Le Infezioni in Medicina, n. 4, 489-498, 2024
doi: 10.53854/liim-3204-7
ORIGINAL ARTICLE
Clinical and epidemiological aspects of spondylodiscitis in a tertiary care hospital in South India
Latha Thimmappa1, Saraswathi Hebbar2, Shyamasunder N Bhat3
1 College of Nursing, All India Institute of Medical Sciences, Kalyani, West Bengal, India;
2 Manipal School of Nursing, Manipal Academy of Higher Education, Manipal, Karnataka, India;
3 Department of Orthopedics, Kasturba Medical College, Manipal, Manipal Academy of Higher Education, Manipal, Karnataka, India
Article received 11 June 2024 and accepted 10 October 2024
Corresponding author
Shyamasunder N Bhat
E-mail: shyambhat.n@manipal.edu
SummaRY
This study aims to bridge the informational gap regarding the clinical and epidemiological aspects of spondylodiscitis in India, addressing the dearth of substantial evidence in this domain. This study was conducted in a tertiary care hospital over three years, involving 145 adult patients diagnosed with spondylodiscitis. Among them, 28 (19.3%) had Brucellar spondylodiscitis with a younger mean age of 40.1 years, 76 (52.4%) had tubercular spondylodiscitis with a higher mean age of 50.7 years, and 27 (18.6%) had pyogenic spondylodiscitis. Common symptoms included pain while walking (82.1% Brucellar, 93.4% TB, 77.8% pyogenic), fever (39.3% Brucellar, 38.2% TB, 33.3% pyogenic), and limb weakness (25% Brucellar, 46.1% TB, 66.7% pyogenic). CRP (mg/L) levels were elevated in Brucellar (mean 58.75) and pyogenic (mean 60.4) spondylodiscitis patients, with debridement and decompression performed in 50% Brucellar, 55.3% tubercular, and 70.4% pyogenic patients. The study reveals the clinical and epidemiological aspects of spondylodiscitis in South India, contributing to the enrichment of existing knowledge in diagnosis and management.
Keywords: Brucellosis, epidemiological monitoring, india; spinal tuberculosis, spondylodiscitis.
INTRODUCTION
Spondylodiscitis is a rare but significant infectious disease that affects the spinal column, including the intervertebral discs and adjacent vertebral bodies [1-3]. This inflammatory disorder can be caused by various microorganisms, including bacteria, fungi, and mycobacteria [2, 4-6]. The incidence of spondylodiscitis has been increasing in recent years, making it a growing public concern worldwide [7-11]. Several studies have reported the incidence of spondylodiscitis in various populations worldwide.
In England and Wales, an incidence of 3.67/100,000 persons per year represents a 150% increase from 1995-1999. The estimated the annual incidence in Europe was 0.4 to 2.4/100,000 [7, 9]. Data from Denmark reported an incidence of 5.8/100,000 persons per year, while Germany reported an age-standardized incidence of 30/250,000 persons per year [10]. In South Korea, the incidence rate of pyogenic spondylodiscitis per 100,000 people was 33.75 in 2019 [12].
In India, the incidence of spondylodiscitis is not well documented, but it is believed to be relatively high due to the prevalence of TB spine [13].
In India, spondylodiscitis is a significant cause of morbidity, and the incidence of the disease is increasing. According to previous studies, infective spondylodiscitis shows a different microbiological profile, and pyogenic spondylodiscitis is an uncommon but important infection [13].
Spondylodiscitis is caused by a variety of organisms in India. Durovic (2020) identified Parvimonas micra, an anaerobic bacterium, as a rare but significant cause, particularly in cases with recent dental inflammation [14]. Aspergillus species were also found to be a causative organism in spondylodiscitis cases [15]. Jain (2020) highlighted the potential influence of genetics, gut microbiome, and environmental factors on the disease, suggesting a complex interplay of factors in India [16]. Lastly, Hughes (2020) reported a unique case of spondylodiscitis caused by Phyllobacterium myrsinacearum, an organism not previously associated with human infections [17].
Recent studies have investigated the prevalence, clinical features, and management strategies of spondylodiscitis. For instance, a study conducted in Iran reported that Brucella is a significant cause of spondylodiscitis and should be considered in the differential diagnosis of patients presenting with back pain and fever [18]. The clinical features of spondylodiscitis can vary depending on the severity of the infection and the causative organism. Common symptoms include back pain, fever, and neurological deficits such as weakness or numbness in the extremities [19, 20].
The diagnosis of spondylodiscitis is complex and often delayed due to its non-specific symptoms and the need for various diagnostic tests. Imaging studies, blood culture, and biopsy help diagnose the disease [9, 21-23].
The present study addresses the lack of sufficient evidence regarding the clinical and epidemiological aspects of spondylodiscitis in India. The existing gap in knowledge has prompted the need for this investigation. Therefore, the study aimed to identify the prevalence of spondylodiscitis and find its association with tuberculosis spine, spondylitis and brucellosis in a tertiary care hospital in South India. This study aimed to determine the clinical features, diagnostic measures, and management strategies of spondylodiscitis in a tertiary care hospital in South India.
PATIENTS AND METHODS
Study design and setting
This study was conducted at a tertiary care hospital that provides specialized medical care and advanced diagnostic and treatment services. Adopting a retrospective design, the research aimed to comprehensively analyze the clinical-epidemiological aspects of spondylodiscitis among patients admitted to this healthcare facility.
Sample and sample size determination
Between January 1, 2017 and December 31, 2019, adult patients (≥18 years), both men and women diagnosed with spondylodiscitis, were included in the study. An attempt was made in all cases of clinically suspected spondylodiscitis to identify the causative organism by serology, culture and histopathology. The diagnosis of brucellar spondylodiscitis was established either by detecting antibodies using the standard agglutinin test or by culturing blood/bone tissue, often complemented by Magnetic Resonance Imaging (MRI). Tubercular spondylodiscitis diagnosis relied mainly on microbiological and histopathological tests frequently accompanied by radio imaging studies like MRI. Pyogenic spondylodiscitis diagnosis involved culturing blood or tissue, sometimes combined with MRI. Additionally, there were patients with other forms of spondylodiscitis, including fungal and parasitic, which were detected through various investigations. Patients who did not complete their diagnostic workups for various reasons were excluded. This resulted in a total of 145 eligible patients being identified over these three years.
Patients presenting with manifestations of spondylodiscitis were examined by a spine expert in the Department of Orthopedics, who made a provisional diagnosis. To establish this diagnosis, standard radiographs and magnetic resonance imaging (MRI) scans were performed on the affected spine segments. Blood tests were conducted for all suspected patients, including a complete blood count, erythrocyte sedimentation rate, C-reactive protein, and blood cultures. A standard agglutination test was also performed for patients suspected of having brucellosis.
Patients with neurological deficits or rapidly deteriorating conditions underwent surgical treatments, such as decompression or debridement, with or without spinal stabilization. A closed biopsy was performed in the absence of a precise surgical indication. Biopsy tissues underwent bacterial cultures, GeneXpert®, and histopathology analysis.
Data collection approach
The primary data source for this investigation comprised the meticulous review of medical records pertaining to patients admitted with spondylodiscitis. Demographic proforma was used to collect the basic demographic details of the included patients. A comprehensive clinical-epidemiological checklist was prepared to extract pertinent information. This checklist encompassed detailed parameters, including presenting symptoms, diagnostic methodologies employed, and the spectrum of treatment modalities administered to the patients. In cases where certain data points were found to be incomplete within the medical records, they were appropriately identified and treated as missing data in the subsequent analysis to maintain the integrity of the study.
Statistical analysis
The collected data were coded and entered into SPSS 23. Descriptive statistics, such as frequency and percentage, were calculated to summarize the demographic and clinical characteristics of patients.
Ethical considerations and clearances
Prior to the commencement of the study, necessary ethical clearances were obtained from the Institutional Ethics Committee (IEC). Given the retrospective nature of the study, individual consent was not sought. However, stringent measures were implemented to maintain the anonymity of the subjects.
RESULTS
This study investigated the clinical and epidemiological profile of spondylodiscitis among patients admitted to a tertiary care hospital. Data were retrospectively gathered from medical records of individuals diagnosed with spondylodiscitis, with a focus on demographic characteristics, clinical presentations, laboratory parameters, and treatment strategies among distinct infectious categories - brucellar, tubercular, pyogenic spondylodiscitis, and other categorized cases. The “Others” category included patients with spondylodiscitis not caused by Brucella, tuberculosis, or pyogenic infections; specifically, it included cases of fungal, parasitic, or immunodeficiency-associated spondylodiscitis.
The total sample comprised 145 patients, distributed across various types of spondylodiscitis: Brucellar (n=28), Tubercular (n=76), Pyogenic (n=27), and others (n=14). This section comprehensively analyses the demographic data and associated clinical features observed within each spondylodiscitis type.
Demographic characteristics
The demographic profile of patients with spondylodiscitis varied across different infectious types (Table 1). Brucellar spondylodiscitis patients (n=28) had a relatively younger mean age of 40.1 years (SD 14.6), while tubercular spondylodiscitis patients (n=76) exhibited a higher mean age of 50.7 years (SD 16.9). Pyogenic spondylodiscitis patients (n=27) and those categorized under ‘Others’ (n=14) had mean ages of 55.5 years (SD 17.6) and 52.3 years (SD 8.8), respectively. Gender distribution also varied among the different types of spondylodiscitis, with discernible differences in occupation and duration of hospital stay. Patients with brucellar spondylodiscitis needed a more extended hospital stay compared with others.
Table 1 - Demographic characteristics of patients with spondylodiscitis.
Commonly affected areas
Specific areas of spinal involvement were observed among different types of spondylodiscitis (Figure 1). Brucellar spondylodiscitis commonly affected the lumbar region (Figure 1A), while tubercular spondylodiscitis predominantly involved the thoracic area (Figure 1B). Pyogenic spondylodiscitis frequently affected the lumbosacral region (Figure 1C), whereas the ‘Others’ category predominantly presented with lumbar involvement (Figure 1D).
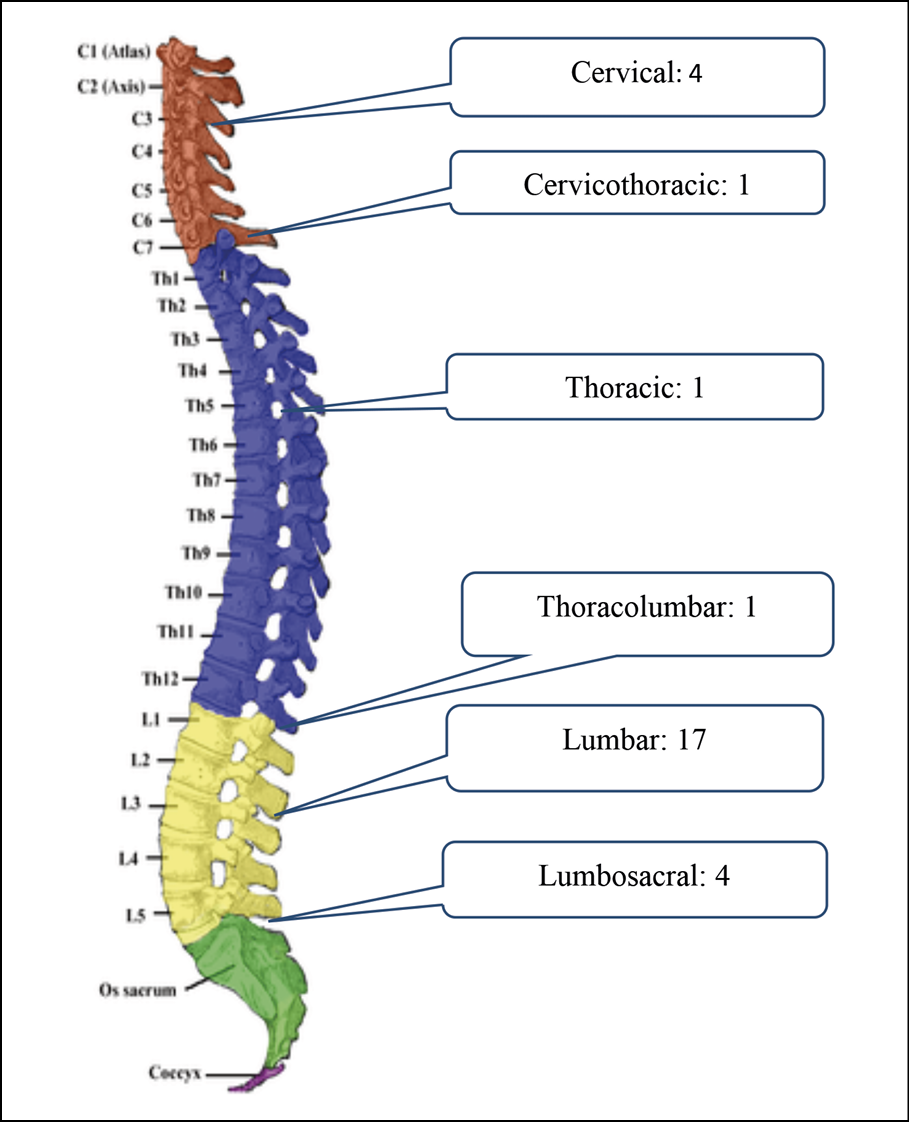
Figure 1A - Location of Brucellar Spondylodiscitis.

Figure 1B - Location of Tubercular Spondylodiscitis.
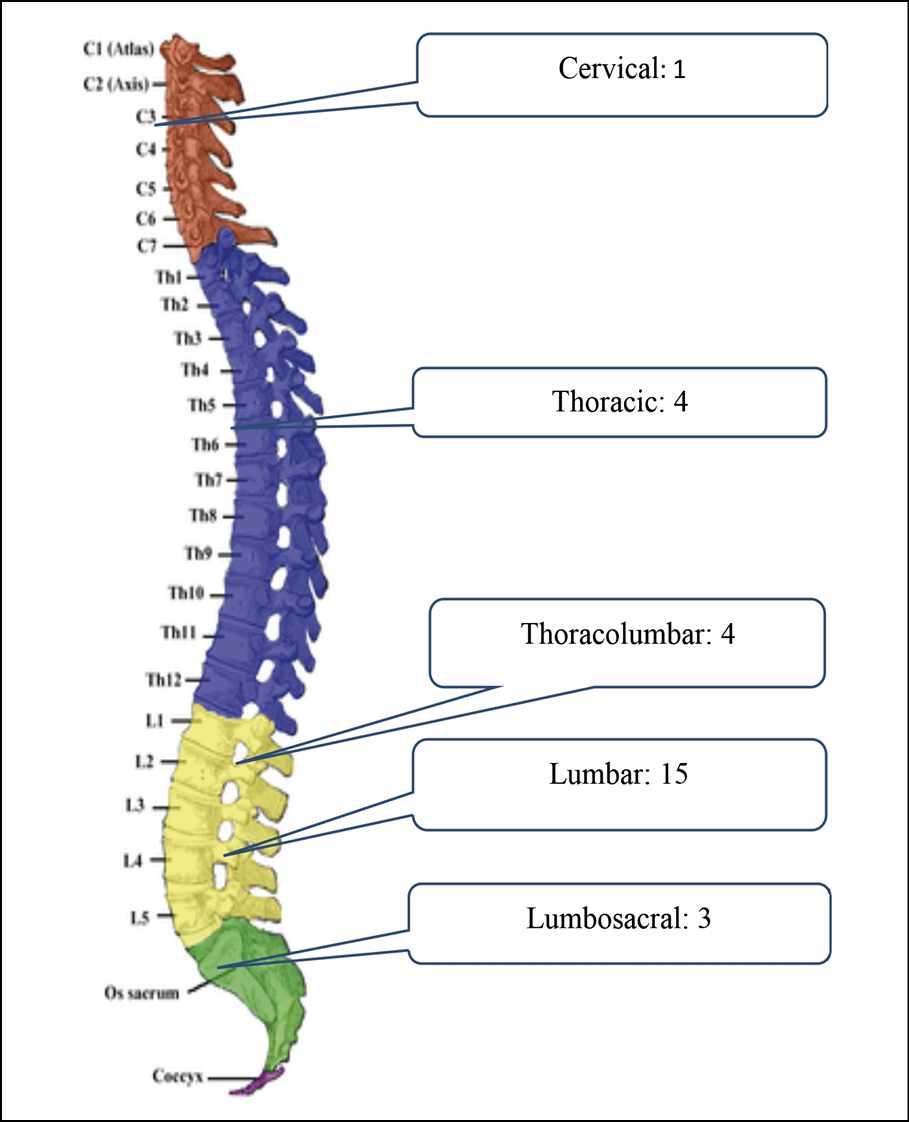
Figure 1C - Location of Pyogenic Spondylodiscitis.
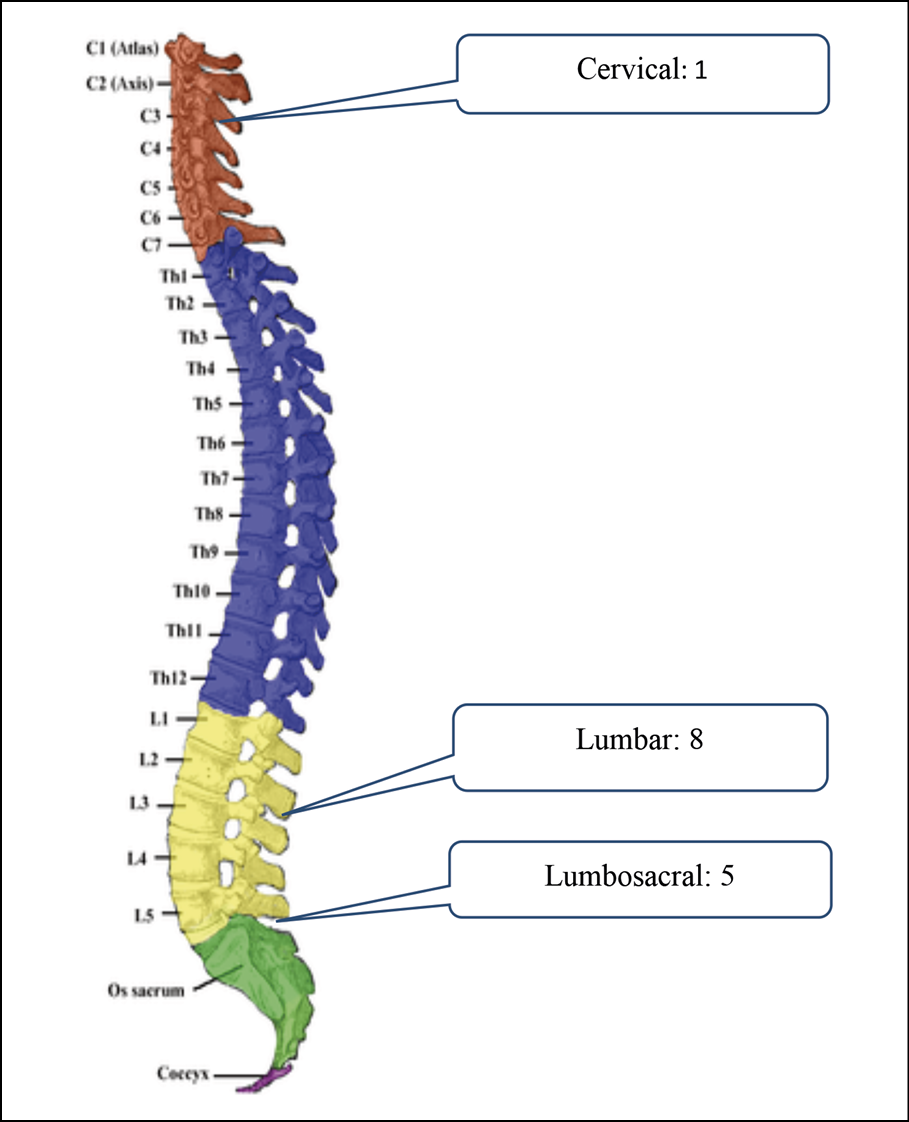
Figure 1D - Location of Other type of Spondylodiscitis.
Risk factors and co-morbidities
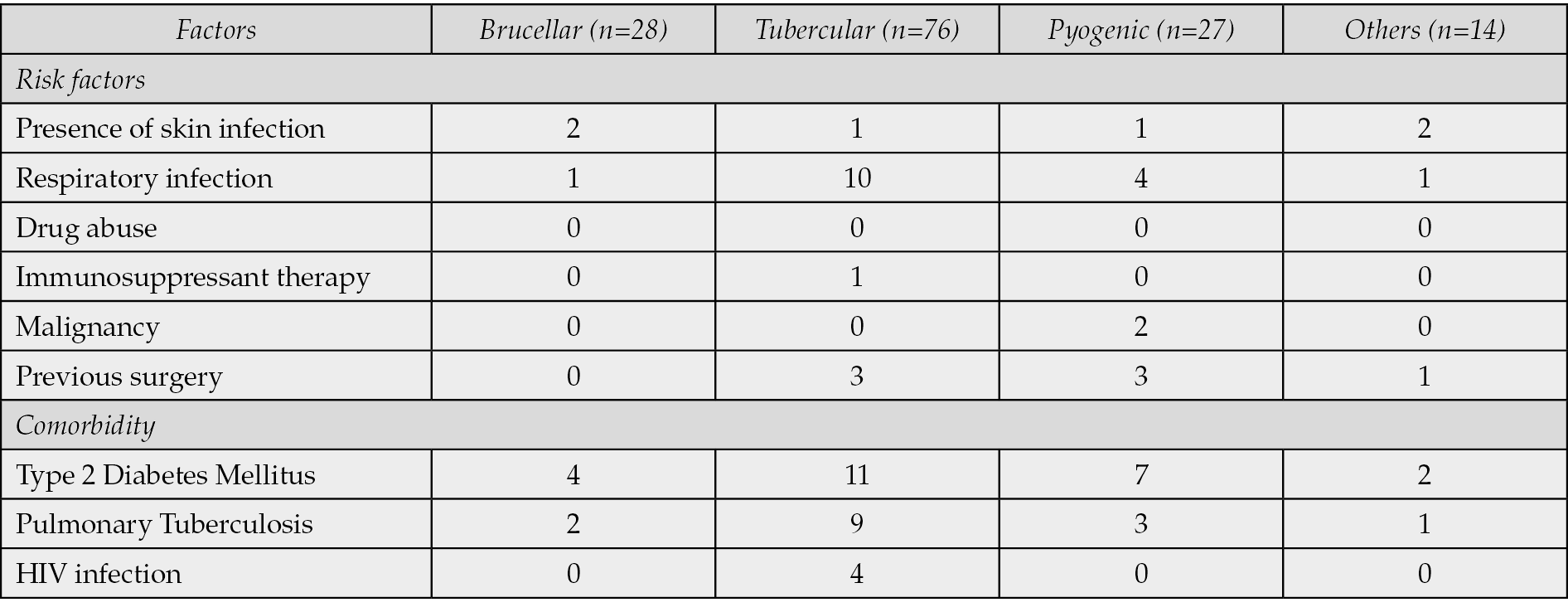
Among the identified risk factors, skin infections were minimally present across all spondylodiscitis types, while respiratory infections were more prevalent among tubercular spondylodiscitis patients (Table 2). Factors such as drug abuse, immunosuppressant therapy, and malignancy were also observed. Comorbid conditions such as Type 2 Diabetes Mellitus and Pulmonary Tuberculosis exhibited varying frequencies among different spondylodiscitis categories, highlighting the heterogeneous nature of these infectious types.
Table 2 - Presence of risk factors and comorbidities of patients diagnosed with spondylodiscitis.
Clinical presentation
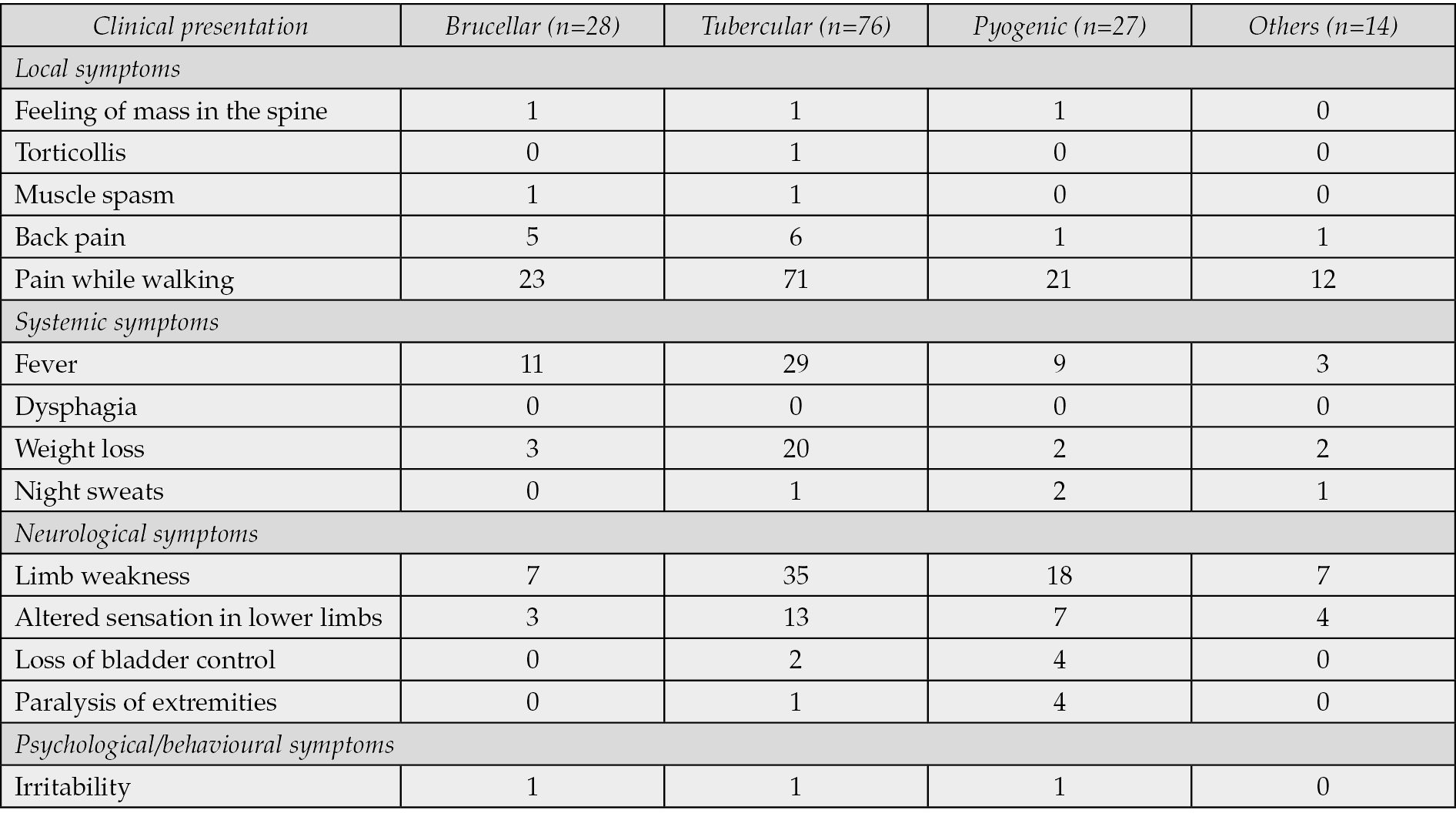
The clinical presentation of spondylodiscitis manifested differently across the infectious categories (Table 3). Common local symptoms, including back pain and pain while walking, were prevalent across all groups. Systemic symptoms such as fever and weight loss showed variations in occurrence, and neurological symptoms such as limb weakness and altered sensation of lower limbs displayed varying frequencies among the different infectious types.
Table 3 - The clinical presentations of patients with spondylodiscitis.
Laboratory parameters
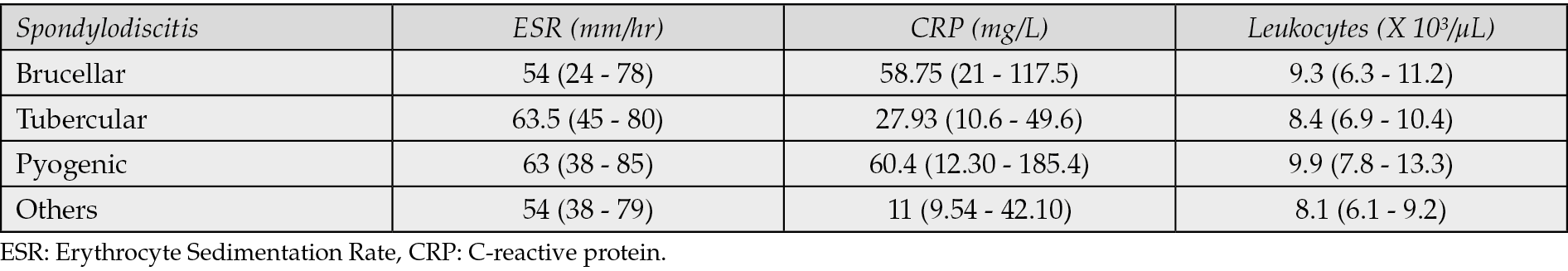
Laboratory findings revealed notable differences in erythrocyte sedimentation rate (ESR), C-reactive protein (CRP) levels, and leukocyte counts among different spondylodiscitis types (Table 4). Tubercular spondylodiscitis patients exhibited higher median ESR levels compared to other categories, while CRP levels varied significantly across the groups.
Table 4 - Median and IQR of Biochemical parameters of patinas with spondylodiscitis.
MRI scans
Magnetic Resonance Imaging (MRI) revealed significant abnormalities across various conditions. In the case of tubercular spondylodiscitis, changes were observed at the T12-L1 vertebrae (Figure 2A), with GenExpert® and histopathology confirming the presence of M. tuberculosis. The brucella antigen test returned positive for the patient with spondylodiscitis at L3-L4 (Figure 2B). In another patient with pyogenic spondylodiscitis (Figure 2C), a culture from the disc space tested positive for Staphylococcus aureus.
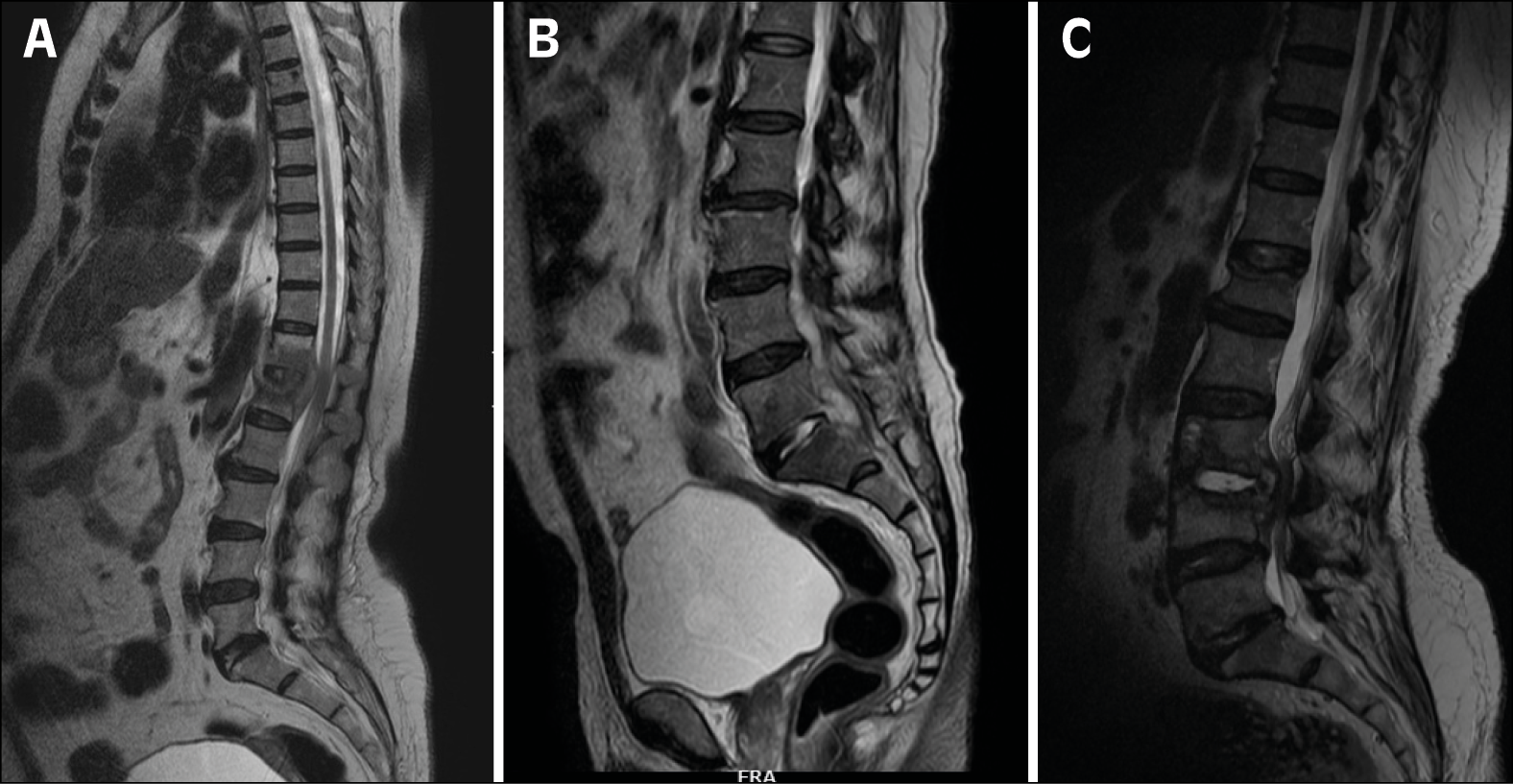
Figure 2 - Magnetic Resonance Imaging of different etilogies.
2A. Tubercular spondylodiscitis at T12-L1,
2B. L3-L4 spondylodiscitis.
2C Pyogenic spondylodiscitis.
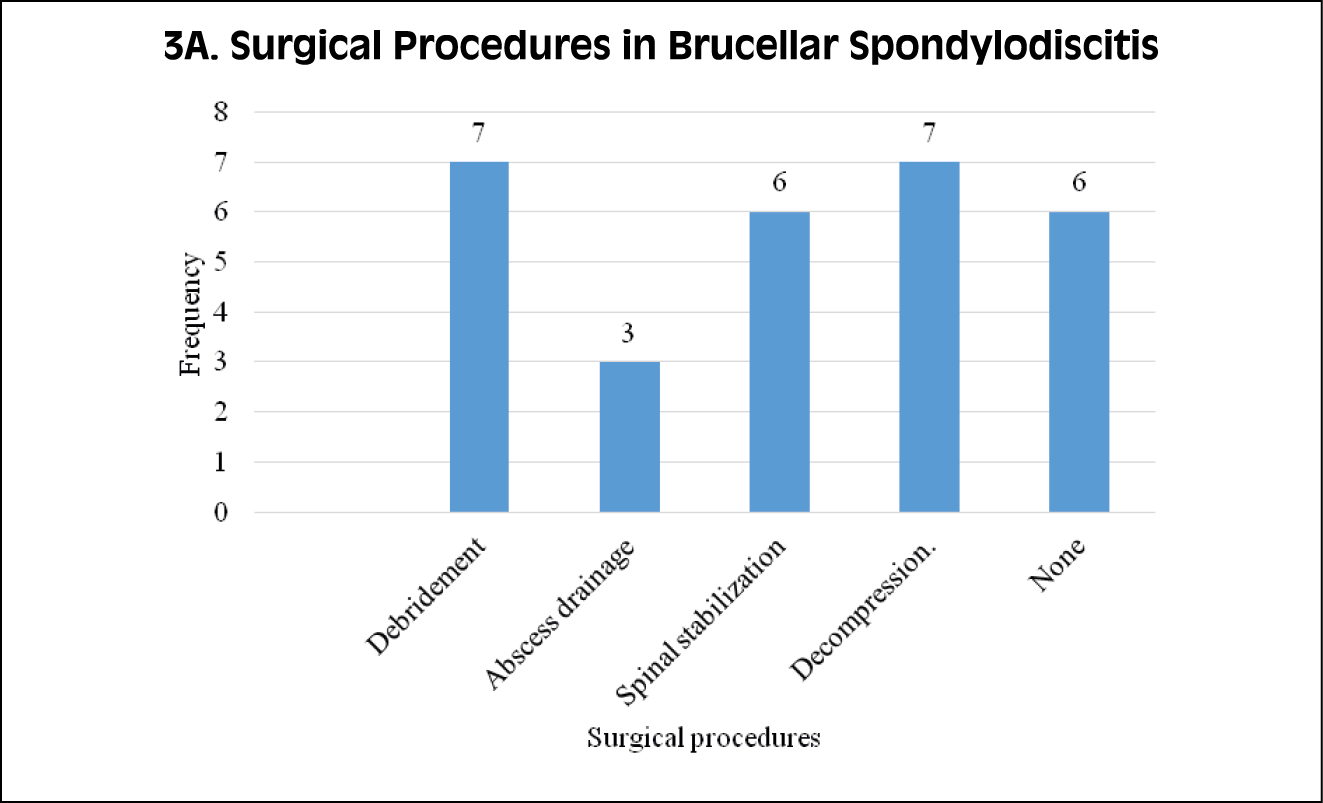
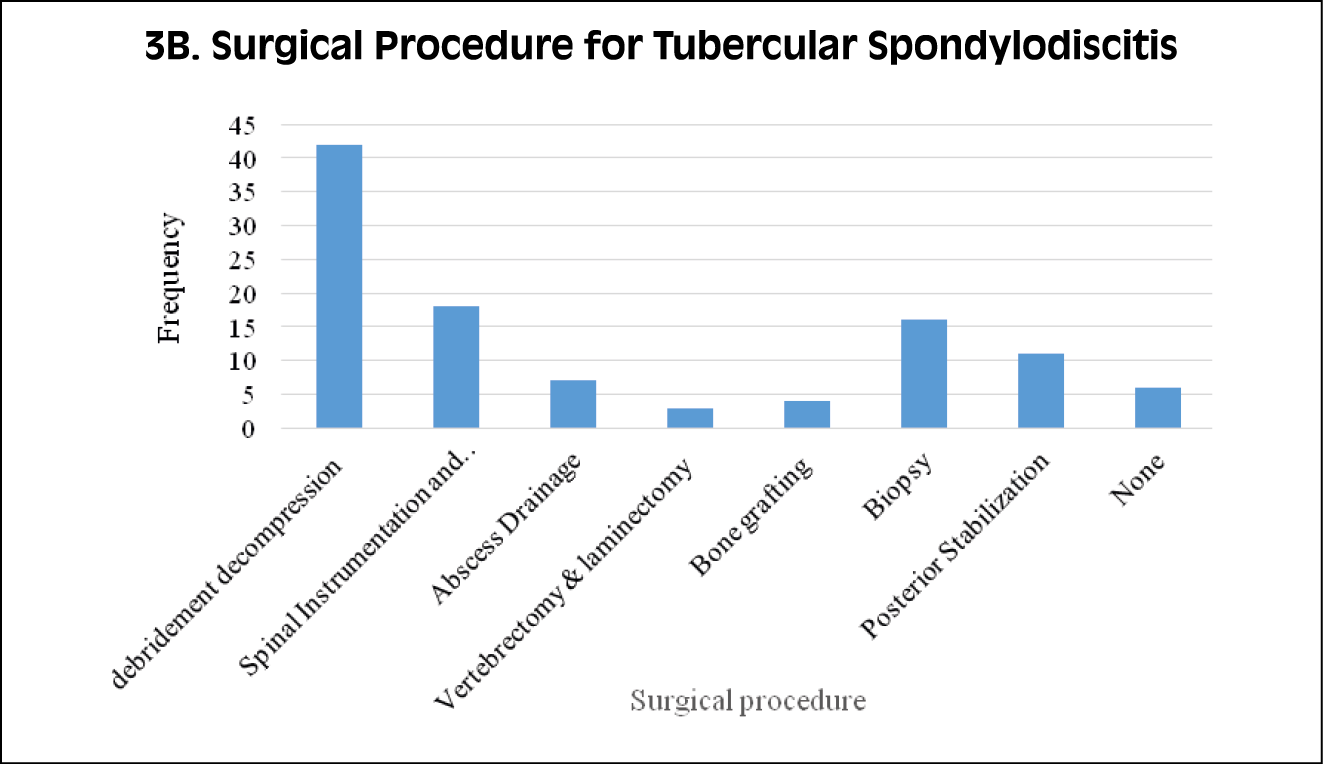
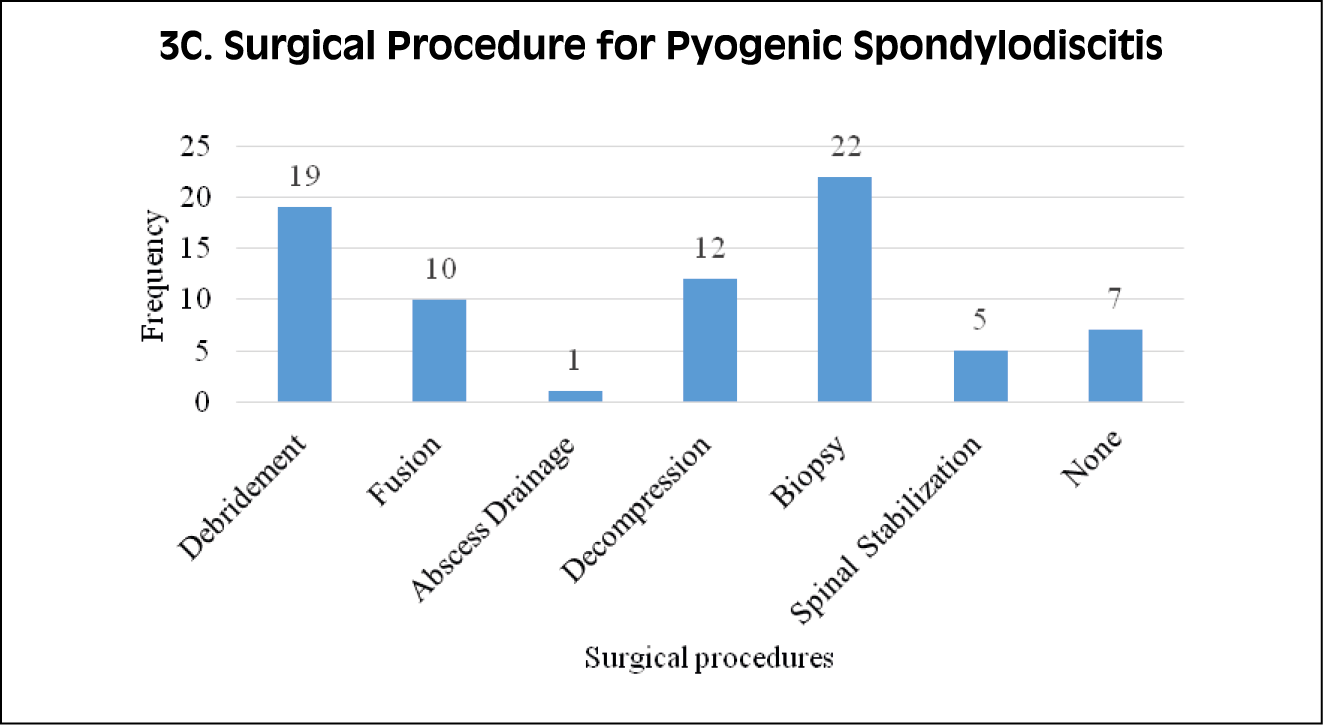
Figure 3A, 3B, 3C - Different surgical procedures performed for patients
with spondylodiscitis.
Treatment of spondylodiscitis
Patients with brucellar spondylodiscitis were treated with Rifampicin, Doxycycline and Gentamicin for two weeks. For the next 10 weeks, they received Rifampicin and Doxycycline. Tubercular spondylodiscitis cases were managed with a 2-month course of Ethambutol, Isoniazid, Rifampicin and Pyrazinamide, followed by 13 months of Ethambutol, Isoniazid, and Rifampicin. For pyogenic spondylodiscitis, treatment was tailored based on the aetiology and guided by culture sensitivity. In instances of non-specific spondylodiscitis, exhaustive efforts were made to identify the underlying cause. If no cause was identified despite thorough investigation, empirical management was provided.
Surgical treatment for spondylodiscitis
The primary surgical approach for both tubercular and brucellar spondylodiscitis commonly involved debridement and decompression procedures (Figure 3). In the case of pyogenic spondylodiscitis, the surgical interventions performed included biopsies, and six individuals required multiple surgeries.
DISCUSSION
Spondylodiscitis is a serious and debilitating condition that can cause significant morbidity and mortality if not diagnosed and treated promptly [2, 3]. The findings of this study reflect trends in the epidemiology of spondylodiscitis, aligning with global patterns reported in various studies [7-11]. This trend is particularly evident in countries like Tunisia, Mauritania, India, and South Africa, where the diagnosis and treatment of spinal tuberculosis are becoming increasingly important [24-27].
Tubercular spondylodiscitis has a higher incidence than other types due to several factors. It is more frequent in younger patients and often involves the thoracolumbar tract and multiple vertebrae [28].
The diverse demographic profiles across different types of spondylodiscitis highlight significant variations in age distribution and gender prevalence [10, 29]. Notably, brucellar spondylodiscitis affected a younger cohort compared to other infectious categories, a trend consistent with prior studies focusing on brucellosis [29, 30].
The findings from this study resonate with previous observations, highlighting unique symptoms that delineate each spondylodiscitis type. These distinct clinical manifestations include persistent back pain accompanied by fevers that often spike, spinal tenderness, and noticeable neurological issues [19, 20, 31]. Our research found that Brucellar spondylodiscitis typically affected the lumbar region, whereas tubercular spondylodiscitis predominantly impacted the thoracic area. Conversely, pyogenic spondylodiscitis commonly affected the lumbosacral region.
A consensus document by Lazzeri et al. (2019) provides a diagnostic flow chart for spondylodiscitis, emphasizing the role of nuclear medicine, radiology, and microbiology [21]. Blood cultures, CT-guided or surgical biopsy, and MRI are crucial for an etiological and radiological diagnosis [3, 32-34]. Homagk et al. developed a scoring system, SponDT, based on CRP, pain, and MRI to support diagnosis and treatment optimization [35]. The challenge of diagnosis is further highlighted by Pasku et al. (2021), who reported a rare case of spondylodiscitis caused by Fusobacterium nucleatum, emphasizing the importance of surgical biopsy and targeted antibiotics [36].
However, the shift towards pyogenic spondylodiscitis in some regions, as seen in India, underscores the need for accurate microbiological diagnosis through techniques like percutaneous biopsy [26]. Most of the patients included in our study have undergone MRI, and for a few of them, based on the clinical manifestations, blood/tissue culture with or without pathological tests were done.
The laboratory findings, particularly elevated ESR and CRP levels, underscore their significance as valuable indicators in diagnosing and monitoring spondylodiscitis [3, 37]. However, their specificity in distinguishing different infectious etiologies warrants further exploration [38].
Surgical intervention is required if there is neurological compromise resulting in progressive neurological deficits such as motor weakness, bowel or bladder dysfunction, or rapidly evolving radiculopathy to relieve pressure on the spinal cord or nerve roots. Additionally, in some instances, a tailored treatment approach combining prolonged antibiotic therapy and surgery is suggested for managing spondylodiscitis caused by non-tuberculous mycobacteria, highlighting the importance of comprehensive and individualized care in these complex infections [39].
Spondylodiscitis can lead to spinal instability, manifested by severe pain exacerbated by movement or deformity. Surgical stabilization may be required to restore spinal alignment and prevent further damage. Large epidural or paraspinal abscesses can compress neural structures, leading to neurological compromise or sepsis. Surgical drainage of abscess and debridement of infected tissue are crucial in preventing further spread of infection.
The primary goal of surgery is to achieve source control by debriding infected tissue and stabilizing the affected spinal segment. This often involves anterior or posterior approaches to the spine, depending on the location and extent of infection. Debridement of necrotic tissue, removal of infected intervertebral discs, and placement of bone grafts with or without implants for fusion are performed to eradicate the infection and restore spinal stability.
Limitations
The study exhibits several limitations. Firstly, its retrospective study design signifies a weaker research approach. Additionally, it lacks an exploration into the duration of the illness. Moreover, the sample size was not calculated, incorporating all available samples, which could compromise the study’s precision. Furthermore, inconsistencies arose in diagnostic measures among patients, predominantly due to financial constraints, leading to disparate evaluations. However, a notable strength lies in its contribution to the limited information available on spondylodiscitis in South India. The study’s three-year inclusion of patients adds valuable insights to this underexplored area.
CONCLUSIONS
The clinical-epidemiological analysis of spondylodiscitis provides valuable insights into the prevalence, risk factors, clinical characteristics, microbiological profile, and management strategies. Enhancing our understanding of the disease burden and optimizing diagnostic and therapeutic approaches can improve clinical outcomes and mitigate its impact on patients’ lives. Further prospective studies are warranted to validate these findings and guide future interventions to reduce the morbidity associated with this condition.
Authors’ contribution
Conceptualization and Methodology: LT, SNB; Data Collection: SH, LT; Analysis: SH, LT, SNB; Writing – Original Draft: LT; Writing – Review & Editing: LT, SH, SNB
Conflict of interest
Authors declare no conflict of interest.
Funding
None to declare.
REFERENCES
[1] Nickerson EK, Sinha R. Vertebral osteomyelitis in adults: an update. Br Med Bull. 2016; 117(1): 121-138.
[2] Tsantes AG, Papadopoulos DV, Vrioni G, et al. Spinal infections: an update. Microorganisms. 2020; 8(4): 476.
[3] Gupta N, Kadavigere R, Malla S, Bha SN, Saravu K. Differentiating tubercular from pyogenic causes of spine involvement on Magnetic Resonance Imaging. Infez Med. 2023; 31(1): 62-69.
[4] Feki A, Akrout R, Masmoudi K, et al. Infectious spondylodiscitis: A twenty-year experience from a single tertiary referral center. Egypt Rheumatol. 2019; 41(3): 231-235.
[5] Shanmuganathan R, Ramachandran K, Shetty AP, Kanna RM. Active tuberculosis of spine: current updates. N Am Spine Soc J. 2023; 16: 100267.
[6] Li W, Lai K, Chopra N, et al. Gut-disc axis: A cause of intervertebral disc degeneration and low back pain? Eur Spine J. 2022; 31(4): 917-925.
[7] Sur A, Tsang K, Brown M, Tzerakis N. Management of adult spontaneous spondylodiscitis and its rising incidence. Ann R Coll Surg Engl. 2015; 97(6): 451-455.
[8] Kehrer M, Pedersen C, Jensen TG, Lassen AT. Increasing incidence of pyogenic spondylodiscitis: a 14-year population-based study. J Infect. 2014; 68(4): 313-320.
[9] Herren C, Jung N, Pishnamaz M, et al. Spondylodiscitis: diagnosis and treatment options: a systematic review. Dtsch Arztebl Int. 2017; 114(51-52): 875.
[10] Lang S, Walter N, Schindler M, et al. The epidemiology of spondylodiscitis in Germany: a descriptive report of incidence rates, pathogens, in-hospital mortality, and hospital stays between 2010 and 2020. J Clin Med. 2023; 12(10): 3373.
[11] Fantoni M, Trecarichi EM, Rossi B, et al. Epidemiological and clinical features of pyogenic spondylodiscitis. Eur Rev Med Pharmacol Sci. 2012; 16: 2-7.
[12] Son HJ, Kim M, Kim DH, et al. Incidence and treatment trends of infectious spondylodiscitis in South Korea: A nationwide population-based study. PLoS One. 2023; 18(6): 1-11.
[13] Jose N, Ralph R, Mani T, Zachariah A. Infective spondylodiscitis-an Indian perspective. Indian J Appl Res. 2016; 6: 8.
[14] Durovic A, Eberhard N, Schären S, Widmer AF. Parvimonas micra as a rare cause of spondylodiscitis–case series from a single centre. Swiss Med Wkly. 2020; 150 (2526).
[15] Koutserimpas C, Chamakioti I, Naoum S, et al. Spondylodiscitis caused by Aspergillus species. Diagnostics (Basel). 2021; 11(10): 1899.
[16] Jain N, Pandya S, Srivastava P, et al. Is there any heterogeneity of spondyloarthritis disease manifestations between two ethnic subgroups?: A multicentre international comparative study between India and UK. Ann Rheum Dis. 2020; 79(Suppl 1): 1642-1643.
[17] Hughes G, Pollard C, Chavda S, David MD. The first case report of human infection with Phyllobacterium myrsinacearum causing spondylodiscitis. Clin Infect Pract. 2020; 7: 100029.
[18] Abbasi A, Jafarpour H, Shahmirzadi AR, et al. Frequency and characteristics of brucellosis in Golestan Province, Iran. Chron Dis J. 2020; 124-130.
[19] Ferri I, Ristori G, Lisi C, et al. Characteristics, management and outcomes of spondylodiscitis in children: a systematic review. Antibiotics (Basel). 2020; 10(1): 30.
[20] Gentile L, Benazzo F, De Rosa F, et al. A systematic review: characteristics, complications and treatment of spondylodiscitis. Eur Rev Med Pharmacol Sci. 2019; 23: 117-128.
[21] Lazzeri E, Bozzao A, Cataldo MA, et al. Joint EANM/ESNR and ESCMID-endorsed consensus document for the diagnosis of spine infection (spondylodiscitis) in adults. Eur J Nucl Med Mol Imaging. 2019; 46: 2464-2487.
[22] Gasbarrini A, Boriani L, Salvadori C, et al. Biopsy for suspected spondylodiscitis. Eur Rev Med Pharmacol Sci. 2012; 16: 26-34.
[23] Cardoso A, Barbosa L, Coelho AM, et al. Spondylodiscitis: a retrospective analysis of clinical, etiological, and radiological diagnosis. Int J Spine Surg. 2020; 14(2): 226-231.
[24] Saidane O, Sellami M, Cheikhrouhou S, et al. Clinical features and prognosis factors of spinal tuberculosis in Northern Tunisia: a case series of 60 patients. Bull Soc Pathol Exot. 2019; 112(2): 71-78.
[25] Boushab BM, Kone N, Basco LK. Contribution of computed tomography scan to the diagnosis of spinal tuberculosis in 14 cases in Assaba, Mauritania. Radiol Res Pract. 2019; 2019: 7298301.
[26] Aithala JP, Attar A, Imthiaz AK, Rai M. Is there a change in trend towards pyogenic spondylodiscitis compared to tubercular spondylodiscitis in India–a study of percutaneous biopsy evaluation in spondylodiscitis. Indian J Tuberc. 2020; 67(4): 509-514.
[27] Mann TN, Schaaf HS, Dunn RN, et al. Child and adult spinal tuberculosis at tertiary hospitals in the Western Cape, South Africa: 4-year burden and trend. Epidemiol Infect. 2018; 146(16): 2107-2115.
[28] Bassetti M, Merelli M, Di Gregorio F, et al. Higher fluorine-18 fluorodeoxyglucose positron emission tomography (FDG-PET) uptake in tuberculous compared to bacterial spondylodiscitis. Skeletal Radiol. 2017; 46: 777-783.
[29] Gupta N, Bhat SN, Reddysetti S, et al. Clinical profile, diagnosis, treatment, and outcome of patients with Tubercular versus non-tubercular causes of spine involvement: A retrospective cohort study from India. Int J Mycobacteriol. 2022; 11(1): 75-82.
[30] Ye HY, Xing FF, Yang J, et al. High index of suspicion for brucellosis in a highly cosmopolitan city in southern China. BMC Infect Dis. 2020; 20: 1-9.
[31] Kang HM, Choi EH, Lee HJ, et al. The etiology, clinical presentation and long-term outcome of spondylodiscitis in children. Pediatr Infect Dis J. 2016; 35(4): e102-e106.
[32] Kafle G, Garg B, Mehta N, et al. Diagnostic yield of image-guided biopsy in patients with suspected infectious spondylodiscitis: a prospective study from a tuberculosis-endemic country. Bone Joint J. 2022; 104(1): 120-126.
[33] Salaffi F, Ceccarelli L, Carotti M, et al. Differentiation between infectious spondylodiscitis versus inflammatory or degenerative spinal changes: How can magnetic resonance imaging help the clinician? La Radiol Med. 2021; 126(6): 843-859.
[34] Cheung WY, Luk KD. Pyogenic spondylitis. Int Orthop. 2012; 36: 397-404.
[35] Homagk L, Marmelstein D, Homagk N, Hofmann GO. SponDT (Spondylodiscitis Diagnosis and Treatment): spondylodiscitis scoring system. J Orthop Surg Res. 2019; 14: 1-8.
[36] Pasku D, Shah S, Aly A, Quraishi NA. Rare, post-periodontitis spondylodiscitis caused by Fusobacterium nucleatum in a patient with multiple sclerosis: challenge of diagnosis and treatment. BMJ Case Rep CP. 2021; 14(3): e239664.
[37] Ahn KS, Kang CH, Hong SJ, et al. The correlation between follow-up MRI findings and laboratory results in pyogenic spondylodiscitis. BMC Musculoskelet Disord. 2020; 21: 1-8.
[38] Bray C, Bell LN, Liang H, et al. Erythrocyte sedimentation rate and C-reactive protein measurements and their relevance in clinical medicine. WMJ. 2016; 115(6): 317-321.
[39] Smimmo A, Perna A, Fantoni M, et al. Non tuberculous mycobacteria related spondylodiscitis: a case report and systematic literature review. Infez Med. 2020; 28(3): 425-435.