Le Infezioni in Medicina, n. 1, 114-124, 2025
doi: 10.53854/liim-3301-11
ORIGINAL ARTICLES
Impact of Enterococcus infection in HSCT recipients: a national analysis
Barath Prashanth Sivasubramanian1, Ajay Sriram Antony Raj2, Diviya Bharathi Ravikumar3, Aneela Satya Ravanam4, Rutvi Balkrishna Patel5, Samhitha Mudumalagurthy6, Devi Meghana Kotharu7, Mohd Zeeshan8, Manisha Chavan9, Jay Patel10, Akhila Vala11, Raghavendra Tirupathi12, Rutul Dalal13, Saketh Palasamudram Shekar14
1Internal Medicine, Northeast Georgia Medical Centre, Gainesville, GA, USA;
2Institute of Internal Medicine, Madras Medical College, Chennai, Tamil Nadu, India;
3Internal Medicine, Employee State Insurance Corporation and Postgraduate Institute of Medical Sciences and Research, Chennai, Tamil Nadu, India;
4Department of Medicine, Government Medical College, Anantapur, Andhra Pradesh, India;
5Department of Medicine, Narendra Modi Medical College, Ahmedabad, Gujarat, India;
6Department of Medicine, Kurnool Medical College, Kurnool, Andhra Pradesh, India;
7Department of Medicine, MNR Medical College, Sanga Reddy, Telangana, India;
8Department of Medicine, Career Institute of Medical Sciences and Hospital, Lucknow, Uttar Pradesh, India;
9Department of Medicine, Kakatiya Medical College, Warangal, Telangana, India;
10Department of Medicine, B.J. Medical College, Ahmedabad, Gujarat, India;
11Department of Medicine, Prathima Institute of Medical Sciences, Nagunur, Telangana, India;
12Department of Infectious Diseases, Keystone Health, Chambersburg, PA, USA;
13Infectious Diseases, Penn State Health Eastern Region, Penn State Health St Joseph Medical Centre, PA, USA;
14Interventional Pulmonologist, Pulmonary and Sleep Associates of Huntsville, Huntsville, AL, USA.
Article received 4 December 2024 and accepted 29 January 2025
Corresponding author
Diviya Bharathi Ravikumar
E-mail: diviyabharathi2612@gmail.com
SummaRY
Background: Enterococci are the third most common cause of healthcare-associated infections in the United States, affecting 10-12% of all transplant recipients worldwide. Enterococcal bacteremia complicates the post-transplant recovery and raises mortality to 18%. This study aims to identify factors linked to mortality in hematopoietic stem cell transplant (HSCT) recipients with Enterococcus infection.
Methods: We conducted a retrospective analysis using the National Inpatient Sample Database (2018-2021). Adults treated with HSCT and admitted for Enterococcus infection were identified using ICD-10 codes. The analysis utilized descriptive statistics and multivariate regression, with significance at a p-value ≤0.05.
Results: A total of 40,462 HSCT patients, 1.3% were admitted with Enterococcus infection. The crude mortality rate was higher in the Enterococcus cohort than in the non-Enterococcus cohort (10.4% vs. 4.6%); however, the mortality risk was substantially lower in the Enterococcus cohort (p<0.001). In the Enterococcus cohort, patients with fungal infections, sepsis, acute respiratory failure, and those with invasive ventilation had a high mortality risk (p<0.05). In leukemia patients treated with HSCT, the Enterococcus cohort had higher mortality than the non-Enterococcus cohort (12.5% vs. 5.0%; aOR 1.8, 95% CI 1.9-2.6, p=0.005).
Conclusions: In HSCT recipients, Enterococcus infection is a clinical marker for poor prognosis. Early broad-spectrum antibiotic therapy is essential for patients with sepsis, respiratory compromise, and fungal co-infections. Additionally, a multidisciplinary team is crucial for managing respiratory and renal failure.
Keywords: Enterococcus, Hematopoietic Stem Cell Transplant, mortality, Leukemia, infections.
INTRODUCTION
Bacteremia is the most common cause of morbidity and mortality in hematopoietic stem cell transplant (HSCT) recipients. Both Gram-positive and Gram-negative bacteremia have been found to occur in HSCT recipients, typically in the early post-transplant phase [1]. Enterococci are the third most common cause of healthcare-associated infections in the United States and affect 10-12% of all transplant recipients worldwide [2-4]. Among the 58 recognized species of enterococci, the most common are E. faecalis and E. faecium [5].
There has also been a noticeable increase in the prevalence of E. faecium, constituting 40% of bloodstream isolates in liver and stem cell transplant recipients [6, 7]. Septic shock, urinary tract infections, meningitis, intra-abdominal and biliary tract infections, and endocarditis are common presentations of bacteremia [8-11]. Immunocompromised patients, those with prolonged neutropenia and gastrointestinal mucositis, are at increased risk for sepsis because of gut translocation into the bloodstream [1, 12]. Risk factors for bacteremia include age over 60 years at the time of transplantation, use of central venous and urinary catheters, prior antibiotic use, underlying acute lymphocytic leukemia, and previous hospitalizations [3, 8]. The risk of mortality also rises in HSCT recipients from 18% on day 7 to as high as 31% on day 30 [11, 13, 14].
However, distinguishing the impact of various risk factors that may co-occur or occur at different times following HSCT is challenging [15]. Studies focusing on the epidemiology and mortality of multidrug-resistant pathogens affecting the HSCT population are limited [16, 17]. The literature has not addressed the effects of bone marrow dysfunction such as anemia, neutropenia, and thrombocytopenia [18-21]. There is a dire need to identify effective strategies for preventing infections and sepsis during HSCT [22].
Our study focused on identifying the risk factors associated with inpatient mortality in HSCT recipients who develop Enterococcus infections, including those presenting with severe sepsis. Additionally, we sought to determine the impact of enterococci on solid organ transplant recipients and leukemia patients who received HSCT.
METHODS
Design and data source
We queried the 2018-2021 National Inpatient Sample (NIS) database for a retrospective cross-sectional study. The NIS was developed by the Healthcare Cost and Utilization Project (HCUP) and was sponsored by the Agency for Healthcare Research and Quality (AHRQ) [23]. The objective of this project was to produce comprehensive data on the utilization, costs, and outcomes of inpatient care at both the regional and national levels in the United States. The document consists of several elements, including patient demographics (such as age, sex, and race), diagnosis and procedure codes obtained from the International Classification of Diseases, Tenth Revision, Clinical Modification/Procedure Coding System (ICD-10-CM/PCS), indicators of severity and comorbidity, hospital characteristics, and discharge status.
Ethical Consideration, Sample Size, and Study Population
The NIS is a de-identified, publicly accessible database. A predetermined sample size was not calculated. Adults aged 18 years or older who were admitted with an ICD10-CM code for Hematopoietic Stem Cell Transplant and Enterococcus infection were included. Our study focused on Enterococcus infections, including those presenting with sepsis and other presentations. The ICD-10-CM/PCS codes were used to identify patients and are provided in Supplementary File Table 1. Before accessing the NIS databases, we adhered to AHRQ’s data user agreement. The databases utilized adhere to the HIPAA (Health Insurance Portability and Accountability Act) Privacy Rule’s definition of limited datasets. These databases do not contain any explicit identifiers of patients. Ethics consent is not required for our study as this is a publicly available repository.
Table 1 - Prevalence, sepsis, mortality, and palliative care rates in different Solid Organ Transplant (SOT) recipients with enterococcal infection.

Variables Assessed
Gender was delineated as male or female. The patient’s race was defined as White (referent), African American, Hispanic, Asian or Pacific Islander, or Native American. The ICD-10 codes encompass acute kidney injury, myocardial infarction, invasive ventilation, severe sepsis, acute respiratory failure, acute respiratory distress syndrome, acute heart failure, and vasopressor usage.
Statistical Methods
We analyzed continuous variables using means and t-tests, while qualitative variables were analyzed using the chi-square test. We established a threshold of statistical significance at p≤0.05. The analysis was conducted using Stata v18. In our analysis, we employed two distinct techniques to account for confounding variables: Propensity score matching and multivariate regression analysis. Propensity scores were employed to pair HSCT recipients with Enterococcus infection with those without. A multivariate logistic regression model was created to calculate the propensity score for mortality utilizing the variables of age, gender, race, and the number of comorbidities. The researchers employed the double robust approach to calculate treatment weights. They then utilized the inverse likelihood of treatment weighting to match cases with controls using generalized linear models, as described by Dugoff et al. [24]. The second analysis employed multivariable regression models to control for potential confounding factors. Multiple regression models were constructed by incorporating all confounding variables that were significantly associated with the outcome in the univariate analysis, with a threshold p-value of 0.2. The models included variables that were identified as significant determinants of the outcomes according to the literature. The Crude Mortality is an unadjusted rate reported as percentages (%). Adjusted mortality risks were calculated using multivariable logistic regression, controlling for confounders like age, gender, race, and number of co-morbidities. Results were reported as adjusted odds ratios (aOR) with 95% confidence intervals.
Outcomes
The primary aim of our study was to evaluate mortality in enterococcal-infected HSCT patients. The secondary aim of our study was to identify the impact of Enterococcus infection in solid organ transplant recipients. The tertiary aim was to assess the impact of Enterococcus in leukemia patients who were treated with HSCT.
RESULTS
Among the 198,045 patients who received organ, tissue, and stem cell transplantation (all transplants), 1.6% (n=3,175) of the admissions had Enterococcus infection. In comparison, 0.8% (n=173,813) of 23 million non-transplant patients had Enterococcus (p≤0.001). In the all-transplant group, individuals aged 61 to 75 years had the highest rate of Enterococcus infections (45.2%, p=0.4742) and experienced higher mortality rates (51.3%, p=0.2698). Overall, those over the age of 60 had a higher risk of mortality compared to younger patients (63.3% vs. 55.7%, p=0.0672). Additionally, males had a higher prevalence of Enterococcus infections than females (55.6% vs. 44.4%) and experienced greater mortality (64.7% vs. 35.3%, p=0.0215); however, these differences were not statistically significant. Patients with Enterococcus infections had a prevalence of peritoneal abscess of 1.5% and urinary tract infection of 54.9%. In terms of bone marrow dysfunction or immunosuppression, the prevalence of anemia was 54.2%, that of thrombocytopenia was 12.2%, that of neutropenia was 5.4%, and that of pancytopenia was 13.5%. Supplementary file Table 2 depicts the Source of infection, bone marrow dysfunction, co-morbidities, interventions, and outcomes of all transplant recipients with and without enterococcal infection. The prevalence of Enterococcus infection in patients with pancreatic transplants was 2.1%, that in kidney transplants was 1.7%, that in hematopoietic stem cell transplantation was 1.3%, that in liver transplants was 1.5%, that in lung transplants was 1.1%, and that in heart transplants was 1.0%. Table 1 shows the prevalence, sepsis, mortality, and palliative care rates in different solid organ transplant recipients with enterococcal infection. For all-transplants, the mortality rate of the Enterococcus cohort was 4.7% (95% CI 4.0%-5.5%), whereas that of the non-Enterococcus cohort was 3.1% (95% CI 3.0% to 3.1%). Patients who faced mortality tended to be over the age of 60 years (p=0.0672), male (p=0.0215), belonged to the White race (p=0.6726), and had three or more co-morbidities (p=0.0526). Supplementary file Table 3 shows the socio-demographic factors and number of co-morbidities in all transplant recipients with and without enterococcal infection. Crude analysis revealed the prevalence of comorbidities such as severe sepsis, pancytopenia, diabetes, fungal infections, acute respiratory failure, acute heart failure, life-threatening arrhythmias, stroke, acidosis, and metabolic encephalopathy was significantly higher in the patients who faced mortality compared to those who survived (p<0.01). However, the mortality risk of Enterococcus infection did not differ from the non-Enterococcus infection (aOR 1.1; 95% CI 0.9 to 1.4; p=0.351) when adjusted for age over 60 years, gender, comorbidities, and interventions (invasive ventilation and vasopressors). We also identified the predictors of mortality via regression analysis. The risk factors for mortality, showing high odds with a p≤0.05, included severe sepsis, pancytopenia, fungal infections, acute respiratory failure, arrhythmia, invasive ventilation, and vasopressor usage. Table 2 depicts the mortality predictors in all transplants with enterococcal infection. Compared with the non-Enterococcus cohort, the Enterococcus cohort required more palliative care (4.9% vs. 3.6%, p=0.0001) as shown in Table 3.
Table 2 - Mortality predictors in all-transplants with enterococcal infection.

Table 3 - All transplantation and HSCT transplantation.

Among the 198,045 patients who received transplants, 20.4% (n=40,462) received hematopoietic stem cell transplantation (HSCT). In the HSCT group, 1.3% (n=531) were admitted for Enterococcus infection. In the HSCT population, individuals aged 61-75 years were most frequently affected by Enterococcus infections (49.2%). Compared with male patients, female patients had more Enterococcus admissions (51.0% vs. 49.0%, p<0.0001). The race did not differ between the Enterococcus and non-Enterococcus cohorts (p=0.0612). Supplementary file Table 4 shows the socio-demographic factors and number of co-morbidities in HSCT recipients with and without enterococcal infection. Among the patients with Enterococcus infection, 0.6% had peritoneal abscesses and 39.0% had acute urinary tract infections. Severe sepsis was seen in 20.2% of the patients. The factors associated with bone marrow dysfunction or immunosuppression were anemia (68.4%), thrombocytopenia (21.3%), neutropenia (18.6%), and pancytopenia (46.3%) in the Enterococcus cohort. Supplementary file Table 5 depicts the sources of infection, bone marrow dysfunction, co-morbidities, interventions, and outcomes of HSCT recipients with and without enterococcal infection. On crude analysis, the mortality rate of the Enterococcus cohort was higher than the non-Enterococcus cohort by 10.4% (95% CI 8.1-13.2%) vs. 4.6% (95% CI 4.4% to 4.8%). However, after matching and adjusting for age over 60 years, gender, comorbidities mentioned above, and interventions (invasive ventilation and vasopressor usage), patients with Enterococcus infection had a lower mortality risk than did those without Enterococcus infection (aOR 0.1, 95% CI 0.0 to 0.3, p<0.001). We also identified risk factors for mortality in the Enterococcus cohort. These factors, including severe sepsis, fungal infections, acute respiratory failure, acidosis, and invasive ventilation, had high odds of mortality, with a p-value of ≤0.05. Table 4 illustrates the mortality predictors in HSCT recipients with enterococcal infection. The Enterococcus cohort required more palliative care than the non-Enterococcus cohort did (11.1% vs. 6.9%, p=0.0001). The total hospital charges (Coefficient $94,629, 95% CI $-113,069 to $302,327, p=0.372) and the duration of hospital stay (Coefficient 7.6 days, 95% CI -2.8 to 18.1, p=0.152) did not differ between the two cohorts. We also identified the prevalence of post-transplant complications in HSCT patients (2.7%; n=1091). Among these patients, 2.0% had Enterococcus infection.
Table 4 - Mortality predictors in HSCT recipients with enterococcal infection.
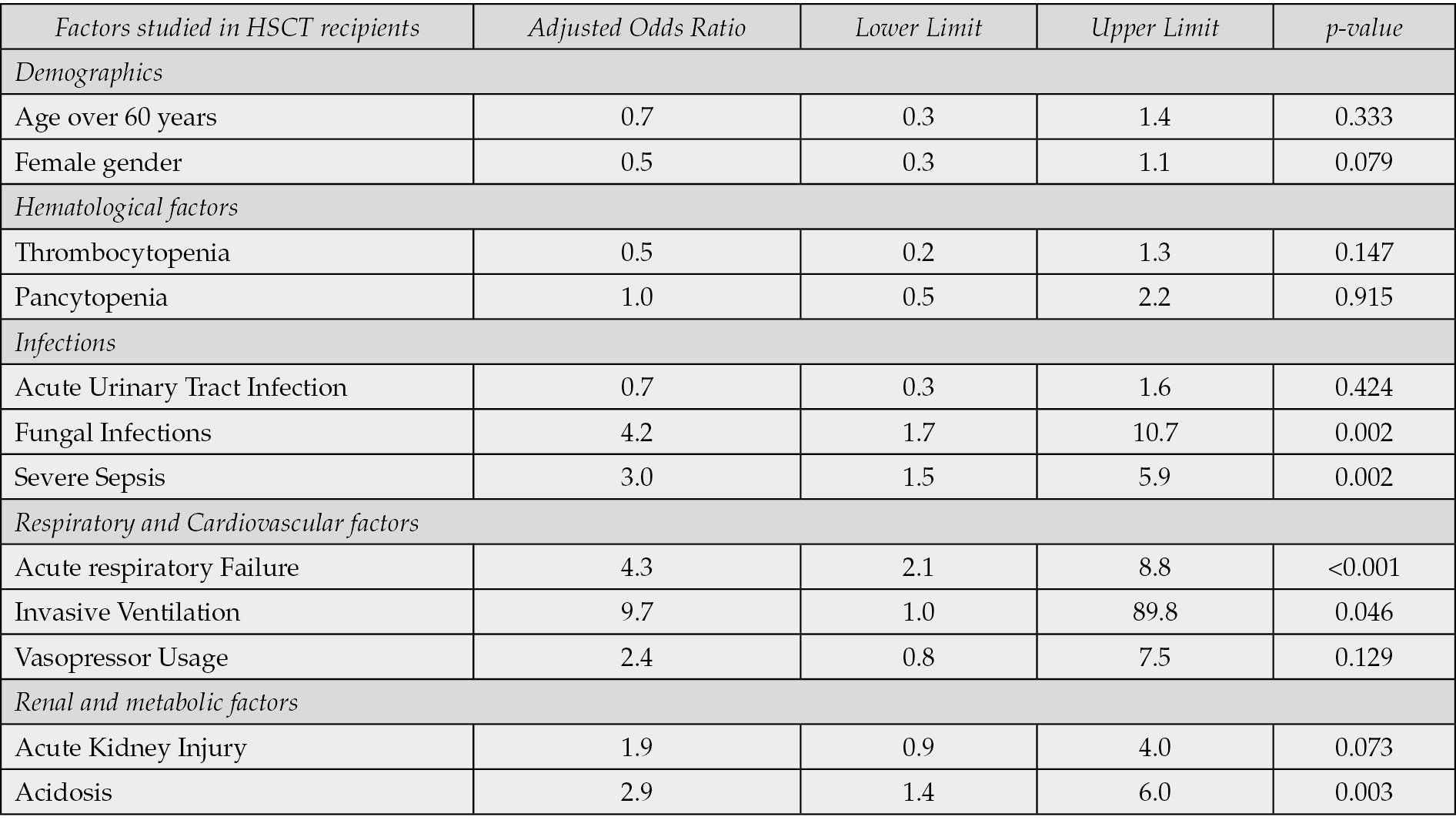
Table 5 - Socio-demographic factors, Bone marrow dysfunction, Comorbidities and Interventions in HSCT-Leukemia and HSCT-AML: With and without Enterococcus infection.

The HSCT population was stratified based on the underlying disease. Among the HSCT recipients, Enterococcus infection was found in 344 (1.4%) of 23,930 leukemia patients and 134 (2.4%) of 5,693 AML (Acute Myeloid Leukemia) patients. Among the 23,930 leukemia patients, 344 (1.4%) had Enterococcus infection. This proportion was higher than that in the non-leukemic population (n=187, 1.1%, p=0.008). In those with leukemia, age over 60 years and race did not differ between the Enterococcus and non-Enterococcus cohorts. Compared to males, females had higher Enterococcus infection (50.3% vs. 49.7%, p=0.0046). Additionally, the Enterococcus cohort had a higher prevalence of urinary tract infections, fungal infections, acute respiratory failure, acute kidney injury, and acidosis. Enterococcus infection was common among patients with signs of bone marrow dysfunction such as neutropenia, anemia, and pancytopenia. Enterococcus infection was also associated with increased rates of severe sepsis and vasopressor usage. In the crude analysis, the mortality rate in the Enterococcus cohort was again higher than that in the non-Enterococcus cohort (12.5% vs. 5.0%, p<0.0001). However, after adjusting for age over 60 years, gender, comorbidities (severe sepsis, acute respiratory failure, acute kidney injury, and acidosis), and vasopressor usage, the mortality risk was found to be higher than that in the non-Enterococcus cohort (aOR 1.8, 95% CI 1.9 to 2.6, p=0.005). The impact of Enterococcus could be understood through the utilization of palliative care services. Patients with Enterococcus infection had a significantly greater need for palliative care than did those without Enterococcus (13.1% vs. 7.7%, p=0.0002). Table 5 outlines the socio-demographic factors, bone marrow dysfunction, comorbidities, and interventions in HSCT-Leukemia and HSCT-AML- with and without Enterococcus infection.
In the AML population, there were no significant differences in socio-demographic factors such as age over 60 years, gender, or race between those with and without Enterococcus infection (p>0.5). Additionally, the Enterococcus cohort had a higher prevalence of urinary tract infections, fungal infections, and acute respiratory failure. Enterococcus infection is common among patients with signs of bone marrow dysfunction such as neutropenia, anemia, and pancytopenia. Enterococcus infection was also associated with increased rates of severe sepsis and increased vasopressor usage. Crude analysis revealed a higher mortality rate in the Enterococcus cohort compared to the non-Enterococcus cohort (16.1% vs. 8.8%, p=0.032). However, after adjusting for age over 60 years, severe sepsis, co-morbidities (fungal infections, acute respiratory failure), and vasopressor usage, the mortality risk did not significantly differ between the two cohorts (aOR 0.9, 95% CI 0.4-1.7, p=0.67). Similarly, the utilization of palliative care services did not differ between the two cohorts (12.7% vs. 9.8%, p=0.2723).
DISCUSSION
To our knowledge, our study is the first to exclusively focus on Enterococcus-related mortality and associated risk factors in a large population of HSCT recipients with enterococcal infections. Additionally, propensity matching and multivariate regression aid in understanding the severity of Enterococcus infection in the transplant population. In the HSCT population, Gram-positive bacteremia is a common cause (41.2%) of infection [25]. Our findings revealed that 1.3% of HSCT recipients had enterococcal infections, in contrast to the higher prevalence of Methicillin-resistant Staphylococcus aureus (2.9%) and gram-negative infections such as Escherichia coli (13%) and Klebsiella pneumoniae (7.2%) [25, 26]. Males and females had the same prevalence of enterococcal infections, despite males facing a higher mortality risk in our study. Urinary tract infection was one of the most common presentations of enterococcal infection [9, 27]. In line with this, in our study, one-third of HSCT recipients with enterococcal infections had a urinary tract infection, and a quarter of this population faced mortality. Additionally, half of all HSCT recipients with enterococcal infections had pancytopenia. We found that one-tenth of the HSCT recipients with enterococcal infections experienced mortality and required palliative care. The crude mortality rates were higher in enterococcal-infected patients, but after adjusting for confounders, the mortality risk was lower compared to uninfected patients. This suggests that the HSCT population is at risk of death from other causes. Similarly, registry studies from the U.S., Australia, and Europe in HSCT cohorts report that infections contribute only marginally (0.3-10%) to mortality, with other factors such as relapse of the primary malignancy (>50%), subsequent secondary malignancies (7.5-25%), and graft versus host disease (25%) having a more substantial impact [28–30]. Similar to these findings, Tavadze et al. and Avery et al. reported that enterococcal bacteremia was a sign of a rapidly deteriorating clinical course and poor overall survival [31, 32]. The risk of mortality increases in patients who need invasive ventilation or concurrently have pancytopenia, severe sepsis, fungal infections, respiratory distress, and acidosis. Avery et al. also reported that fungal infections, multiorgan failure, and ARDS contributed to death [31]. We observed that Enterococcus infections are linked to higher rates of sepsis and vasopressor support. The closer hemodynamic monitoring and managing metabolic disturbances often require ICU level of care. HSCT patients require broad-spectrum antibiotics, respiratory support, and multidisciplinary care to reduce mortality.
With respect to the underlying disease in HSCT recipients, we observed that the mortality risk among those with leukemia caused by enterococci was greater than that among those without enterococcal infection (aOR 1.75, 95% CI 1.18 to 2.58, p=0.005). However, for HSCT recipients with AML, the mortality risk did not differ between those infected with enterococci and those who were not (aOR 0.86, 95% CI 0.4-1.7, p=0.67). Ferreira et al. reported that HSCT patients aged more than 62 years were at a higher risk of enterococcal infections [33]. However, in our study, we noted that age and race were not determinants of Enterococcus infection in either the leukemia or the AML group. Peng et al. reported thrombocytopenia as a risk factor for Enterococcus infection in the pediatric population [34]. We identified similar findings in our study, which included adult patients over 18 years of age. We found that patients with anemia, thrombocytopenia, and pancytopenia were more likely to develop enterococcal infections. In leukemia patients, anemia (p<0.001) and pancytopenia (p<0.001) were significant factors, while in AML patients, anemia (p<0.0001), thrombocytopenia (p=0.0004), and pancytopenia (p<0.0001) were found to be significantly associated with Enterococcus-related admissions.
In patients with Enterococcus infection, severe sepsis, urinary tract infection, or respiratory failure necessitate aggressive treatment with broad-spectrum antibiotics that need to be switched to appropriate antibiotics after resistance patterns are identified. Broad-spectrum antibiotics are implicated in this condition for several reasons, including the multidrug-resistant nature of Enterococcus, the tendency of enterococci to coexist and present as polymicrobial infections, and the vulnerability of HSCT patients [5, 35]. Owing to the high crude mortality rate (10.4%), providing multidisciplinary care is essential to reduce hemodynamic compromise, ensure close monitoring, and manage other comorbidities. Enterococcus should be considered a differential diagnosis for any Leukemia patient with HSCT who presents with a urinary tract infection or respiratory failure.
We found that patients who underwent pancreas, kidney, liver, or renal transplantation presented a slightly greater prevalence of Enterococcus infection (1.5-2.0%) than did those who underwent heart or lung transplantation (1.0-1.1%). Newell et al. reported that among liver transplant recipients, Enterococcus-related sepsis significantly contributed to mortality. This was influenced by factors such as coinfection with multidrug-resistant bacteria, additional laparotomies, and prior vancomycin use [36]. Gotur et al. also reported high sepsis rates in heart and lung transplant recipients [37]. Consistent with these findings, our study revealed high severe sepsis rates in liver (16.8%) and lung (15.8%) transplant recipients. Additionally, we found that among heart transplant recipients, despite the lower prevalence of Enterococcus, there was a higher rate of severe sepsis (14.2%), indicating that these patients are more prone to Enterococcus-associated complications. Furthermore, we observed that mortality rates were particularly high among pancreas (4.0%) and kidney transplant recipients (3.4%). Similarly, Linares et al. reported increased mortality among kidney transplant recipients infected with multidrug-resistant bacteria, with 9.5% of these infections attributed to enterococci [38]. Farges et. al reported high mortality in organ transplant patients with invasive fungal infections despite measures such as early ICU admission, the use of vasopressors, and mechanical ventilation [39]. Similarly, our study revealed a mortality rate of 23.6% for fungal infections in the HSCT Enterococcus cohort. These findings underscore the critical need for early detection and aggressive management of infections in transplant recipients to improve patient outcomes.
Limitations in our study include the inability to gather detailed information regarding the relapse of leukemia, transplant rejection rates, other solid organ transplants, transplant complications, and the number of transplants. Additionally, evidence regarding the patients’ code status, initiation and duration of antibiotic and immunosuppressant therapies, laboratory values, the exact cause of mortality, and the persistence and severity of complications is limited. We also lacked detailed outcome data, such as death at 30 days, follow-up information, and whether patients recovered from illness. Our study could not investigate the antimicrobial resistance patterns in this population. The primary data source was from clinical registries, from which retrospective chart abstractions were made using billing codes, discharge diagnosis codes, and similar identifiers, introducing the possibility of coding errors. Another notable limitation of our analysis is the failure to stratify data by enterococcal species, specifically E. faecium and E. faecalis. Owing to the retrospective design of the research, we could not control for unidentified confounding variables.
Future research should focus on developing predictive models that integrate prognostic factors—such as infection severity, immune profile, and patient demographics—to enhance mortality risk assessment in HSCT recipients with enterococcal infections. In high-risk populations the role of antimicrobial stewardship programs that optimize antimicrobial use, reduce antimicrobial resistance, and improve overall outcomes are critical but could not be studied due to the limitations of the study design. Longitudinal studies examining immediate and late post-transplant complications and optimizing antimicrobial stewardship are necessary to improve survival.
CONCLUSIONS
Our goal of this study was to draw attention to the significance of enterococcal infections in transplant recipients among adults in the United States. Enterococcus infection is a clinical marker for poor prognosis, as evidenced by the high mortality rate. Early broad-spectrum antibiotic therapy that covers Enterococcus is essential for patients with sepsis, respiratory compromise, and fungal co-infections. Additionally, a multidisciplinary team is crucial for managing respiratory and renal failure to improve outcomes.
Conflict of interest
The authors declare that they have no conflicts of interest.
None to declare.
Authors’ contributions
Writing-original drafting: Barath Prashanth Sivasubramanian, Ajay Sriram Antony Raj, Aneela Satya Ravanam, Diviya Bharathi Ravikumar, Samhitha Mudumalagurthy, Rutvi Balkrishna Patel, Devi Meghana Kotharu, Mohd Zeeshan, Manisha Chavan, Jay Patel, Akhila Vala.
Data analysis: Barath Prashanth Sivasubramanian.
Critical review and conception of the manuscript: Raghavendra Tirupathi, Rutul Dalal, Saketh Palasamudram Shekar.
All authors have reviewed the final version to be published and agreed to be accountable for all aspects of the work.
Supplementary file Table 1 - ICD 10 codes.

Supplementary file Table 2 - Sources of infection, bone marrow dysfunction, comorbidities, interventions and outcomes of all-transplant recipients with and without enterococcal infection.

Supplementary file Table 3 - Socio-demographic factors, number of comorbidities in all-transplant recipients with and without enterococcal infection.

Supplementary file Table 4 - Socio-demographic factors, number of comorbidities in HSCT recipients with and without enterococcal infection.

Supplementary file Table 5 - Sources of infection, bone marrow dysfunction, comorbidities, interventions, and outcomes of HSCT recipients with and without enterococcal infection.

REFERENCES
[1] Wingard JR, Hsu J, Hiemenz JW. Hematopoietic stem cell transplantation: an overview of infection risks and epidemiology. Infect Dis Clin North Am. 2010; 24: 257-272.
[2] Macesic N, Morrissey CO, Cheng AC, et al. Changing microbial epidemiology in hematopoietic stem cell transplant recipients: increasing resistance over a 9-year period. Transpl Infect. Dis 2014; 16: 887-896.
[3] Mikulska M, Del Bono V, Prinapori R, et al. Risk factors for enterococcal bacteremia in allogeneic hematopoietic stem cell transplant recipients. Transpl Infect Dis. 2010; 12: 505-512.
[4] Hidron AI, Edwards JR, Patel J, et al. NHSN annual update: antimicrobial-resistant pathogens associated with healthcare-associated infections: annual summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2006-2007. Infect Control Hosp Epidemiol. 2008; 29: 996-1011.
[5] García-Solache M, Rice LB. The Enterococcus: a Model of Adaptability to Its Environment. Clin Microbiol Rev; 32. Epub ahead of print 20 March 2019. DOI: 10.1128/CMR.00058-18.
[6] Mikulska M, Del Bono V, Raiola AM, et al. Blood stream infections in allogeneic hematopoietic stem cell transplant recipients: reemergence of Gram-negative rods and increasing antibiotic resistance. Biol Blood Marrow Transplant. 2009; 15: 47-53.
[7] Bedini A, Codeluppi M, Cocchi S, et al. Gram-positive bloodstream infections in liver transplant recipients: incidence, risk factors, and impact on survival. Transplant Proc. 2007; 39: 1947-1949.
[8] Kang Y, Vicente M, Parsad S, et al. Evaluation of risk factors for vancomycin-resistant Enterococcus bacteremia among previously colonized hematopoietic stem cell transplant patients. Transpl Infect Dis. 2013; 15: 466-473.
[9] Rolston KVI, Jiang Y, Matar M. VRE fecal colonization/infection in cancer patients. Bone Marrow Transplant. 2007; 39: 567-568.
[10] Wang JS, Muzevich K, Edmond MB, et al. Central nervous system infections due to vancomycin-resistant enterococci: case series and review of the literature. Int J Infect Dis. 2014; 25: 26-31.
[11] Satlin MJ, Soave R, Racanelli AC, et al. The emergence of vancomycin-resistant enterococcal bacteremia in hematopoietic stem cell transplant recipients. Leuk Lymphoma. 2014; 55: 2858-2865.
[12] Vedaldi D, Dall’Acqua F, Caffieri S, et al. Sequence specificity in DNA for the interaction with adriamycin or daunomycin. Farmaco Sci. 1982; 37: 571-581.
[13] Kusakabe S, Fukushima K, Yokota T, et al. Enterococcus: A Predictor of Ravaged Microbiota and Poor Prognosis after Allogeneic Hematopoietic Stem Cell Transplantation. Biol Blood Marrow Transplant. 2020; 26: 1028-1033.
[14] Xie O, Slavin MA, Teh BW, et al. Epidemiology, treatment and outcomes of bloodstream infection due to vancomycin-resistant enterococci in cancer patients in a vanB endemic setting. BMC Infect Dis. 2020; 20: 228.
[15] Castagnola E, Faraci M. Management of bacteremia in patients undergoing hematopoietic stem cell transplantation. Expert Rev Anti Infect Ther. 2009; 7: 607-621.
[16] Mikulska M, Del Bono V, Viscoli C. Bacterial infections in hematopoietic stem cell transplantation recipients. Curr Opin Hematol. 2014; 21: 451-458.
[17] Balletto E, Mikulska M. Bacterial Infections in Hematopoietic Stem Cell Transplant Recipients. Mediterr J Hematol Infect Dis. 2015; 7: e2015045.
[18] Papanicolaou GA, Ustun C, Young J-AH, et al. Bloodstream Infection Due to Vancomycin-resistant Enterococcus Is Associated With Increased Mortality After Hematopoietic Cell Transplantation for Acute Leukemia and Myelodysplastic Syndrome: A Multicenter, Retrospective Cohort Study. Clin Infect Dis. 2019; 69: 1771-1779.
[19] Ohta T, Ueno T, Uehara Y, et al. Incidence, Etiology, Risk Factors, and Outcomes of Bloodstream Infection after a Second Hematopoietic Stem Cell Transplantation. Intern Med. 2023; 62: 3305-3316.
[20] Benamu E, Deresinski S. Vancomycin-resistant enterococcus infection in the hematopoietic stem cell transplant recipient: an overview of epidemiology, management, and prevention. F1000Res. 2018; 7: 3.
[21] Styczyński J, Tridello G, Koster L, et al. Death after hematopoietic stem cell transplantation: changes over calendar year time, infections and associated factors. Bone Marrow Transplant. 2020; 55: 126-136.
[22] Satlin MJ, Walsh TJ. Multidrug-resistant Enterobacteriaceae, Pseudomonas aeruginosa, and vancomycin-resistant Enterococcus: Three major threats to hematopoietic stem cell transplant recipients. Transpl Infect Dis; 19. Epub ahead of print December 2017. DOI: 10.1111/tid.12762.
[23] HCUP-US NIS overview, https://hcup-us.ahrq.gov/nisoverview.jsp (accessed 20 February 2024).
[24] DuGoff EH, Schuler M, Stuart EA. Generalizing Observational Study Results: Applying Propensity Score Methods to Complex Surveys. Health Services Research. 2014; 49: 284-303.
[25] Han QZ, Chen Y, Yang H, et al. [Incidence of blood stream infections of 1265 patients with hematopoietic stem cell transplantation and analysis of pathogenic bacteria]. Zhonghua Xue Ye Xue Za Zhi. 2017; 38: 930-933.
[26] Gill J, Busca A, Cinatti N, et al. Bacterial Bloodstream Infections after Allogeneic Hematopoietic Stem Cell Transplantation: Etiology, Risk Factors and Outcome in a Single-Center Study. Microorganisms; 11. Epub ahead of print 14 March 2023. DOI: 10.3390/microorganisms11030742.
[27] Barros M, Martinelli R, Rocha H. Enterococcal urinary tract infections in a university hospital: clinical studies. Braz J Infect Dis. 2009; 13: 294-296.
[28] Bhatia S, Robison LL, Francisco L, et al. Late mortality in survivors of autologous hematopoietic-cell transplantation: report from the Bone Marrow Transplant Survivor Study. Blood. 2005; 105: 4215-4222.
[29] Ashton LJ, Le Marsney RE, Dodds AJ, et al. A population-based cohort study of late mortality in adult autologous hematopoietic stem cell transplant recipients in Australia. Biol Blood Marrow Transplant. 2014; 20: 937-945.
[30] Gratwohl A, Brand R, Frassoni F, et al. Cause of death after allogeneic haematopoietic stem cell transplantation (HSCT) in early leukaemias: an EBMT analysis of lethal infectious complications and changes over calendar time. Bone Marrow Transplant. 2005; 36: 757-769.
[31] Avery R, Kalaycio M, Pohlman B, et al. Early vancomycin-resistant enterococcus (VRE) bacteremia after allogeneic bone marrow transplantation is associated with a rapidly deteriorating clinical course. Bone Marrow Transplant. 2005; 35: 497-499.
[32] Tavadze M, Rybicki L, Mossad S, et al. Risk factors for vancomycin-resistant enterococcus bacteremia and its influence on survival after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2014; 49: 1310-1316.
[33] Ferreira AM, Moreira F, Guimaraes T, et al. Epidemiology, risk factors and outcomes of multi-drug-resistant bloodstream infections in haematopoietic stem cell transplant recipients: importance of previous gut colonization. J Hosp Infect. 2018; 100: 83-91.
[34] Peng X, Zhou W, Zhu Y, et al. Epidemiology, risk factors and outcomes of bloodstream infection caused by ESKAPEEc pathogens among hospitalized children. BMC Pediatr. 2021; 21: 188.
[35] Xu W, Fang Y, Zhu K. Enterococci facilitate polymicrobial infections. Trends Microbiol. 2024; 32: 162-177.
[36] Newell KA, Millis JM, Arnow PM, et al. Incidence and outcome of infection by vancomycin-resistant Enterococcus following orthotopic liver transplantation. Transplantation. 1998; 65: 439-442.
[37] Gotur DB, Masud FN, Ezeana CF, et al. Sepsis outcomes in solid organ transplant recipients. Transpl Infect Dis. 2020; 22: e13214.
[38] Linares L, Cervera C, Cofán F, et al. Epidemiology and outcomes of multiple antibiotic-resistant bacterial infection in renal transplantation. Transplant Proc. 2007; 39: 2222-2224.
[39] Farges C, Cointault O, Murris M, et al. Outcomes of solid organ transplant recipients with invasive aspergillosis and other mold infections. Transpl Infect Dis. 2020; 22: e13200.