri V, et al. Maternal and fetal outcomes of dengue fever in pregnancy: a large prospective and descriptive observational study. Arch Gynecol Obstet. 2021; 304(1): 91-100.
[26] Nujum ZT, Nirmala C, Vijayakumar K, et al. Incidence and outcomes of dengue in a cohort of pregnant women from an endemic region of India: obesity could be a potential risk for adverse outcomes. Trans R Soc Trop Med Hyg. 2019; 113(5): 242-251.
[27] Naik S, Robinson ML, Alexander M, et al. Intensified Short Symptom Screening Program for Dengue Infection during Pregnancy, India. Emerg Infect Dis. 2020; 26(4): 738-743.
[28] Gupta A, Jain P, Venkatesh V, et al. Prevalence of Dengue, Chikungunya, and Zika Viruses in Febrile Pregnant Women: An Observational Study at a Tertiary Care Hospital in North India. Am J Trop Med Hyg. 2021; 106(1): 168-173.
[29] Mewada BN, Gandhi D, Diwan F, et al. Prospective study of pyrexia with pregnancy. Ann Afr Med. 2023; 22(1): 1-4.
[30] Singkibutr T, Wuttikonsammakit P, Chamnan P. Effects of Dengue Infection on Maternal and Neonatal Outcomes in Thai Pregnant Women: A Retrospective Cohort Study. J Med Assoc Thai. 2020; 103: 155-162.
[31] Tougma SA, Zoungrana/Yaméogo WN, Dahourou DL, et al. Dengue virus infection and pregnancy outcomes during the 2017 outbreak in Ouagadougou, Burkina Faso: A retrospective cohort study. PloS One. 2020; 15 (9): e0238431.
[32] Mubashir M, Ahmed KS, Mubashir H, et al. Dengue and malaria infections in pregnancy: Maternal, fetal and neonatal outcomes at a tertiary care hospital. Wien Klin Wochenschr. 2020; 132(7-8): 188-196.
[33] Sagili H, Krishna RS, Dhodapkar R, et al. Maternal & perinatal outcome of fever in pregnancy in the context of dengue - A retrospective observational study. Indian J Med Res. 2022; 156(4 & 5): 619-623.
[34] Paixão ES, Campbell OM, Teixeira MG, et al. Dengue during pregnancy and live birth outcomes: a cohort of linked data from Brazil. BMJ Open. 2019; 9(7): e023529.
[35] Mulyana RS, Pangkahila ES, Pemayun T. Maternal and Neonatal Outcomes during Dengue Infection Outbreak at a Tertiary National Hospital in Endemic Area of Indonesia. Korean J Fam Med. 2020; 41(3): 161-166.
[36] Ortiz-Mesina J, Caballero-Hoyos JR, Trujillo X, et al. Obstetric complications of dengue and chikungunya in the pregnant patient: case-control study. Rev Med Inst Mex Seguro Soc. 2019; 57(3): 162-169.
[37] Rathore SS, Oberoi S, Hilliard J, et al. Maternal and foetal-neonatal outcomes of dengue virus infection during pregnancy. Trop Med Int Health. 2022; 27(7): 619-629.
[38] Machain-Williams C, Raga E, Baak-Baak CM, et al. Maternal, Fetal, and Neonatal Outcomes in Pregnant Dengue Patients in Mexico. Biomed Res Int. 2018; 2018: e9643083.
[39] Sharma S, Jain S, Rajaram S. Spectrum of Maternofetal Outcomes during Dengue Infection in Pregnancy: An Insight. Infect Dis Obstet Gynecol. 2016; 2016: e5046091.
[40] Xiong YQ, Mo Y, Shi TL, et al. Dengue virus infection during pregnancy increased the risk of adverse fetal outcomes? An updated meta-analysis. J Clin Virol. 2017; 94: 42-49.
[41] Paixão E, Teixeira M, Costa M, et al. Dengue during pregnancy and adverse fetal outcomes: a systematic review and meta-analysis. Lancet Infect Dis. 2016; 16(7): 857-865.
[42] Sinha R, Datta MR. Dengue in Early Pregnancy: A Neglected Problem? Cureus. 15(5): e38740.
[43] Romero-Machado CR, Machado ES, Rohloff RD, et al. Is pregnancy associated with severe dengue? A review of data from the Rio de Janeiro surveillance information system. PLoS Negl Trop Dis. 2013; 7(5): e2217.
[44] Annan E, Nguyen US, Treviño J, et al. Moderation effects of serotype on dengue severity across pregnancy status in Mexico. BMC Infect Dis. 2023; 23(1): 147.
[45] Adorno M, Maher-Griffiths C, Grush-Abadie HR. HELLP Syndrome. Crit Care Nurs Clin North Am. 2022; 34(3): 277-288.
[46] Mei JY, Afshar Y. Hypertensive complications of pregnancy: Hepatic consequences of preeclampsia through HELLP syndrome. Clin Liver Dis. 2023; 22(6): 195-199.
[47] Howard-Jones AR, Pham D, Sparks R, et al. Arthropod-Borne Flaviviruses in Pregnancy. Microorganisms. 2023; 11(2): 433.
[48] Patnaik R, Kulkarni S, Karan N. Dengue and HELLP: Beware of the Masquerade. Indian J Crit Care Med. 2022; 26(5): 639-640.
[49] Harde M, Bhadade R, deSouza R, et al. Thrombocytopenia in Pregnancy Nearing Term: A Clinical Analysis. Indian J Crit Care Med. 2019; 23(11): 503-508.
[50] Agarwal K, Malik S, Mittal P. A retrospective analysis of the symptoms and course of dengue infection during pregnancy. Int J Gynecol Obstet. 2017; 139(1): 4-8.
[51] Wang WH, Urbina AN, Chang MR, et al. Dengue hemorrhagic fever - A systemic literature review of current perspectives on pathogenesis, prevention and control. J Microbiol Immunol Infect. 2020; 53(6): 963-978.
[52] Sookaromdee P, Wiwanitkit V. Dengue Shock Syndrome, Anaphylaxis, and Homeopathic Medicine. Homeopathy. 2022; 111(3): 232.
[53] Rajapakse S, Wattegama M, Weeratunga P, et al. Beyond thrombocytopaenia, haemorrhage and shock: the expanded dengue syndrome. Pathog Glob Health. 2018; 112(8): 404-414.
[54] McBride A, Chanh HQ, Fraser JF, et al. Microvascular dysfunction in septic and dengue shock: Pathophysiology and implications for clinical management. Glob Cardiol Sci Pract. 2020; 2020(2): e202029.
[55] Shrivastava G, Valenzuela-Leon PC, Calvo E. Inflammasome Fuels Dengue Severity. Front Cell Infect Microbiol. 2020; 10: 489.
[56] Rathore AP, Mantri CK, Aman SA, et al. Dengue virus-elicited tryptase induces endothelial permeability and shock. J Clin Invest. 2019; 129(10): 4180-4193.
[57] Sudulagunta SR, Sodalagunta MB, Sepehrar M, et al. Dengue shock syndrome. Oxf Med Case Rep. 2016; 2016(11): omw074.
[58] Boon PL, Martins AS, Lim XN, et al. Dengue Virus Capsid Protein Facilitates Genome Compaction and Packaging. Int J Mol Sci. 2023; 24(9): 8158.
[59] Watterson D, Modhiran N, Muller DA, et al. Plugging the Leak in Dengue Shock. Adv Exp Med Biol. 2018; 1062: 89-106.
[60] Kok BH, Lim HT, Lim CP, et al. Dengue virus infection - a review of pathogenesis, vaccines, diagnosis and therapy. Virus Res. 2023; 324: 199018.
[61] López-Barroso R, Deulofeu-Betancourt I, Fayad-Saeta Y, et al. Repercusión del dengue serotipo 3 sobre el embarazo y producto de la concepción. Rev Cuba Obstet Ginecol. 2010; 36(2): 42-50.
[62] Romero-Angarita LC, Angarita SV, Correa M, et al. Transmisión perinatal del virus dengue en el binomio madre-hijo. Arch Venez Pueric Pediatría. 2013; 76(3): 99-104.
[63] Feitoza HA, Koifman S, Koifman RJ, et al. Dengue infection during pregnancy and adverse maternal, fetal, and infant health outcomes in Rio Branco, Acre State, Brazil, 2007-2012. Cad Saude Publica. 2017; 33(5): e00178915.
[64] Tejo AM, Hamasaki DT, Menezes LM, et al. Severe dengue in the intensive care unit. J Intensive Med. 2024; 4(1): 16-33.
[65] Organización Panamericana de la Salud. PAHO. 2024 [citado 22 de julio de 2024]. Situación Epidemiológica del Dengue en la Región de las Américas - Semana Epidemiológica 26. Available in: https://www.paho.org/en/documents/situation-report-no-26-dengue-epidemiological-situation-region-americas-epidemiological.
[66] Kharwadkar S, Herath N. Clinical manifestations of dengue, Zika and chikungunya in the Pacific Islands: A systematic review and meta-analysis. Rev Med Virol. 2024; 34(2): e2521.