Le Infezioni in Medicina, n. 4, 506-517, 2024
doi: 10.53854/liim-3204-9
ORIGINAL ARTICLE
Pediatric invasive pneumococcal disease in Bolívar, Colombia: a descriptive cross-sectional study
Wilfrido Coronell Rodríguez1, Andrés Felipe Mora-Salamanca2, José Santacruz-Arias2, Juan Carlos Alvarado-Gonzalez1-3, Laura Saavedra1,3, Hernando Pinzón-Redondo1,3, Nelson Rafael Alvis Guzmán1,4, Nelson Rafael Alvis-Zakzuk2, Josefina Zakzuk1,2
1 Universidad de Cartagena, Cartagena, Bolívar, Colombia;
2ALZAK Foundation, Cartagena, Bolívar, Colombia;
3Fundación Hospital Infantil Napoleón Franco Pareja, Cartagena, Colombia;
4Universidad de la Costa, Barranquilla, Colombia
Article received 12 July 2024 and accepted 29 October 2024
Corresponding author
Josefina Zakzuk
E-mail: jzakzuks@unicartagena.edu.co
SummaRY
Introduction: Invasive pneumococcal disease (IPD) remains a pediatric health challenge despite national vaccination efforts in Colombia. We described the socio-demographic, epidemiological, and clinical characteristics of children (<18 years of age) with IPD at a pediatric reference center in Bolívar, Colombia.
Methods: Descriptive cross-sectional study of all pediatric patients (under 18 years of age) diagnosed with IPD between 2016 and 2023. Data was collected retrospectively from medical records. IPD was defined as identifying Streptococcus pneumoniae (Spn) in blood, cerebrospinal, pleural, synovial, peritoneal, or pericardial fluid. Spn serotyping data was provided by the Colombian National Institute of Health. Descriptive statistics were performed to describe all variables.
Results: Between 2016-2023, we identified fifty-four pediatric IPD cases. Most cases were reported among children in the 2-9 age group (44.4%), male sex (57.4%), low socio-economic strata (100%), and previous medical conditions (61.1%). Half of the patients were vaccinated. Serotyping data were available from 35 (64.8%) isolates. Fifteen Spn serotypes were identified, Spn19A being the most frequent (20.4%). All Spn isolates were vancomycin sensitive, while 34% had meropenem-decreased sensitivity. Three-quarters of the patients (76.0%) were diagnosed with bacteremia (bacteremic pneumonia/meningitis and bacteremia without known focus). The 79.6% of children were admitted to the pediatric intensive care unit (PICU). The median hospitalization days were 13.5 (IQR 5.5-23.5) while the median PICU length of stay was 9.5 (IQR 4-18) days. Nineteen patients died (35.2%).
Conclusion: IPD disproportionately affects vulnerable children, resulting in high PICU admission and mortality rates and prolonged hospital stay in Bolívar, Colombia. In addition, the emergence of resistance to carbapenems is of concern.
Keywords: Pneumococcal infection, serotype, antimicrobial drug resistance, children, cross-sectional study, Colombia.
INTRODUCTION
Invasive pneumococcal disease (IPD) is one of the greatest health challenges worldwide in children under five years of age [1]. IPD encompasses a diverse group of infections characterized by the identification of Streptococcus pneumoniae (Spn) in the blood, cerebrospinal, pleural, synovial, peritoneal, or pericardial fluid (normally sterile sites) [2]. Specifically, pleural (pneumonia/lower respiratory tract infections) and cerebrospinal fluid (CSF) infections (meningitis) were the first and tenth leading causes of mortality in children under five years of age globally in 2019, which is one of the main etiological agents of these infections [3-5].
In Colombia, although national data are not available, the case-fatality risk (CFR) due to IPD was estimated at 7.5% (18 of 239 identified cases) in Bogotá between 2008-2014, with meningitis (33%), bacteremia without an apparent focus of infection (25%), and pneumonia (27%) being the most common diagnoses [6]. From 2006 to 2018, Bolivar was the fourth and fifth leading Colombian department according to the number of invasive Spn isolates reported in children under five years of age and in children between 5 and 14 years of age (4.2% and 4.3%, respectively) [7]. Simultaneously, the predominant serotypes recovered from isolates in children under five years of age at the national level were 14, 19A, 1, 3, 6B, 6A, and 23F, representing 66.6% of all isolates [7].
Several vaccines against Spn are currently available. PCV7 was the first vaccine against Spn for children approved in the United States in 2000 and protected against seven serotypes (4, 6 B, 9V, 14, 18C, 19F, and 23F) [8]. Subsequently, PCV10 (PCV7 + serotypes 1, 5, and 7F) and PCV13 (PCV10 + serotypes 3, 6A, and 19A) were developed [8], with the latter being administered in Colombia according to the Expanded Programme on Immunization (EPI) since May 2022 [9].
Although IPD global incidence and mortality have decreased due to widespread vaccination against Spn, the incidence and clinical disease outcomes vary according to geographical region, Spn serotype, underlying illnesses, and immunization status in children and adults [10-15]. While national vaccination coverage against Spn has improved since PCV7 introduction in 2008 and PCV10 in 2012 (exceeding 90% nationwide coverage in 2019) [16], Colombian EPI aims to vaccinate 100% of children [17]. This is particularly important in Bolívar, where immunization coverage against Spn has historically been below the national average. According to recent local data, the average vaccination coverage in Bolivar reached 80%-89% by 2021, though still lagging behind the national goal of 90%+ coverage as outlined by the Expanded Programme on Immunization [16]. Therefore, we described the sociodemographic, epidemiological, and clinical characteristics of children (<18 years of age) diagnosed with IPD treated at a pediatric reference center in Bolívar, Colombia.
METHODS
Type of Study
Descriptive cross-sectional study of pediatric patients (aged <18 years) diagnosed with IPD between January 2016 and February 2023 at a regional pediatric hospital (Napoleon Franco Pareja Children’s Foundation Hospital) located in Cartagena de Indias, Bolívar, Colombia.
Population
Bolívar, one of the 32 administrative divisions (departments) of Colombia, is located in the Colombian Caribbean Region (Northern Colombia). In 2023, the Departamento Nacional de Estadística (Colombia’s Office of National Statistics) estimated that 2,247,283 citizens live in Bolívar, of which 30.5% (685,633 individuals) were children and adolescents [18].
We included all pediatric patients diagnosed with IPD between 2016 and 2023 at the Napoleon Franco Pareja Children’s Foundation Hospital, a pediatric reference center in the Colombian Caribbean Region, dedicated to medical education and research since 2002 [19].
Data were collected retrospectively from the medical records of all individuals aged <18 years who were diagnosed with IPD. Serotyping data from the Colombian National Institute of Health (Instituto Nacional de Salud, INS) were also included. Patients with confirmed co-infections (two or more microorganisms) from a sterile site were excluded.
Statistical Analysis
Sociodemographic, Spn serotype, and clinical data from 2016-2023 period were included in the analysis. Vaccination status (PCV7 or PCV10 vaccination) was sourced from the Colombian EPI website or PAIweb (https://paiweb2.paiweb.gov.co/login). PAIweb, an electronic health record system, is responsible for compiling vaccination data in Colombia [20]. The patients were classified according to the number of PCV7/10 received (one, two, or three doses). The outcomes were admission to the pediatric or neonatal intensive care unit, hospital stay duration, invasive mechanical ventilation requirement, and hospital discharge status (alive or deceased). Other variables included in the analysis were as follows: age, sex, place of residence, health coverage program, child’s schooling, previous medical conditions, diagnosis, Spn serotype, laboratory results, empiric antimicrobial treatment, and Spn antibiotic resistance profile for nine antibiotics (PEN: Penicillin; AMC: Amoxicillin/Clavulanate; CXM: Cefuroxime; CTX: Cefotaxime; MEM: Meropenem; VAN: Vancomycin; CLI: Clindamycin; SXT: Trimethoprim/sulfamethoxazole; ERY: Erythromycin).
Categorical variables are reported as frequencies and percentages. Continuous variables were expressed as mean or median according to their distribution, and their dispersion was described using standard deviation (SD) or interquartile range (IQR), respectively. All data analyses were performed using STATA 17.0 software (College Station, Texas, USA). A heatmap of antibiotic decreased sensitivity for S. pneumoniae isolates was generated with the Seaborn package using the Python programming language.
Ethical considerations
According to the Helsinki Declaration [21] and the regulations stipulated in Resolution No. 8430 of 1993 by the Ministry of Health of Colombia [22] regarding ethical aspects of health research, this study was a minimal-risk research. Data were evaluated and retrospectively extracted from the medical records; thus, it did not involve direct patient contact was not considered. The research protocol was reviewed and approved by the Research Ethics Committee of the ALZAK Foundation (Acta No. 007, December 14, 2022).
RESULTS
A total of 54 individuals were included in this study. Almost two-thirds (63.0%) were reported in 2017, 2018, and 2022. The number of IPD cases has decreased in 2020 and 2021 (Figure 1). Most cases were reported in children in the 2-9 age group (44.4%), and the number of boys (n=31) was slightly higher than that of girls (n=23). Almost all children were born in the Bolívar Department (94.4%), lived in an urban area (83.3%), were affiliated with the subsidized healthcare system (87.0%), and were not enrolled in school (77.8%) (Table 1). All cases were diagnosed in children from the lowest socioeconomic strata (stratum 1) in Colombia.
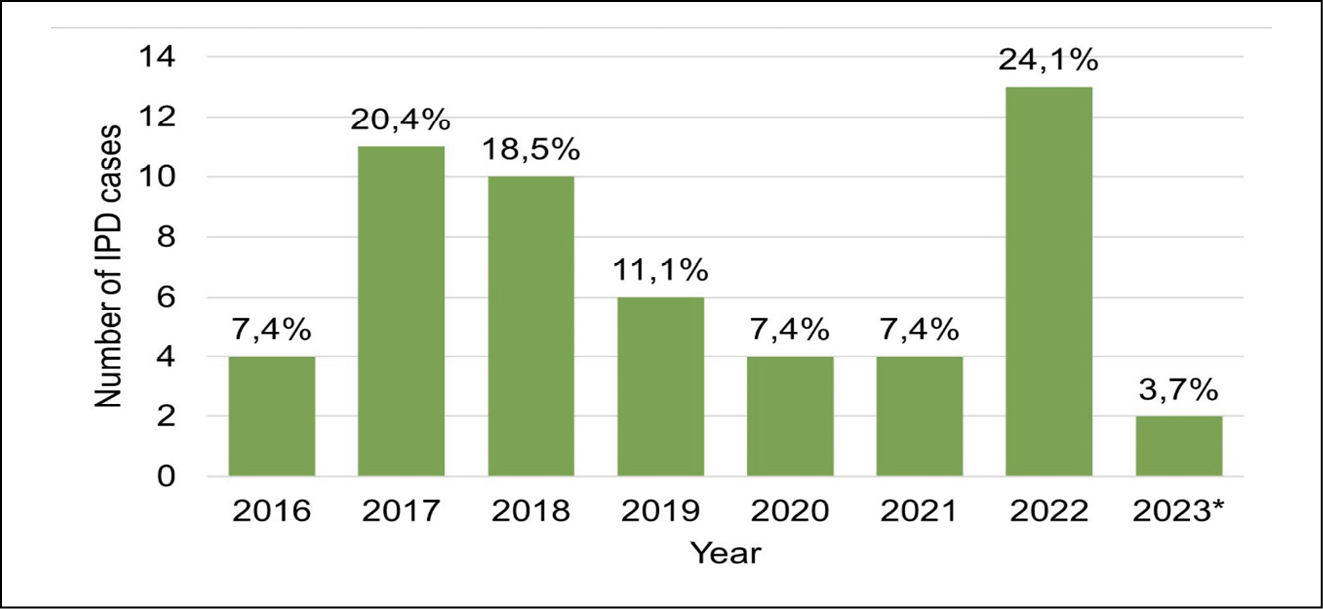
Figure 1 - Number of pediatric invasive pneumococcal disease (IPD) cases by year reported at the Hospital Pediátrico Fundación Infantil Napoleón Franco Pareja from January 2016 to February 2023.
*Cases reported in 2023 only include January and February.
Table 1 - Demographic and clinical characteristics of patients with invasive pneumococcal disease.
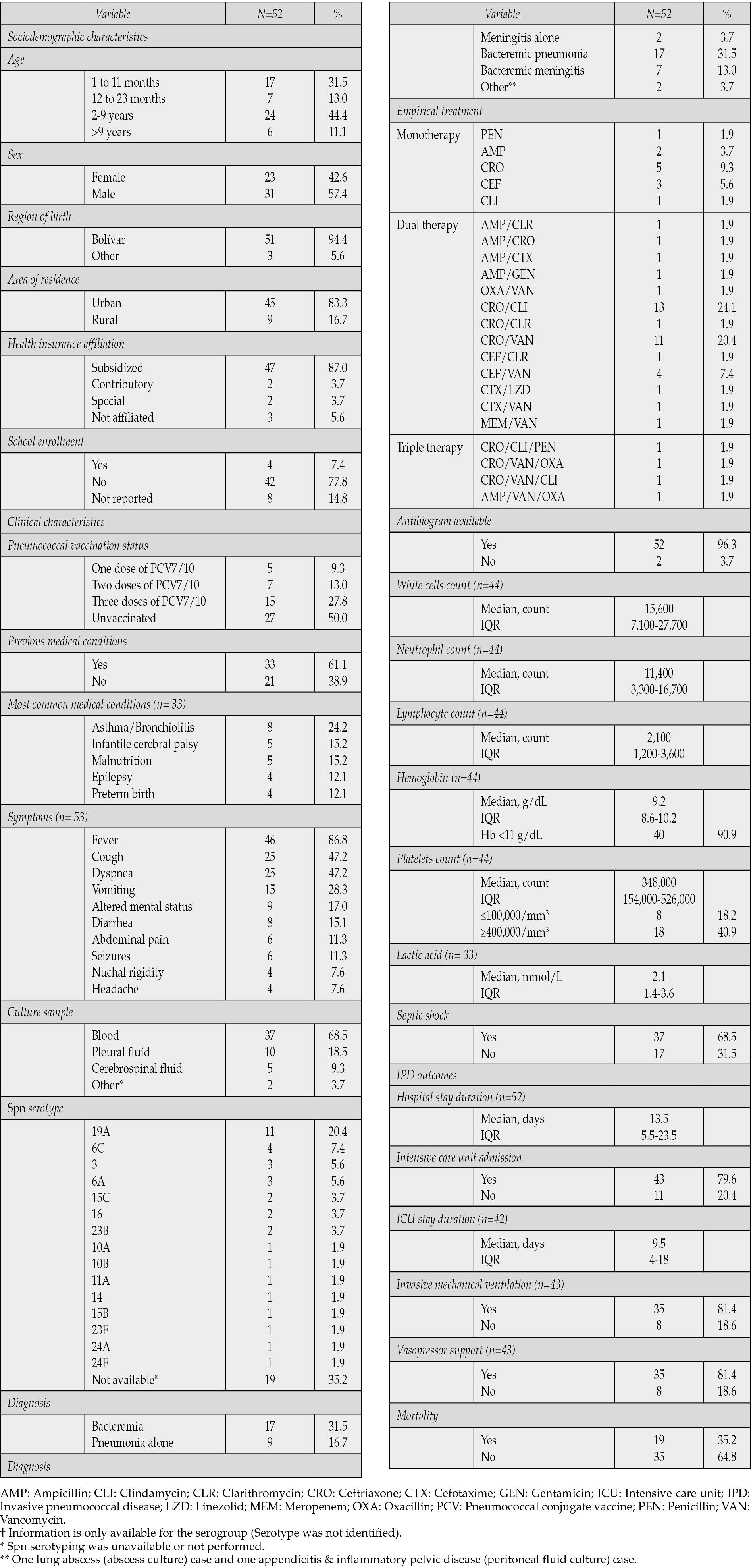
The proportion of patients who received at least one dose of PCV10 was the same as the proportion of children not vaccinated (50%). Among those vaccinated, 15 children completed the vaccination scheme (15 of 27), and only one was vaccinated with PCV7. None of the children were vaccinated with PCV13. Thirty-three children had a previous medical condition (61.1%) and asthma/bronchiolitis was the most common underlying illness (24.2%). The most common symptoms were fever, cough, dyspnea, and vomiting (Table 1).
As described in Table 1, bacteremia was identified in 41 out of 53 cases (76.0%). However, Spn serotyping in blood culture was only available for 35 children. Fifteen different Spn serotypes were identified, with Spn 19A being the most common (20.4%). Other common serotypes were Spn 6C (7.4%), Spn 3 (5.6%), and Spn 6A (5.6%).
Ceftriaxone was the most common empirical antibiotic prescribed to IPD patients (63.0%), followed by vancomycin (38.9%) and clindamycin (29.6%). Dual antibiotic prescription was the most common treatment (70.4%), with ceftriaxone/clindamycin (24.1%) and ceftriaxone/vancomycin (20.4%) used in almost half of the patients. Most children had elevated leukocyte counts and developed septic shock (68.5%). Twenty-six patients had altered platelet levels (59.1%). Notably, 40 of 44 children with hemoglobin measurements were anemic (Hb <11 g/dL) (Table 1).
Clinical outcomes
Regarding clinical outcomes, forty-three patients were admitted to PICU (79.6%). The median hospitalization days was 13.5 (IQR 5.5-23.5) days, while the median PICU length of stay was 9.5 (IQR 4-18) days. Thirty-five (81.4%) children in the PICU required vasopressor support and invasive mechanical ventilation. Nineteen (35.2%) patients died (Table 1).
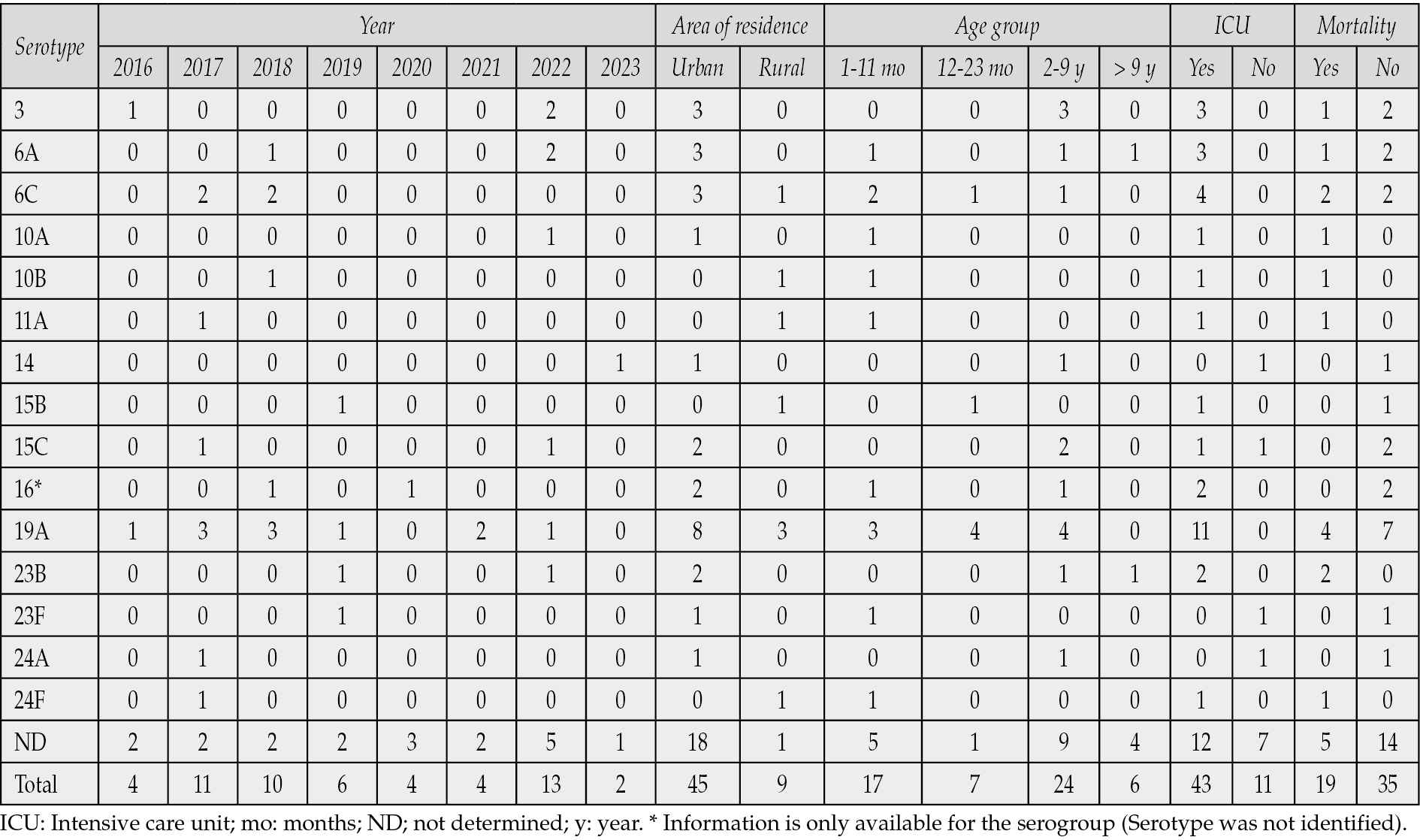
Serotypes 3, 6A, and 19A were detected more frequently in vaccinated children than in unvaccinated children (11 vs. six isolates, respectively). Both PCV10 vaccine-preventable serotypes (14 and 23F) were isolated from unvaccinated children (Table 2). Spn19A isolates were reported every year, except for 2020 and 2023. The PCV10 vaccine-preventable serotypes were exclusively detected in children living in urban areas. PCV13 exclusive vaccine-preventable serotypes were more common among children living in urban areas than among those from rural areas (14 vs. 3 cases, respectively). All IPD cases caused by PCV13 exclusive vaccine-preventable serotypes were admitted to the PICU, whereas PCV10 vaccine-preventable cases did not require intensive care management. All patients with Spn19A were admitted to the PICU, and over one-third died (36.4%) (Table 3).
Table 2 - S. pneumoniae serotype distribution according to patient’s vaccination status.
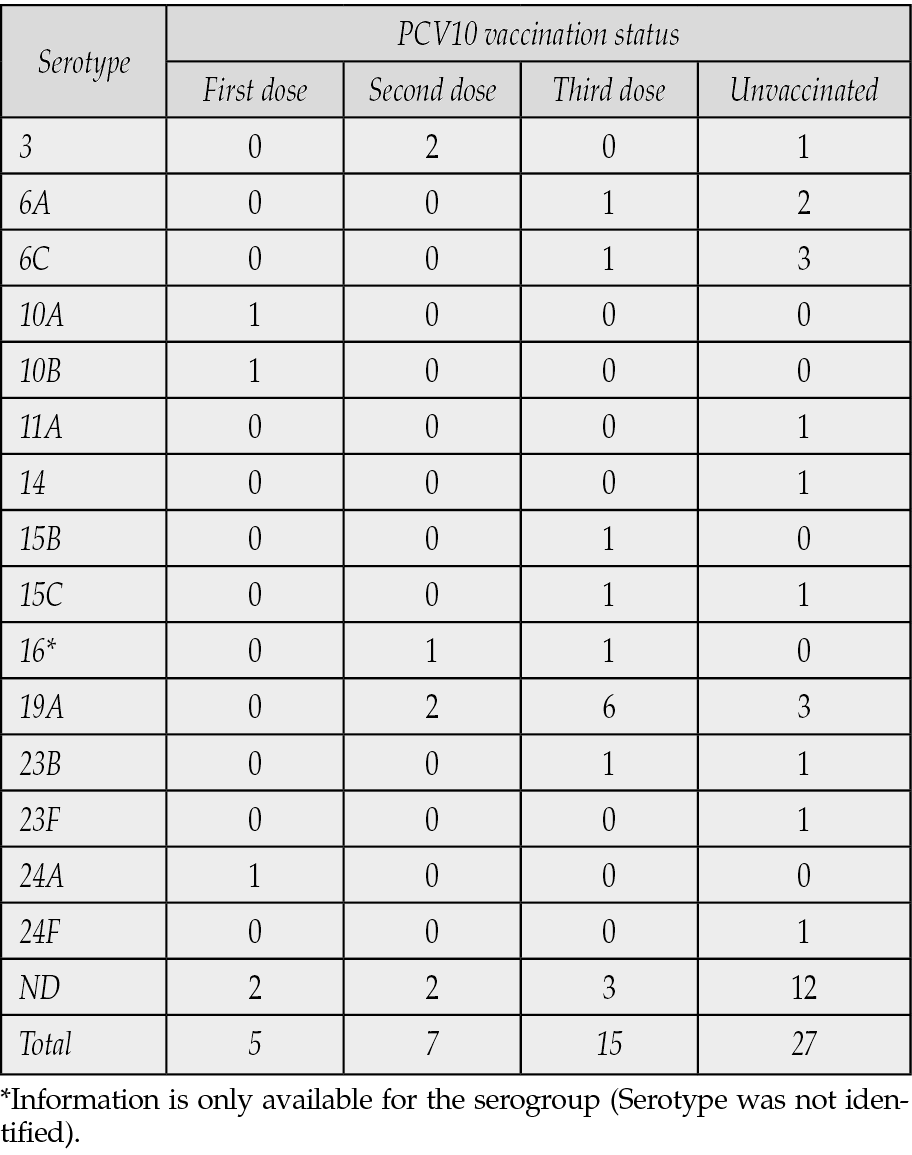
Table 3 - S. pneumoniae serotype distribution by study year, area of residence, age group, and clinical outcome.
The proportion of antibiotic resistance for seven out of the nine antibiotics (PEN, AMC, CXM, MEM, CLI, SXT, and ERY) was higher than 24.5%. However, the proportion of decreased antibiotic sensitivity ranged from 30.2% (CTX) to 50.0% (PEN) in nine of the antibiotics. Remarkably, all Spn isolates were vancomycin-sensitive; however, meropenem resistance was detected (34%) (Figure 2). Considering the Spn serotype, only four serotypes (14, 10A, 15B and 24) were susceptible to all antibiotics evaluated, and five serotypes (6C, 10 B, 19A, 23F, and 24F) showed decreased sensitivity to all antibiotics. Likewise, serogroup Spn16 displayed resistance to all antibiotics, except third generation cephalosporins (CTX) (Figure 3).
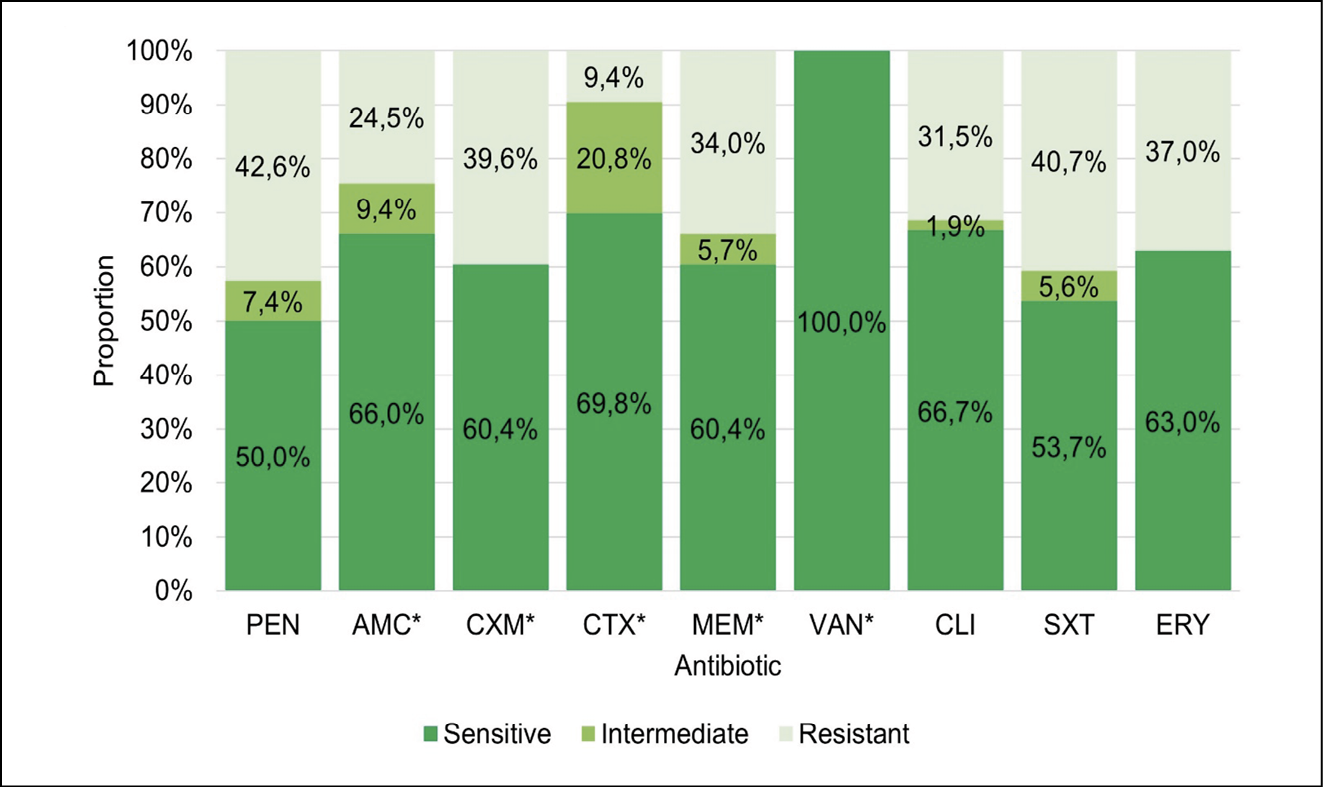
Figure 2 - S. pneumoniae antimicrobial resistance profile among 54 isolates from pediatric patients with invasive pneumococcal disease in Bolívar, Colombia. PEN: Penicillin; AMC: Amoxicillin/Clavulanate; CXM: Cefuroxime; CTX: Cefotaxime; MEM: Meropenem; VAN: Vancomycin; CLI: Clindamycin; SXT: Trimethoprim/sulfamethoxazole; ERY: Erythromycin.
*Data from 53 isolates.
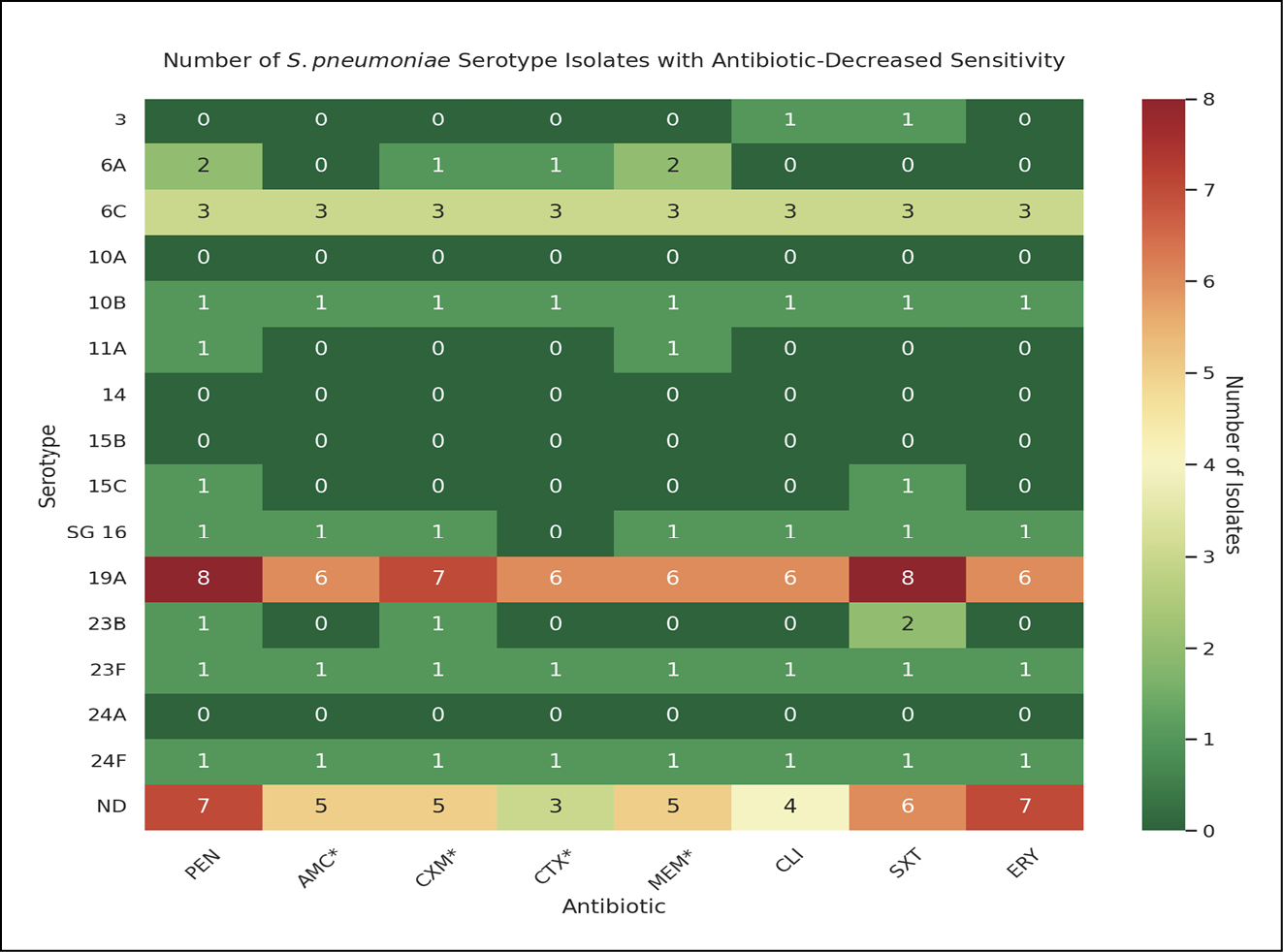
Figure 3 - Number of S. pneumoniae serotype isolates with antibiotic-decreased sensitivity. This heatmap shows the distribution of antibiotic sensitivity among S. pneumoniae serotype isolates, quantifying decreased sensitivity as a sum of resistant and intermediate phenotypes. Color bar indicates the number of isolates with decreased resistance (green: the lowest, red: the highest). Antibiotics are abbreviated as follows: PEN (Penicillin), AMC (Amoxicillin/Clavulanate), CXM (Cefuroxime), CTX (Cefotaxime), MEM (Meropenem), CLI (Clindamycin), SXT (Trimethoprim/sulfamethoxazole), ERY (Erythromycin). *Data from only 53 isolates. SG 16: Information is only available for the serogroup 16; serotype was not identified.
DISCUSSION
This study describes one of the largest cohorts of pediatric patients diagnosed with IPD in Bolívar, Colombia. INS reported 106 Spn isolates in Bolívar from 2016 to 2020, while we analyzed 54 cases from 2016 to 2023 [23]. Thus, pediatric hospital cases accounted for almost half of all pediatric IPD cases in Bolívar. The study cohort primarily consisted of vulnerable children, including those with pre-existing medical conditions, hailing from low-income households, and enrolled in the subsidized healthcare system. High PICU admissions, mortality rates, and prolonged hospital stays were also observed. As observed in other reports from Colombia and other countries in the world, the most common serotype was Spn19A [14, 24].
We found that a higher proportion of IPD cases were reported among boys, children living in urban areas, and children with previous medical conditions. A higher proportion of IPD cases among boys than among girls has been reported previously, although clinical outcome (PICU admissions and mortality) differences attributable to sex have not been observed [24, 25]. A similar proportion of children living in urban settings was reported in our study and in England (83.3% vs. 87.6%) [26]. Concerning previous medical conditions, two case-control studies conducted in the United States reported that underlying illnesses were associated with IPD [13, 27].
In this study, bacteremia was identified as the most prevalent diagnosis, accounting for 76.0% of cases, wherein 40% was primary bacteremia, followed by pneumonia, which constituted 48.2% of cases. Both conditions are frequently reported in the literature, but their proportions are usually smaller. For example, Versluys et al. identified 888 pediatric IPD cases from 1999 to 2019, of which 59.2% and 25.9% were diagnosed with bacteremia and pneumonia, respectively [24]. In 2002, Queensland Health (Queensland, Australia) reported that 65.1% and 23.5% of pediatric IPD cases were categorized as bacteremia and pneumonia, respectively [28]. Marín-Portocarrero et al. reported a 62.1% proportion of bacteremia and 48.3% of pneumonia cases in a descriptive study of IPD cases in a pediatric hospital in Perú, which is similar to our results [29]. Results from a multicentric study in Colombia showed that among IPD cases, 17.9% were diagnosed as primary bacteremia, and most cases had pneumonia [30].
IPD threatens public health because of their high PICU admission and mortality rates. However, our results exceeded those of the previous studies. In a cohort of 253 pediatric patients in Barcelona, Spain, 19.4% were admitted to the PICU, and only two patients died [31]. Versluys et al. reported 5.5% CFR [24]. Jiang et al. reported a CFR of 21.5% CFR among 186 pediatric patients in Beijing [32]. In a previous study conducted in Bogotá, the pediatric IPD CFR was 7.5% and the CFR associated with meningitis was 44% [6]. Another study from Colombia reported a lethality rate of 11.3 %, that was even higher in patients with meningitis (with or without pneumonia) and an admission to PICU rate of 50% [30]. Our study reports even higher case fatality and PICU admission rates, possibly due to the increased proportion of critically ill patients and the presence of multidrug-resistant strains like Spn19A. This was especially higher for meningitis (CFR: 77.8%). These findings may be influenced by several factors, including poor nutritional status, delays in seeking medical care, and limited access to healthcare in low-income populations - factors that were unexplored in this study but may be crucial for future investigation [33, 34]. Moreover, since the Napoleon Franco Pareja Children’s Foundation Hospital is a regional referral center, most inter-institutional patient transfers may involve critically ill patients.
Antimicrobial resistance is a growing problem worldwide and Spn is no exception. Compared to the 2006-2018 national Spn surveillance data, resistance rates to penicillin and erythromycin have increased [7]. Meropenem resistance in Spn is particularly troublesome because carbapenems are among the last antibiotic options. Spn15A was associated with meropenem resistance in Japan, and a multicenter study conducted in China reported a 40.6% meropenem resistance rate, similar to our results [35, 36]. Factors contributing to the rising Spn antimicrobial resistance are the worldwide emergence of Spn19A after PCV7 and PCV10 (especially multidrug-resistant clones; clonal complex 320), antibiotic selective pressure, and serotype replacement [37]. One case of multiple antibiotic resistance (including meropenem) but sensitivity to a third-generation cephalosporin was also found (Spn 16). Although not common, this phenotype has also been described in other studies [38] and may be explained by genetic variations in penicillin binding proteins (PBP). Depending on which PBPs carry mutations, this can lead to varying degrees of susceptibility to antibiotics [39]. Fortunately, vancomycin resistance has not been detected in Colombia, though decreased susceptibility (‘tolerance’) has been reported overseas [40, 41]. In a recent multi-city report from Colombia carried out by the Neumocolombia network, resistance to macrolides was observed in 56% of IPD patients, 43.2% to clindamycin, and 51.5% to trimethoprim/sulfamethoxazole. Rates from our cohort tended to be lower for these antibiotics [30].
Importantly, this study highlights the effectiveness of PCV10 vaccine in preventing IPD. Before PCV10 implementation, the most common causes of IPD in Colombian children under 15 years of age were Spn14, Spn1, and Spn6B [7]. Only two cases of PCV10 vaccine-preventable serotypes (14 and 23F) were reported in this study in two unvaccinated children. Additionally, our results support the introduction of PCV13 into the Colombian Expanded Program on Immunization. One-third of the IPD cases and deaths were caused by PCV13 exclusive vaccine-preventable serotypes (3, 6A, and 19A). Spn19A exhibited the greatest antimicrobial resistance profile among all the identified serotypes. Furthermore, PCV13 is associated with a reduction in the incidence of pneumonia, severe pneumonia, pneumonia hospitalization rates, and pneumonia mortality in children aged <5 years, particularly in high-income countries [10, 42].
As other countries in the world, including the Latin American region, we reported a high circulation rate of Spn19, being the most frequent serotype in our cohort. Another study from Colombia also reported Spn19A as the most frequent serotype found during the study period (51.3%) [30]. An increase in the incidence of invasive infections caused by Spn19A, such as meningitis and pneumonia, has been observed. This serotype has been responsible for outbreaks in several countries, including Brazil [43, 44] from the Latin American Region, and is frequently associated with higher rates of morbidity and mortality compared to other serotypes. Most cases with Spn19A infection were vaccinated with the PCV10. Thus, given the recent vaccine change in Colombia (PCV13), the prevalence in the population over 2 years old, and the prevalence of patients with comorbidities, it would be necessary to evaluate the cost-effectiveness of the implementation of a catch-up strategy and a vaccination strategy for at-risk groups.
Although PCV13 is the current vaccination standard for Spn in the pediatric population, other vaccines have been developed and approved in the United States, the European Union and other countries. For instance, according to the Advisory Committee on Immunization Practices, PCV13 and PCV15 (PCV13 + serotypes 22F and 33F) have been used interchangeably since June 2022 [45]. Our results indicate that there have been no cases of IPD caused by Spn22F or Spn33F in children in Bolívar, Colombia. From 2006 to 2018, only seven IPD cases among children under 15 years of age were attributable to Spn22F (Spn33F caused zero cases) [7].
Similarly, in April 2023, the U.S. Food and Drug Administration approved PCV20 (PCV15 + serotypes 8, 10A, 11A, 12F, and 15 B) for pediatric use [46]. In contrast to PCV15, PCV20 could have prevented three more IPD cases (two fatal) caused by Spn10A, Spn11A, or Spn15B. Nationwide, 96 cases of PCV20 exclusive vaccine-preventable serotypes were documented from 2006 to 2018 [7]. However, continuous epidemiological surveillance in the following years is required to evaluate the effectiveness of PCV13 in Colombia. The introduction of PCV13 will change the distribution of Spn serotypes (serotype replacement) in Colombia, as has previously been observed after PCV7 introduction in 2008 [7] and PCV10 in 2012 [47], and has been reported after PCV13 worldwide [48-50]. Several public health assessments are required at least 5-7 years after post-PCV13 introduction before recommending a new vaccine against Spn (most likely PCV20).
The limitations of this study include its cross-sectional design and retrospective nature. Serotype distribution among cases may be impacted by the current vaccination program, however, no causality can be ascertained. Assessment of potential risk factors for IPD was not possible because this information was barely collected from the medical records derived from the emergency room or ICU management. For example, information about the history of breastfeeding, day care attendance, previous antibiotic use, previous ear infections, household income, household exposure to smoking, and household crowding has been explored previously to identify risk factors associated with IPD [13, 27]. Another area for improvement is the need for more information on interhospital patient transfers. Since the hospital from which the population was studied is a regional pediatric center, not all study patients attended this facility directly. Some patients received critical and specialized care. However, information from the original healthcare institution regarding length of stay, previous treatment, and other medical data was not always available. Therefore, the length of hospital stay and ICU duration may be biased owing to the lack of information. Since the study was limited to 54 patients, it was not possible to perform inferential statistics such as multivariate analyses. In addition, Spn serotyping information accounted for a considerable proportion of missing/unavailable data (35.2%). The PAIweb was implemented in 2012 [51]; thus, the immunization status of children vaccinated before 2012 (n=11) was not recorded in the system. It is possible that some children were vaccinated with PCV7.
Other limitation to be declared is the potential risk of selection bias in our study, as it is based solely on cases from a single institution in the city that serves a pediatric population with limited economic resources. Children included in this study were from families categorized as Colombia’s lowest socioeconomic status (level 1). Socioeconomic stratification (SES) in Colombia is determined according to the classification of residential dwellings in a city or town, ranging from the lowest SES level (level 1) to the highest (level 6) [52]. Additionally, most children were not enrolled in school and belonged to a subsidized health insurance program. Since the study did not include children from middle- or high-income families, we cannot generalize that our results are entirely representative of Cartagena.
This study summarizes the socio-demographic, epidemiological, and clinical characteristics of 54 children diagnosed with IPD in Bolívar, Colombia, between 2016 and 2023. The results of this study indicate that IPD continues to be a severe cause of morbidity and mortality among children, despite vaccination programs and medical advances. These findings also suggest that, in general, a replacement in the Spn serotype distribution has taken place in Bolívar since PCV10 implementation. Overall, this study strengthens the idea that PCV13 introduction into Colombian EPI is necessary to prevent a significant number of IPD cases and deaths, mainly due to Spn19A. Further studies could assess the long-term effects of PCV13 on the epidemiology, antimicrobial resistance profile, and clinical presentation of Spn-associated infections, particularly IPD. This study’s findings have several important implications. For instance, greater efforts are needed to ensure that 100% vaccination rates among children are achieved in Colombia, as IPD vaccine-preventable cases are still observed. Moreover, to better understand the impact of PCV13 in Colombia and recommend new vaccines, most Spn isolates should be successfully serotyped and prospective studies should be conducted.
Conflict of interest
To carry out this study, the authors declare having received funding from Pfizer through a grant.
Funding
This work was supported by Pfizer (Grant number 76501397) and ALZAK Foundation.
Acknowledgements
We want to thank the administrative ALZAK Foundation team and Fundación Hospital Infantil Napoleón Franco Pareja for their support during the development of this study.
REFERENCES
[1] Wahl B, O’Brien KL, Greenbaum A, et al. Burden of Streptococcus pneumoniae and Haemophilus influenzae type b disease in children in the era of conjugate vaccines. Lancet Glob Health. 2018; 6(7): 744-757.
[2] Randle E, Ninis N, Inwald D. Invasive pneumococcal disease. Arch Dis Child Educ Pract. 2011; 96(5): 183-190.
[3] Institute of Health Metrics. Global burden of disease study 2019 (GBD 2019) disability weights. GHDx. 2020. Available at: http://www.healthdata.org/gbd/2019 [accessed 11, October, 2024].
[4] Torres A, Cilloniz C, Niederman MS, et al. Pneumonia. Nat Rev Dis Primers. 2021; 7(1): 25.
[5] Oordt-Speets AM, Bolijn R, van Hoorn RC, et al. Global etiology of bacterial meningitis: a systematic review and meta-analysis. PLoS One. 2018; 13(6).
[6] Rojas JP, Leal AL, Patiño J, et al. Caracterización de pacientes fallecidos por enfermedad neumocóccica invasiva en la población infantil de Bogotá, Colombia. Rev Chil Pediatr. 2016; 87(1): 48-52.
[7] Instituto Nacional de Salud (INS). Vigilancia por laboratorio de aislamientos invasores de Streptococcus pneumoniae Colombia 2006-2018. Instituto Nacional de Salud. 2019:1-16. Available at: https://www.ins.gov.co/buscador-eventos/Informacin%20de%20laboratorio/Vigilancia%20por%20laboratorio%20S.%20pneumoniae%202006-2018.pdf [accessed 11, October, 2024].
[8] Kellner JD. Update on the success of the pneumococcal conjugate vaccine. Paediatr Child Health. 2011; 16(4): 233-236.
[9] Ministerio de Salud y Protección Social. PAI incorporó actualización de vacuna contra el neumococo. Bol Prensa. 2022. Available at: https://www.minsalud.gov.co/Paginas/PAI-incorporo-actualizacion-de-vacuna-contra-el-neumococo.aspx [accessed 11, October, 2024].
[10] Reyburn R, Tsatsaronis A, von Mollendorf C, et al. Systematic review on the impact of the pneumococcal conjugate vaccine ten valent (PCV10) or thirteen valent (PCV13) on all-cause, radiologically confirmed and severe pneumonia hospitalisation rates and pneumonia mortality in children 0-9 years old. J Glob Health. 2023; 13: 05002.
[11] Greenwood B. The epidemiology of pneumococcal infection in children in the developing world. Philos Trans R Soc Lond B Biol Sci. 1999; 354(1384): 777-785.
[12] Song JY, Nahm MH, Moseley MA. Clinical implications of pneumococcal serotypes: invasive disease potential, clinical presentations, and antibiotic resistance. J Korean Med Sci. 2013; 28(1): 4.
[13] Levine OS, Farley M, Harrison LH, et al. Risk factors for invasive pneumococcal disease in children: a population-based case-control study in North America. Pediatrics. 1999; 103(3).
[14] Narváez PO, Gomez-Duque S, Alarcon JE, et al. Invasive pneumococcal disease burden in hospitalized adults in Bogotá, Colombia. BMC Infect Dis. 2021; 21(1): 1059.
[15] Hu T, Song Y, Done N, et al. Incidence of invasive pneumococcal disease in children with commercial insurance or Medicaid coverage in the United States before and after the introduction of 7- and 13-valent pneumococcal conjugate vaccines during 1998–2018. BMC Public Health. 2022; 22(1): 1677.
[16] Ministerio de Salud y Protección Social. Coberturas en vacunación departamental - Programa Ampliado de Inmunizaciones (PAI). Vacunación. 2020. Available at: https://www.minsalud.gov.co/salud/publica/Vacunacion/Paginas/pai.aspx [accessed 11, October, 2024].
[17] Ministerio de Salud y Protección Social. Lineamientos técnicos y operativos para la transición de la vacuna polisacárida contra el neumococo de PCV10 a PCV13 en Colombia 2022. Ministerio de Salud y Protección Social. 2022:1-36. Available at: https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/VS/PP/ET/lineamiento-tecnico-operativo-transicion-vacuna-polisacarda-contra-neumococo-pcv10-pcv13-colombia-2022.pdf [accessed 11, October, 2024].
[18] Departamento Administrativo Nacional de Estadística (DANE). Proyecciones de población departamentales por área. DANE. 2020. Available at: https://www.dane.gov.co/salud/publica/Vacunacion/Paginas/pai.aspx [accessed 11, October, 2024].
[19] Pérez IST. Un hospital pediátrico de referencia en el Caribe colombiano como espacio para la docencia e investigación. Investig En Educ Médica. 2018; 7(25): 87. doi: 10.1016/j.riem.2017.01.150 [accessed 11, October, 2024].
[20] Ministerio de Salud y Protección Social. PAIWEB se actualiza para proceso de vacunación contra el covid-19. Ministerio de Salud y Protección Social. 2021. Available at: https://www.minsalud.gov.co/Paginas/PAIWEB-se-actualiza-para-proceso-de-vacunacion-contra-el-covid-19.aspx [accessed 11, October, 2024].
[21] Asociación Médica Mundial. Declaración de Helsinki de la AMM – Principios éticos para las investigaciones médicas en seres humanos. Asociación Médica Mundial. 1964. Available at: https://www.wma.net/es/policies-post/declaracion-de-helsinki-de-la-amm-principios-eticos-para-las-investigaciones-medicas-en-seres-humanos/ [accessed 11, October, 2024].
[22] Ministerio de Salud y Protección Social. Resolución 8430 de 1993. Ministerio de Salud y Protección Social. 1993:1-60. Available at: https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/DE/DIJ/RESOLUCION-8430-DE-1993.PDF [accessed 11, October, 2024].
[23] Instituto Nacional de Salud (INS). Vigilancia por laboratorio de Streptococcus pneumoniae en Colombia, 2016-2020. Instituto Nacional de Salud. 2021:1-4. Available at: https://www.ins.gov.co/BibliotecaDigital/vigilancia-por-laboratorio-s-pneumoniae%20-colombia-2016-2020.pdf [accessed 11, October, 2024].
[24] Versluys KA, Eurich DT, Marrie TJ, et al. Invasive pneumococcal disease and long-term outcomes in children: A 20-year population cohort study. Lancet Reg Health Am. 2022; 14: 100341.
[25] Wagenvoort GHJ, Sanders EAM, Vlaminckx BJ, et al. Sex differences in invasive pneumococcal disease and the impact of pneumococcal conjugate vaccination in the Netherlands, 2004 to 2015. Eurosurveillance. 2017; 22(10): 6-18.
[26] Mohanty S, Podmore B, Cuñado Moral A, et al. Incidence of pneumococcal disease from 2003 to 2019 in children ≤17 years in England. Pneumonia. 2023; 15(1): 2.
[27] Pilishvili T, Zell ER, Farley MM, et al. Risk factors for invasive pneumococcal disease in children in the era of conjugate vaccine use. Pediatrics. 2010; 126(1).
[28] Queensland Health. Invasive pneumococcal disease in children aged under five years in Queensland, in 2002. Queensland Health. 2003; 1-14. Available at: https://www.health.qld.gov.au/__data/assets/pdf_file/0025/422287/20434.pdf [accessed 11, October, 2024].
[29] Marín-Portocarrero JG, Quispe-Sanchez A, Charca-Rodriguez F de M, et al. Enfermedad neumocócica invasiva en pacientes de un hospital pediátrico de Perú, 2017-2020. Rev Peru Med Exp Salud Pública. 2022; 39(4): 469-473.
[30] Camacho-Moreno G, Leal AL, Patiño-Niño J, et al. Serotype distribution, clinical characteristics, and antimicrobial resistance of pediatric invasive pneumococcal disease in Colombia during PCV10 mass vaccination (2017–2022). Front Med. 2024; 11: 1380125.
[31] Izquierdo C, Ciruela P, Hernández S, et al. Pneumococcal serotypes in children, clinical presentation and antimicrobial susceptibility in the PCV13 era. Epidemiol Infect. 2020; 48: 1-37.
[32] Jiang M, Wang X, Zhu L, et al. Clinical characteristics, antimicrobial resistance, and risk factors for mortality in paediatric invasive pneumococcal disease in Beijing, 2012–2017. BMC Infect Dis. 2022; 22(1): 338.
[33] Ricketson LJ, Vanderkooi OG, Wood ML, et al. Clinical features and outcomes of serotype 19A invasive pneumococcal disease in Calgary, Alberta. Can J Infect Dis Med Microbiol. 2014; 25(2).
[34] Lai CY, Huang LM, Lee PY, et al. Comparison of invasive pneumococcal disease caused by serotype 19A and non-19A pneumococci in children: more empyema in serotype 19A invasive pneumococcal disease. J Microbiol Immunol Infect. 2014; 47(1): 23-27.
[35] Ono T, Watanabe M, Hashimoto K, et al. Serotypes and antibiotic resistance of Streptococcus pneumoniae before and after the introduction of the 13-valent pneumococcal conjugate vaccine for adults and children in a rural area in Japan. Pathogens. 2023; 12(3): 493.
[36] Chen T, Li W, Wang F, et al. Antibiotics prescription for targeted therapy of pediatric invasive pneumococcal diseases in China: a multicenter retrospective study. BMC Infect Dis. 2021; 21(1): 1156.
[37] Ruiz García Y, Nieto Guevara J, Izurieta P, et al. Circulating clonal complexes and sequence types of Streptococcus pneumoniae serotype 19A worldwide: The importance of multidrug resistance: A systematic literature review. Expert Rev Vaccines. 2021; 20(1): 45-57.
[38] Varghese R, Basu S, Neeravi A, et al. Emergence of meropenem resistance among cefotaxime non-susceptible Streptococcus pneumoniae: evidence and challenges. Front Microbiol. 2022; 12: 810414.
[39] Hakenbeck R, Brückner R, Denapaite D, et al. Molecular mechanisms of β-lactam resistance in Streptococcus pneumoniae. Future Microbiol. 2012; 7(3): 395-410.
[40] Normark BH, Novak R, Ortqvist A, et al. Clinical isolates of Streptococcus pneumoniae that exhibit tolerance of vancomycin. Clin Infect Dis. 2001; 32(4): 552-558.
[41] Ataee RA, Habibian S, Mehrabi-Tavana A, et al. Determination of vancomycin minimum inhibitory concentration for ceftazidime-resistant Streptococcus pneumoniae in Iran. Ann Clin Microbiol Antimicrob. 2014; 13(1): 53.
[42] Aternina-Caicedo A, Smith AD, Buchanich J, et al. Reductions in childhood pneumonia mortality after vaccination in the United States. Pediatr Infect Dis J. 2023; 42(8): 723-729.
[43] Brandileone MC, Almeida SC, Minamisava R, et al. Distribution of invasive Streptococcus pneumoniae serotypes before and 5 years after the introduction of 10-valent pneumococcal conjugate vaccine in Brazil. Vaccine. 2018; 36(18): 2559-2566.
[44] Cassiolato AP, Almeida SC, Andrade AL, et al. Expansion of the multidrug-resistant clonal complex 320 among invasive Streptococcus pneumoniae serotype 19A after the introduction of a ten-valent pneumococcal conjugate vaccine in Brazil. PLoS One. 2018; 13(6).
[45] Kobayashi M, Farrar JL, Gierke R, et al. Use of 15-valent pneumococcal conjugate vaccine among U.S. children: Updated recommendations of the advisory committee on immunization practices - United States, 2022. MMWR Morb Mortal Wkly Rep. 2022; 71(37): 1174-1181.
[46] Food and Drugs Administration (FDA). PREVNAR 20. Vaccines Blood Biol. Published online June 2023. Available at: https://www.fda.gov/vaccines-blood-biologics/vaccines/prevnar-20 [accessed 11, October, 2024].
[47] Gutiérrez-Tobar IF, Londoño-Ruiz JP, Mariño-Drews C, et al. Epidemiological characteristics and serotype distribution of culture-confirmed pediatric pneumococcal pneumonia before and after PCV 10 introduction, a multicenter study in Bogota, Colombia, 2008–2019. Vaccine. 2022; 40(20): 2875-2883.
[48] Wu CJ, Lai JF, Huang IW, et al. Serotype distribution and antimicrobial susceptibility of Streptococcus pneumoniae in pre- and post-PCV7/13 eras, Taiwan, 2002–2018. Front Microbiol. 2020; 11: 18977.
[49] Løchen A, Croucher NJ, Anderson RM. Divergent serotype replacement trends and increasing diversity in pneumococcal disease in high-income settings reduce the benefit of expanding vaccine valency. Sci Rep. 2020; 10(1): 18977.
[50] Lee JK, Yun KW, Choi EH, et al. Changes in the serotype distribution among antibiotic-resistant carriage Streptococcus pneumoniae isolates in children after the introduction of the extended-valency pneumococcal conjugate vaccine. J Korean Med Sci. 2017; (9): 1431-1438.
[51] Ministerio de Salud y Protección Social. Minsalud implementa Sistema de Información Nominal de Vacunación. Minsalud. 2012. Available at: https://www.minsalud.gov.co/Paginas/Minsalud%20implementa%20Sistema%20de%20Informaci%C3%B3n%20Nominal%20de%20vacunaci%C3%B3n.aspx [accessed 11, October, 2024].
[52] Congreso de Colombia. Ley 142 de 1994. Congreso de Colombia. 1994; 1-60. Available at: https://www.funcionpublica.gov.co/eva/gestornormativo/norma.php?i=2752 [accessed 11, October, 2024].