Le Infezioni in Medicina, n. 4, 478-488, 2024
doi: 10.53854/liim-3204-6
REVIEW
RSV vaccination as the optimal prevention strategy for older adults
Massimo Andreoni1, Paolo Bonanni2, Giovanni Gabutti3, Stefania Maggi4, Roberta Siliquini5, Andrea Ungar6
1 Infectious Diseases Unit, University Tor Vergata, Rome, Italy;
2 Department of Health Sciences, University of Florence, Florence, Italy;
3 Coordinator Working Group “Vaccines and Immunization Policies”, Italian Scientific Society of Hygiene, Preventive Medicine and Public Health (SItI), Cogorno (Ge), Italy;
4 National Research Council (CNR), Aging Section, Padua, Italy;
5 Department of Public Health University of Torino, AOU Città della Salute e della Scienza, Torino, Italy;
6 Department of Geriatrics, University of Florence, Italy
Article received 17 October 2024 and accepted 12 November 2024
Corresponding author
Massimo Andreoni
E-mail: andreoni@uniroma2.it
SummaRY
Respiratory syncytial virus (RSV) is a respiratory virus that usually causes mild upper respiratory tract symptoms. However, it can lead to a severe lower respiratory tract disease in high-risk populations, with severe complications such as pneumonia and respiratory failure. RSV poses a significant public health threat not only to children, but also to adults, particularly those over 75 years of age and individuals with comorbidities. The high incidence and severity of RSV infections in these vulnerable groups highlights the urgent need for effective preventive strategies.
Despite advancements in the clinical management of respiratory infections, preventing RSV remains a significant challenge. The symptoms of RSV are often underestimated and misdiagnosed due to their similarity to other respiratory infections like influenza and COVID-19. This leads to a substantial disease burden not only for patients but also for healthcare systems, due to increased hospitalizations and prolonged treatments.
In recent years, there has been a significant progress in the development of RSV vaccines, offering new hope for disease prevention. Our aim is to promote RSV vaccination, emphasizing the importance of large-scale prevention efforts to enhance the quality of life for at-risk individuals and optimize healthcare resource allocation.
This paper will report efficacy and safety data for the three main vaccines currently available for healthcare practitioners, providing a comprehensive and balanced overview of vaccine options and supporting informed decision-making by healthcare professionals and regulatory authorities.
Keywords: RSV, vaccines, efficacy, safety, overview.
INTRODUCTION
Respiratory Syncytial Virus (RSV) is a widespread virus of the respiratory tract with which almost all children are infected by 2 years of age [1]. It is an RNA-based virus of the genus Orthopneumovirus, family Pneumoviridae, order Mononegavirales [2]. RSV spreads through respiratory droplets and contaminated surfaces, typically causing mild, cold-like symptoms, and its infection can be prevented by regular handwashing, avoiding contacts with sick people, and covering coughs. Usually, RSV infections resolve on their own without the need for medical intervention in healthy individuals. However, it can also become a major cause of respiratory diseases, especially in infants, older adults, and patients with chronic conditions or a weakened immune system [3-5]. In these high-risk populations, RSV can lead to complications like bronchiolitis and pneumonia. Common RSV illness symptoms include sore throat, sputum production, cough, fever, dyspnoea, myalgia and wheezing [6]. The diagnosis is complex due to the similarity of symptoms with other viral diseases like influenza and COVID-19, thus requiring a laboratory confirmation of the infection. The lack of specific antiviral treatments reduces the incentive for a high-cost molecular test, increasing the spreading risk and poor outcomes [7].
Taking a closer look at the molecular structure of the RSV, there are two structural proteins with antigenic determinants on the viral membrane that can elicit the production of neutralizing antibodies and are mostly investigated for the development of monoclonal antibodies or vaccines: the attachment protein G and the fusion protein F. RSV F is the preferred target since it plays a pivotal role in the host cell viral entry, and is highly conserved among RSV A and B subtypes. The RSV F protein can either exist in its pre-fusion (pre-F) or post-fusion (post-F) conformation, with the latter structure being irreversible once the fusion happens. This conformation rearrangement makes the protein lose its top antigenic sites, lowering the number of targets for immune-mediated neutralization [8]. Another mechanism of immune evasion used by RSV is mediated through the NS1 and NS2 non-structural proteins, that inhibit both the production and effector functions of type I interferon, thereby dampening the innate immune response. Moreover, once the cells are infected, a soluble form of the G protein is released, acting as a decoy and altering the immune cell response [9]. On top of that, the RSV RNA-dependent replication cycle lacks a proofreading mechanism and is thus error-prone, leading to mutations that allow for changes in virulence [10]. All these factors contribute in circumventing the efficacy of treatments and of the host immune response.
To prevent severe RSV complications in infants, the Food and Drugs Administration (FDA) and the European Medicines Agency (EMA) recently approved nirsevimab, a new monoclonal antibody targeting the pre-F RSV protein, quickly neutralizing the virus upon exposure [11]. In older adults and in those with chronic conditions, infection management currently involves only supportive care, despite the severe consequences.
Recent epidemiological studies in Europe indicate that RSV is one of the leading causes of acute respiratory tract infections, which could reach 5.7 million in the five largest European countries (Germany, Italy, France, the United Kingdom and Spain) [12, 13]. These infections lead to significant hospitalization and pose a considerable health burden. The incidence of RSV infection in hospitalized adults varies based on the age group. For adults aged 18 to 49 years, the rate ranges from 7.72 to 11.94 per 100,000 population, for the 50 to 64 years group the rate is 33.46-63.03 per 100,000. The highest incidence is observed in individuals older than 65, with rates ranging from 136.92 to 255.56 per 100,000, demonstrating that the incidence of severe respiratory complications is notably higher in older individuals compared to other age groups, with a significant impact on their health status [14, 15].
In this context, vaccination is the most appropriate strategy to prevent severe RSV outcomes and reduce healthcare costs, with enough data to support it as a key prevention strategy [16-20]. Recently, three new vaccines that provide promising evidence in lowering RSV-related complications have been approved. Our review aims to provide comprehensive information on the available vaccination options to support their effectiveness in preventing RSV and guiding informed decisions to save lives and healthcare resources.
PROTECTING VULNERABLE POPULATION
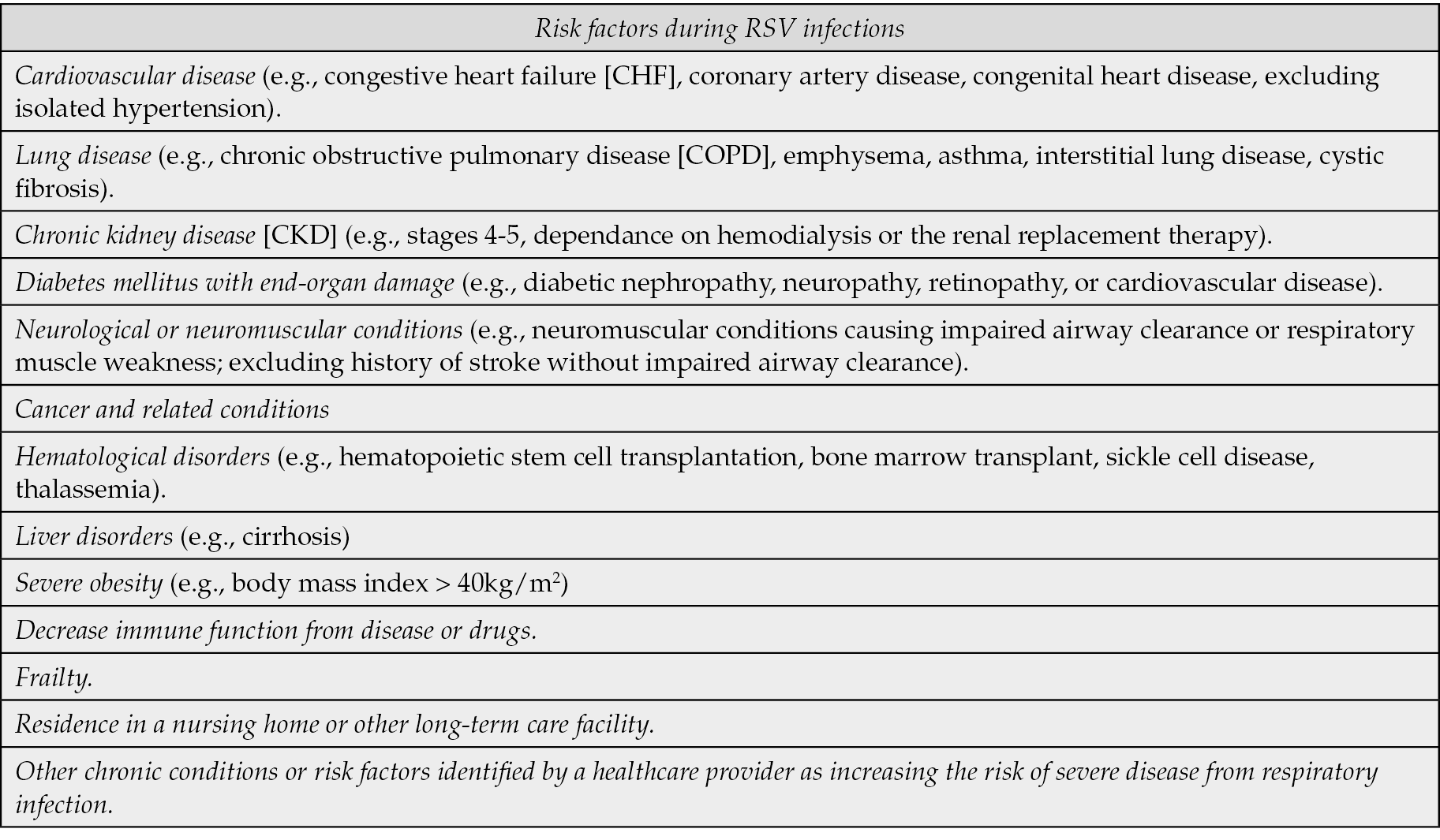
The global demographic shift towards an ageing population is creating significant challenges for healthcare systems worldwide. Older individuals are more susceptible to infections due to immunosenescence, the gradual deterioration of the immune system associated with aging. This renders the elderly population more vulnerable, impairing the ability to respond to pathogens. Patients with RSV infection are often older, making the virus a substantial burden in terms of morbidity and disease severity [21]. Additionally, pre-existing medical conditions, such as cardiovascular disease, lung and kidney disease, severe obesity, immunocompromising conditions, neurologic and neuromuscular conditions, liver disorders, hematologic conditions, cancer-related conditions, and frailty significantly increase the risk of poor outcomes when there is a concomitant RSV infection [22-24]. A comprehensive study involving US Medicare health insurance beneficiaries revealed alarming statistics: 53.7% of those hospitalized with RSV had chronic obstructive pulmonary disease (COPD), 41.1% had congestive heart failure (CHF), and 31.0% had chronic kidney disease (CKD). Hypertension, present in 65% of patients, and diabetes, in 28%, further exacerbate the risk and complexity of RSV management in these patients [15, 24]. A list of medical conditions that are risk factors for RSV can be found in Table 1. In this context, the increased risk is both due to the increased severity of the infection and the pre-existing condition, as well as the potential aggravation of pre-existing conditions due to treatment discontinuation, thus increasing the likelihood of complications. Additionally, individuals with compromised immune systems or advanced age face a higher risk of hospitalization, with prolonged hospital stays that increase healthcare costs [24-26]. Individuals residing in a nursing home or other long-term care facility are particularly vulnerable to RSV infection, which can lead to serious complications. These risks make preventive measures, such as vaccination, essential for safeguarding this at-risk population. Recently, the German Standing Committee in Vaccination STIKO issued its recommendation on RSV vaccination in adults [27], highlighting the significant impact caused by underreporting of the disease in this population. Specifically, the underreporting factor, estimated to range between 8 and 14 times, was determined by comparing the RSV hospitalization incidence from population-based studies and modeling to those recorded in hospital discharge diagnoses. This comparison indicated that the actual incidence of RSV-related hospitalizations could be significantly higher than what is reflected in routine hospital data. The discrepancy arises because hospital discharge records often rely on non-standardized RSV diagnostics, and RSV may not be consistently identified or recorded in older adults. Consequently, the true burden of RSV is likely underreported, and applying this correction factor helps provide a more accurate estimate of RSV-related hospitalizations [27].
Table 1 - Conditions that have been identified as risk factors for individual over 65 years old for the risk of poor outcomes during RSV infections. The list is inspired by ACIP’s general best practice guidelines for COVID-19 immunizations and is not exhaustive of all potential risk conditions [36].
RSV is rarely diagnosed in adults, despite its potential to be a serious condition. Raising awareness among healthcare professionals about the incidence of this disease is essential to enhance understanding RSV epidemiology and clinical manifestations. Enhanced knowledge will facilitate a more accurate assessment of the costs and benefits of vaccination. Planning targeted and effective interventions will ensure optimal resource utilization, reducing the burden of RSV and improving the quality of life for the most vulnerable populations.
DIVERSE VACCINE TECHNOLOGIES TO OPTIMIZE RSV PREVENTION STRATEGIES
Given the significant impact RSV has among elderly and vulnerable individuals, particularly due to infection-related complications, there is a significant interest in developing more effective strategies for preventing RSV infection and outcomes. RSV transmission is common to many other respiratory viruses, occurring through droplet inoculation in the eyes, nose, mouth, with close contact with an infected subject and with autoinoculation after touching infected surfaces, where the virus can be infective for up to 6 hours. When the virus reaches the mucosae, it infects airway epithelial cells in the upper respiratory tract, spreading afterwards towards bronchioles, where the replication is more effective [28]. Vaccination remains one of the most effective tools in preventing infectious diseases, including RSV. It is essential not only for protecting individuals from infection but also for reducing viral transmission within the community, thereby protecting the most vulnerable populations, such as the elderly, immunocompromised patients, and those with comorbidities. For individuals with a pre-existing medical condition, RSV can exacerbate symptoms, leading to severe complications like pneumonia and potentially causing long-term damage to lungs and heart. In these cases, vaccination is crucial to prevent the worsening of chronic diseases and to promote healthier lives.
Three main types of RSV vaccines are currently approved: two protein-based vaccines and an mRNA-based vaccine. Each of these offers unique advantages and potential limitations, representing different approaches in the fight against RSV. The major characteristics of the available RSV vaccines are listed in Table 2.
Table 2 - Major characteristics of three available RSV vaccines. LRTD: lower respiratory tract disease.

The two newly developed protein-based RSV vaccines are the recombinant, adjuvanted RSV pre-fusion F glycoprotein (RSVPreF3) introduced by GSK with the commercial name of Arexvy, and the bivalent RSV prefusion F protein-based (RSVPreF) vaccine containing F glycoproteins from RSV subtype A and subtype B, named Abrysvo, by Pfizer. These vaccines utilize protein subunits to elicit an immune response, providing targeted protection against the respiratory syncytial virus, and are both approved for the treatment of adults 60 years of age and older [29, 30]. Additionally, Abrysvo is also approved for use in pregnant women to protect newborns from birth until 6 months of age [29].
The messenger RNA (mRNA)-based RSV PreF vaccine developed by Moderna (mRNA-1345, mRESVIA) represents a significant advancement, being the first mRNA-based vaccine since COVID-19 to receive approval from the FDA and from EMA for the immunization of adults over 60 years of age [31, 32]. This vaccine contains an mRNA filament encoding the RSV F glycoprotein stabilized in its pre-fusion conformation. This technology, successfully tested with COVID-19 vaccines, offers advantages in terms of rapid development and production flexibility. The availability of these three vaccines provides a wide range of options for RSV prevention, fostering better implementation of such vaccination in target populations. Understanding the specific attributes and performances of each vaccine can guide healthcare practitioners in making informed decisions for their patients’ immunization plans.
EFFICACY OF RSV VACCINES
The efficacy of the approved RSV vaccines was demonstrated through rigorous clinical trials. However, the efficacy of each vaccine cannot be directly compared due to differences in study designs, populations, and methodologies, as well as the specific seasons when the trials were conducted [16-20].
GSK’s RSVPreF3 was evaluated in a phase III randomized and placebo-controlled clinical trial involving 24,966 adults aged 60 and older, with 39.3% of participants with at least one comorbidity. Participants received a single dose of RSVPreF3 or a placebo and were followed for a median of 6.7 months. The RSVPreF3 vaccine demonstrated an efficacy of 82.6% in preventing RT-PCR-confirmed RSV-associated lower respiratory tract disease (LRTD) and 94.1% against severe RSV-related LRTD over a median follow-up of 6.7 months (maximum 10.1 months). The vaccine’s efficacy was similar between RSV A and B subtypes [18]. Over the course of two seasons, with a median follow-up time of 17.8 months, the vaccine’s efficacy was 67.2% against RSV-associated LRTD and 78.8% against severe LRTD. The reactogenicity was comparable between participants that received one or two doses, indicating the efficacy of only one dose [20].
Pfizer’s bivalent vaccine was also studied in adults ages 60 years and older in a separate clinical trial. The study involved 34,284 participants in a phase 3, multicenter, double-blind, randomized, placebo-controlled trial. The result showed an efficacy of 66.7% in preventing RSV-associated LRTD with two signs or symptoms, 85.7% in preventing LRTD with three or more symptoms and 62.1% in preventing RSV-associated acute respiratory illness over the course of two RSV seasons [17]. RSVPreF was also studied in a randomized study including 7,392 pregnant women that received a single RSVpreF injection or placebo at 24 through 36 weeks gestation, showing varying efficacy in newborns protection. Severe LRTD was prevented in newborns, with a vaccine efficacy of 81.8% at 90 days after birth and of 69.4% at 180 days after birth, with no difference in the adverse events (AEs) occurrence between the vaccine and the placebo groups [16].
Moderna’s mRNA vaccine mRNA-1345 was tested in a randomized, double-blind, placebo-controlled phase 2-3 trial with 35,541 participants of 60 years of age or older. mRNA-1345 showed an overall efficacy of 83.7% in the whole cohort, with mostly mild to moderate transient AEs. The mRNA-based vaccine showed high efficacy in older populations, with an efficacy of 76% in those aged 60 to 69 years and 95.4% in those aged 70 to 79 years for RSV-associated LRTD with 2 signs or symptoms, and 76% in those aged 60 to 69 years and 100% in those aged 70-79 years for LRTD with 3 or more signs or symptoms, with a median follow-up of 112 days (range 1 to 379) [19]. Based on the most recent presented data, an additional analysis of efficacy was performed after a median follow-up of 8.6 months (range 15 to 530 days). A single dose of mRNA-1345 met the same criterion as defined in the primary analysis: the vaccine efficacy against RSV-LRTD with 2 or more symptoms was 63.3%, and 63.0% for RSV-LRTD with 3 or more symptoms. Furthermore, point estimates of vaccine efficacy in the subgroup analyses by age, comorbidity, and frailty were generally consistent with the vaccine efficacy observed in the overall population [33].
The availability of these three vaccines provides a wide range of options for RSV-related severe LRTS prevention, allowing vaccination strategies to be tailored to the diverse needs of the population.
SAFETY OF RSV VACCINES
Ensuring the safety of RSV vaccines is critical for public health. The safety profiles for the GSK, Pfizer and Moderna vaccines have been thoroughly evaluated.
GSK’s RSVPreF3 AReSVi-006 clinical study involving 24,966 adults over 60 years and with a median follow-up of 6.7 months (maximum 10.1 months) indicated that the most common AEs were pain at injection site (60.9%), fatigue (33.6%), myalgia (28.9%), headache (27.2%) and arthralgia (18.1%). Most solicited AEs were mild or moderate and resolved within 4 days. The unsolicited AEs occurring within 30 days after injection were comparable between the vaccine and the placebo groups, indicating that they were not linked to the vaccine administration. Three cases of fatal serious AEs (cardiopulmonary failure, pulmonary embolism and unknown cause of death) were considered to be related to the vaccine or placebo injection by the investigators, but after a blinded assessment an alternative explanation for these deaths was considered plausible on the basis of time to onset and the presence of preexisting risk factors [18].
Pfizer’s RSVPreF vaccine was assessed both in 34,284 subjects aged 60 years and over and in 7,392 pregnant women. In subjects aged 60 years and over, the most frequently reported local AE was pain at the vaccination site (11%) which was higher than in the placebo group (6%), whereas other reported systemic AEs (fatigue, headache, muscle and joint pain, nausea and diarrhea) were comparable between the two groups (27% vaccine vs 26% placebo). Most reactions were, however, mild to moderate in severity and resolved within 1-2 days after onset. AEs were considered during the follow-up over the course of two RSV seasons [17]. In the Capitalize study on pregnant women, the most frequently reported local AEs for women were pain at the injection site (41% vaccine vs 10% placebo), whereas other systemic AEs, both in pregnant women and in newborns at 1 month after birth were comparable between the two groups. AEs were assessed until 180 days after birth. The only AEs considered to be directly related to the RSVpreF vaccine were lymphadenopathy and injection-site bruising, reported in 0.1% of the vaccine recipients [16].
Moderna’s mRNA-1345, involving 35,541 participants over 60 years of age, showed more solicited AEs related to the vaccine respect to the placebo group (58.7% vs 16.2%) during the median follow-up of 112 days (range 1 to 379), but with a favorable safety profile with most AEs being mild or moderate and resolving within 1 or 2 days after onset, with symptoms like injection site pain, headache, myalgia and fatigue. The frequency of unsolicited AEs was balanced between the two groups within 28 days after injection (20.4% vaccine vs 18.8% placebo), suggesting no correlation with vaccine administration. No cases of acute myocarditis, pericarditis or severe neurological syndromes such as Guillain-Barré syndrome were reported in relation to the trial injection, and discontinuation due to AEs was balanced between the two groups (0.2% in both). None of the deaths that occurred during the study were reported by the investigators as related to the trial injection [19].
RECOMMENDATIONS FOR RSV VACCINE IMPLEMENTATION
To maximize the impact of available vaccines, it is essential to follow a series of recommendations for their effective implementation. These recommendations are based on considerations of efficacy, safety, and ease of access of the vaccines.
It is crucial that vaccination campaigns target people over 75 years of age and those over 60 with pre-existing medical conditions, regardless of the specific vaccine used [34]. The three available vaccines should be considered complementary and used flexibly to cover the target population [3].
Ensuring that vaccines are administered safely and efficiently is essential for the success of vaccination campaigns. The presentation in pre-filled syringes, as in the case of the Moderna vaccine, can facilitate administration and reduce errors [35].
Implementing a continuous monitoring system to evaluate the real-world efficacy and safety of vaccines is fundamental. This includes collecting post-marketing data to identify rare side effects and assess the duration of protection offered by vaccines. The effectiveness of RSV vaccines in real-world settings has been supported by observational studies. The VISION network study demonstrated that protein-based RSV vaccines have an effectiveness of 80% against RSV-associated hospitalizations among adults aged 60 years and older, studying 28,271 hospitalizations from October 2023 to March 2024 [36]. Similarly, the IVY network study reported an effectiveness of 75% against RSV-associated emergency department visits and hospitalizations in 367 adults over 60 years of age from October 2023 to March 2024 [37, 38]. These data suggest that RSV vaccination provide substantial protection in older adults with underlying health issues [16].
Currently, definitive conclusions on clear recommendations regarding the timing of revaccination against RSV cannot be drawn, especially for the differences from other respiratory illness, like influenza. RSV infection does not confer long-lasting immunity and allows for frequent reinfections. The immune response to RSV is less robust due to the virus’s ability to evade the immune system and inhibit a long-term protective response [39-41]. Clinical trial data regarding the protection of a single RSV vaccination dose are limited, and those available are heterogeneous and not exhaustive.
More specifically, GSK has published immunogenicity data on 24,967 subjects at 12 and 24 months, along with efficacy data at 12 months post-revaccination [20]. These data suggest that administering a second dose at 12 months does not offer additional protection compared to a single dose. On the contrary, Moderna has presented 12-month immunogenicity data on 300 subjects adults aged 65 to 79 years indicative of a booster effect, although these findings are not yet supported by efficacy evidence [42]. Lastly, Pfizer has not yet provided results concerning the revaccination of previously vaccinated individuals. Therefore, it is impossible to establish if a booster is effectively needed and what is the optimal revaccination timing for those who have already received a dose of the RSV vaccine. Currently, CDC recommends only a single dose of RSV vaccine for all adults over 75 years of age and for adults aged 60-74 with increased risk of severe RSV disease.
Continuous education and training for healthcare providers regarding the benefits of RSV vaccination and the specific characteristics of each vaccine are essential. Well-informed healthcare providers are crucial for promoting vaccination and addressing patients’ questions and concerns.
Scientific societies must collaborate closely with healthcare institutions to ensure that vaccination campaigns are well-planned and supported. This includes working with health ministries, regulatory agencies, and non-governmental organizations to facilitate vaccine access and implement large-scale awareness campaigns.
Many countries are still in the process of developing their RSV vaccination guidelines, as the vaccines are relatively new. The first RSV vaccine for adults was approved by the FDA only in May 2023 (GSK’s RSVPreF3) [43], and the most recent was in August 2024, when the EMA approved Moderna’s mRNA-based vaccine for immunizing adults over 60 years of age [32]. Guidelines for RSV vaccination vary across different countries, reflecting regional health priorities and demographic considerations [44]. In the United States, the Center for Disease Control and Prevention (CDC) updated its RSV vaccination recommendations for adults in 2024. The new recommendation advises that all individuals aged 75 and older should receive a single dose of any approved RSV vaccine. Additionally, adults aged 60 to 74 with chronic conditions or those living in nursing homes are also recommended to receive the vaccine. It would be also advisable to obtain immunization for the personnel working in nursing homes, in order to protect the elderly. CDC recommends that adults aged 60 or older who have already received RSV vaccination should not receive another dose, based on evidence that a single dose provides enough protection for at least two RSV seasons [45]. The UK has announced a national RSV vaccination program starting in September 2024, making it the first country to offer a national program using the same vaccine for both infants and older adults. The program includes vaccination for pregnant women over 28 weeks to protect their newborns, a routine vaccination program for those aged 75 and older, and a one-off campaign for people aged 75 to 79 [46]. In Germany, medical associations and the standing committee in vaccination STIKO are recommending RSV vaccination for people over 75 years of age and those over 60 with pre-existing conditions [27, 47]. Sweden recommended vaccination of people aged 75 and older, or 65 and older with pre-existing conditions [48]. The WHO recommendations include active immunization of infants and toddlers (aged 28 days to 23 months), active immunization of pregnant women and older adults (aged 50 and above), including those with co-morbidities [49].
These guidelines reflect a targeted approach to RSV vaccination, prioritizing those most at risk of severe illness, such as older adults, infants, and people with underlying health conditions.
CHALLENGES AND FUTURE PERSPECTIVES
Implementing effective vaccination programs faces several challenges and offers numerous opportunities for the future. One of the main challenges is vaccine acceptance. Vaccine hesitancy, often fuelled by misinformation and scepticism, can pose a significant barrier to widespread adoption. It is essential to develop effective communication strategies to educate not only the population about the benefits of RSV vaccination but also elderly care practitioners of the severity and incidence of RSV in their patients. In this context, providing detailed information about the demographics of the at-risk population and the effect of pre-existing medical conditions is crucial to ensure an optimal vaccination campaign. This information will help the discussion on how the findings could influence prevention strategies for the RSV virus.
Another critical issue is ensuring equitable access to vaccines, especially for vulnerable and disadvantaged populations. Economic, geographical, and logistical barriers must be addressed through inclusive public health policies and efficient distribution programs.
The production capacity and logistics of large-scale distribution represent significant operational challenges. The pharmaceutical industry and healthcare institutions must collaborate to ensure sufficient production and widespread distribution of vaccines. mRNA vaccines offer the advantages that their production is faster and easier to adapt to new virus variants than traditional vaccines, allowing a timely response to health emergencies.
Recent studies have explored the co-administration of RSV vaccines with other vaccines, such as the seasonal inactivated influenza vaccine (SIIV), showing promising results in terms of safety and immunogenicity. For instance, a study on RSVPreF co-administered with SIIV in adults aged 65 and older demonstrated a comparable immune response to the administration of each individual vaccine. This Phase III trial included 1403 participants, with a median age of 70, and found that local reactions and systemic events were mostly mild or moderate, with no vaccine-related serious adverse events reported [50]. The findings suggest that co-administering RSV vaccines with other seasonal vaccines, such as the influenza vaccine, is not only safe but also effective in eliciting a strong immune response. This co-administration strategy could enhance vaccine coverage rates by simplifying the vaccination process, making it more convenient for patients and healthcare providers. By reducing the need for multiple visits, this approach could improve compliance and overall vaccination rates, ultimately contributing to better public health outcomes and lower healthcare costs [51].
Despite these challenges, the future perspectives for RSV vaccination are promising. With continuous commitment to research, innovation, and collaboration, it is possible to significantly improve the prevention of respiratory infections and protect the health of the older population.
DISCUSSION
RSV is a significant burden for patients and for society in general, especially for older populations with comorbidities. Currently, there is no specific RSV antiviral treatment, highlighting the need for a more effective prevention strategy. Epidemiological data clearly indicate the importance of vaccination, especially for individuals older than 75 years old and those over 60 with pre-existing medical conditions [34].
Recently, three new vaccines against RSV have been approved: the protein-based vaccines RSVPreF3 and RSVPreF, and the mRNA-based vaccine mRNA-1345. They represent a crucial breakthrough in protecting the old population from severe respiratory tract infections. Current data confirm that the newly available options are not only effective but also safe, positioning vaccination as a key strategic tool for improving public health.
Recent years have seen not only particularly severe RSV infections in older populations, but also the “tripledemic” of COVID-19, influenza and RSV, combined with rising chronic diseases, exacerbating the pressure on healthcare systems [52]. For this reason, adult immunisation programs for diseases like RSV can offer a significant benefit for both public health and healthcare budgets, easing the pressure during the influenza season. RSV vaccines are considerably more expensive than typical influenza vaccines, with prices more comparable to newly developed COVID-19 vaccines. For this reason, their cost-effectiveness and accessibility are still being evaluated in many regions [53]. Investigating whether vaccines for influenza, COVID-19, and RSV can be administered together would be an excellent way to reduce costs and increase vaccination coverage. Combining these vaccinations into a single appointment could streamline the process for both healthcare providers and patients, increasing convenience and potentially boosting overall vaccine uptake.
Immunizing older individuals with pre-existing conditions shows a significant benefit-cost ratio. An economic analysis indicates that adult immunization programs generate significant economic returns by reducing hospitalizations. Vaccination can be seen as a highly beneficial investment strategy that can offer a return up to 19 times the initial cost for public health. In terms of age groups, the cost-effectiveness is particularly pronounced for individuals over 75 and those over 60 with pre-existing medical conditions. Vaccinating these populations can lead to substantial savings by preventing RSV-related hospitalizations [54].
A gradual implementation of the program is recommended to manage the high costs. Increasing coverage over several years can lead to significant long-term economic and health benefits with an investment that can be more acceptable and manageable. In Europe, applying similar strategies and focusing on high-risk groups can ensure the economic sustainability of RSV vaccination programs, leading to reduced healthcare costs and increased productivity.
An increase in the number of vaccinated people is only possible focusing both on population and healthcare professional education and effective communication to overcome vaccine hesitancy and misinformation. Global cooperation among pharmaceutical industries, governments, and healthcare institutions and the scientific community is essential to support large-scale production and distribution. Vaccines need to be of equitable access to ensure that older people can benefit from vaccination protection, despite their economical, geographical and logistic barrier. Integrating RSV vaccination with other vaccination programs, such as those against influenza, can increase vaccination coverage and improve prevention programs efficiency [55]. This synergistic approach can facilitate administration and reduce operational costs. Governments and policymakers are urged to prioritize adult immunisation as part of a broader public health strategy. Effective vaccination programs can support sustainable healthcare systems and promote long-term societal benefits.
CONCLUSIONS
Despite operational challenges, the future perspectives for RSV vaccination are promising, offering an unprecedented opportunity to improve the health of the elderly. Continuous commitment to promotion, research, and collaboration will ensure the success of this prevention strategy, protecting millions of people from the severe consequences of RSV infection.
Authors contribution
All authors contributed equally to the manuscript and should be considered as joint first authors.
Conflict of interest
All authors declare that they have no conflicts of interest.
Funding
This work was supported with unrestricted grant by Moderna.
Acknowledgements
Editorial assistance was provided by Osmosia medical writing and scientific consultancy and Aristea.
REFERENCES
[1] Obando-Pacheco P, Justicia-Grande AJ, Rivero-Calle I, et al. Respiratory Syncytial Virus Seasonality: A Global Overview. J Infect Dis. 2018; 217(9): 1356-1364.
[2] Griffiths C, Drews SJ, Marchant DJ. Respiratory Syncytial Virus: Infection, Detection, and New Options for Prevention and Treatment. Clin Microbiol Rev. 2017; 30(1): 277-319.
[3] Hernandez-Ruiz V, Letenneur L, Fülöp T, et al. Infectious diseases and cognition: do we have to worry? Neurol Sci. 2022; 43: 6215-6224.
[4] Shi T, Balsells E, Wastnedge E, Singleton R, et al. Risk factors for respiratory syncytial virus associated with acute lower respiratory infection in children under five years: Systematic review and meta-analysis. J Glob Health. 2015; 5: 020416.
[5] Robinson JL, Le Saux N. Preventing hospitalizations for respiratory syncytial virus infection. Paediatr Child Health. 2015; 20: 321-326.
[6] Belongia EA, King JP, Kieke BA, et al. Clinical Features, Severity, and Incidence of RSV Illness During 12 Consecutive Seasons in a Community Cohort of Adults ≥60 years old. Open Forum Infect Dis. 2018; 5(12): ofy316.
[7] Binder W, Thorsen J, Borczuk P. RSV in adult ED patients: Do emergency providers consider RSV as an admission diagnosis? Am J Emerg Med 2017; 35: 1162-1165.
[8] Graham BS. Vaccine development for respiratory syncytial virus. Curr Opin Virol. 2017; 23: 107-112.
[9] Mejias A, Rodríguez-Fernández R, Oliva S, Peeples ME, Ramilo O. The journey to a respiratory syncytial virus vaccine. Ann Allergy Asthma Immunol. 2020; 125(1): 36-46.
[10] Agoti CN, Otieno JR, Gitahi CW, Cane PA, Nokes DJ. Rapid Spread and Diversification of Respiratory Syncytial Virus Genotype ON1, Kenya. Emerg Infect Dis. 2014; 20(6): 950-959.
[11] Mankad VS, Leach A, Chang Y, et al. Comprehensive Summary of Safety Data on Nirsevimab in Infants and Children from All Pivotal Randomized Clinical Trials. Pathogens. 2024; 13: 503.
[12] Domnich A, Calabrò GE. Epidemiology and burden of respiratory syncytial virus in Italian adults: A systematic review and meta-analysis. PLoS One. 2024; 19: e0297608.
[13] Osei-Yeboah R, Spreeuwenberg P, Del Riccio M, et al. Estimation of the Number of Respiratory Syncytial Virus-Associated Hospitalizations in Adults in the European Union. J Infect Dis. 2023; 228: 1539-1548.
[14] Savic M, Penders Y, Shi T, Branche A, Pirçon J. Respiratory syncytial virus disease burden in adults aged 60 years and older in high-income countries: A systematic literature review and meta-analysis. Influenza Other Respir Viruses. 2022; 17(1): e13031.
[15] Branche AR, Saiman L, Walsh EE, et al. Incidence of Respiratory Syncytial Virus Infection Among Hospitalized Adults, 2017-2020. Clin Infect Dis 2022; 74: 1004-1011.
[16] Kampmann B, Madhi SA, Munjal I, et al. Bivalent Prefusion F Vaccine in Pregnancy to Prevent RSV Illness in Infants. N Engl J Med. 2023; 388(16): 1451-1464.
[17] Walsh EE, Pérez Marc G, Zareba AM, et al. Efficacy and Safety of a Bivalent RSV Prefusion F Vaccine in Older Adults. N Engl J Med. 2023; 388: 1465-1477.
[18] Papi A, Ison MG, Langley JM, et al. Respiratory Syncytial Virus Prefusion F Protein Vaccine in Older Adults. N Engl J Med. 2023; 388(7): 595-608.
[19] Wilson E, Goswami J, Baqui AH, et al. Efficacy and Safety of an mRNA-Based RSV PreF Vaccine in Older Adults. N Engl J Med. 2023; 389(24): 2233-2244.
[20] Ison MG, Papi A, Athan E, et al. Efficacy and Safety of Respiratory Syncytial Virus (RSV) Prefusion F Protein Vaccine (RSVPreF3 OA) in Older Adults Over 2 RSV Seasons. Clin Infect Dis. 2024; 78: 1732-44.
[21] Ackerson B, Tseng HF, Sy LS, et al. Severe Morbidity and Mortality Associated With Respiratory Syncytial Virus Versus Influenza Infection in Hospitalized Older Adults. Clin Infect Dis. 2019; 69: 197-203.
[22] Tseng HF, Sy LS, Ackerson B, et al. Severe Morbidity and Short- and Mid- to Long-term Mortality in Older Adults Hospitalized with Respiratory Syncytial Virus Infection. J Infect Dis. 2020; 222(8): 1298-1310.
[23] Versluys AB, Rossen JWA, van Ewijk B, Schuurman R, Bierings MB, Boelens JJ. Strong association between respiratory viral infection early after hematopoietic stem cell transplantation and the development of life-threatening acute and chronic alloimmune lung syndromes. Biol Blood Marrow Transplant. 2010; 16(6): 782-791.
[24] Wyffels V, Kariburyo F, Gavart S, Fleischhackl R, Yuce H. A Real-World Analysis of Patient Characteristics and Predictors of Hospitalization Among US Medicare Beneficiaries with Respiratory Syncytial Virus Infection. Adv Ther. 2020; 37(3): 1203-1217.
[25] Singh AM, Busse WW. Asthma exacerbations. 2: Aetiology. Thorax. 2006; 61(9): 809-816.
[26] Kwong JC, Schwartz KL, Campitelli MA, et al. Acute Myocardial Infarction after Laboratory-Confirmed Influenza Infection. N Engl J Med. 2018;378(4): 345-3453.
[27] RKI - Archiv 2024 - Epidemiologisches Bulletin 32/2024 n.d. https://www.rki.de/DE/Content/Infekt/EpidBull/Archiv/2024/Ausgaben/32_24.html (accessed September 6, 2024).
[28] Kaler J, Hussain A, Patel K, Hernandez T, Ray S. Respiratory Syncytial Virus: A Comprehensive Review of Transmission, Pathophysiology, and Manifestation. Cureus. 2023 Mar 18; 15(3): e36342.
[29] Abrysvo | European Medicines Agency (EMA) 2023. https://www.ema.europa.eu/en/medicines/human/EPAR/abrysvo (accessed October 2, 2024).
[30] Arexvy | European Medicines Agency (EMA) 2023. https://www.ema.europa.eu/en/medicines/human/EPAR/arexvy (accessed September 6, 2024).
[31] Healthcare G. mRESVIA’s FDA approval paves the way for mRNA vaccines across multiple indications. Clinical Trials Arena 2024. https://www.clinicaltrialsarena.com/analyst-comment/mresvias-fda-approval-mrna-vaccines/ (accessed August 27, 2024).
[32] Daily News 23 / 08 / 2024. European Commission - European Commission n.d. https://ec.europa.eu/commission/presscorner/detail/en/mex_24_4381 (accessed September 3, 2024).
[33] RSVVW’24 Conference - ReSViNET 2023. https://resvinet.org/conferences/rsvvw24/ (accessed September 9, 2024).
[34] Board del Calendario per la Vita. Raccomandazioni del Board del Calendario per la Vita sulla vaccinazione contro Virus Respiratorio Sinciziale (VRS o RSV) nella popolazione anziana e negli adulti a rischio. Gennaio 2024.
[35] Mehta D, Kimball-Carroll S, Clark DR, et al. Vaccine preparation time, errors, satisfaction, and preference of prefilled syringes versus RSV vaccines requiring reconstitution: randomized, time and motion study. J Med Econ 2024; 27(1): 887-896.
[36] Payne AB, Watts JA, Mitchell PK, et al. Respiratory syncytial virus (RSV) vaccine effectiveness against RSV-associated hospitalisations and emergency department encounters among adults aged 60 years and older in the USA, October, 2023, to March, 2024: a test-negative design analysis. Lancet. 2024; 404(10462): 1547-1559.
[37] Britton A, Roper LE, Kotton CN, et al. Use of Respiratory Syncytial Virus Vaccines in Adults Aged ≥60 Years: Updated Recommendations of the Advisory Committee on Immunization Practices - United States, 2024. MMWR Morb Mortal Wkly Rep 2024; 73(32): 696-702.
[38] Surie D. Effectiveness of adult respiratory syncytial virus (RSV) vaccines, 2023-2024. CDC (Presentation). Available at: https://www.cdc.gov/acip/downloads/slides-2024-06-26-28/07-RSV-Adult-Surie-508.pdf
[39] Gonzàlez-Parra G, De Ridder F, Huntjens D, Roymans D, Ispas G, Dobrovolny HM. A comparison of RSV and influenza in vitro kinetic parameters reveals differences in infecting time. PLoS One. 2018; 13(2): e0192645.
[40] Waterlow NR, Toizumi M, Leeuwen E van, et al. Evidence for influenza and RSV interaction from 10 years of enhanced surveillance in Nha Trang, Vietnam, a modelling study. PLoS Comput Biol. 2022; 18(6): e1010234.
[41] Kramer SC, Pirikahu S, Casalegno J-S, Cellès M. Characterizing the interactions between influenza and respiratory syncytial viruses and their implications for epidemic control 2022. https://doi.org/10.1101/2022.11.29.22282872.
[42] Shaw CA, Essink B, Harper C, et al. Safety and Immunogenicity of an mRNA-Based RSV Vaccine Including a 12-Month Booster in a Phase I Clinical Trial in Healthy Older Adults. J Infect Dis. 2024; 230(3): e647-e656.
[43] Venkatesan P. First RSV vaccine approvals. The Lancet Microbe 2023; 4(8): e577.
[44] RSV 2023. https://vaccination-info.europa.eu/en/rsv (accessed August 26, 2024).
[45] RSV (Respiratory Syncytial Virus) Immunizations | CDC 2024. https://www.cdc.gov/vaccines/vpd/rsv/index.html (accessed August 26, 2024).
[46] National RSV vaccination programme announced. GOVUK n.d. https://www.gov.uk/government/news/national-rsv-vaccination-programme-announced (accessed August 26, 2024).
[47] Protection against respiratory diseases: Medical associations recommend new RSV vaccination | German Center for Infection Research n.d. https://www.dzif.de/en/protection-against-respiratory-diseases-medical-associations-recommend-new-rsv-vaccination (accessed August 26, 2024).
[48] Vaccination mot RS-virus 2024. https://www.folkhalsomyndigheten.se/smittskydd-beredskap/vaccinationer/vacciner-som-anvands-i-sverige/vaccin-mot-rs-virus/ (accessed August 26, 2024).
[49] Guidelines on the quality, safety and efficacy of respiratory syncytial virus vaccines, Annex 2, TRS No 1024 n.d. https://www.who.int/publications/m/item/respiratory-syncytial-virus-vaccines-annex-2-trs-no-1024 (accessed August 26, 2024).
[50] Athan E, Baber J, Quan K, et al. Safety and Immunogenicity of Bivalent RSVpreF Vaccine Coadministered With Seasonal Inactivated Influenza Vaccine in Older Adults. Clin Infect Dis. 2024; 78(5): 1360-1368.
[51] Bonanni P, Steffen R, Schelling J, et al. Vaccine co-administration in adults: An effective way to improve vaccination coverage. Hum Vaccin Immunother 2023; 19(1): 2195786.
[52] Matias G, Taylor R, Haguinet F, Schuck-Paim C, Lustig R, Shinde V. Estimates of hospitalization attributable to influenza and RSV in the US during 1997-2009, by age and risk status. BMC Public Health. 2017; 17(1): 271.
[53] Crawford R, Bailey S, Cornelissen T. Cost-Effectiveness of Respiratory Syncytial Virus Vaccines for Adults. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; 2024 Feb. Report No.: HE0047.CADTH Health Technology Review.
[54] El Banhawi. Socio-Economic Value of Adult Immunisation Programmes - OHE. OHE - Leading Intellectual Authority on Global Health Economics 2024. https://www.ohe.org/publications/the-socio-economic-value-of-adult-immunisation-programmes/ (accessed August 26, 2024).
[55] Das R. Overview of Moderna’s Investigational RSV Vaccine (mRNA-1345) in Adults ≥60 Years of Age n.d.