Le Infezioni in Medicina, n. 4, 417-420, 2024
doi: 10.53854/liim-3204-1
EDITORIAL
Pregnancy Loss, Oropouche Virus and the Lessons from Pernambuco, Brazil
Shriyansh Srivastava1,2, Dheeraj Sharma1, Sachin Kumar2, Suraj N. Mali3, Rachana Mehta4,5, Vasso Apostolopoulos6, Ranjit Sah7-9, Tania do Socorro Souza Chaves10, Camila Luna11, Alfonso J. Rodriguez-Morales11,12,13
1 Department of Pharmacy, School of Medical and Allied Sciences, Galgotias University, Greater Noida 203201, India;
2 Department of Pharmacology, Delhi Pharmaceutical Sciences and Research University (DPSRU), Sector 3 Pushp Vihar, New Delhi 110017, India;
3 School of Pharmacy, D. Y. Patil University (Deemed to be University), Navi Mumbai, Maharashtra, India;
4 Dr. Lal PathLabs Nepal, Chandol, Kathmandu, Nepal;
5 Clinical Microbiology, RDC, Manav Rachna International Institute of Research and Studies, Faridabad, Haryana 121004, India;
6 School of Health and Biomedical Sciences, RMIT University, Bundoora Melbourne, VIC 3083, Australia;
7 SR Sanjeevani Hospital, Kalyanpur, Siraha 56517, Nepal;
8 Department of Microbiology, Dr. D. Y. Patil Medical College, Hospital and Research Centre, Dr. D. Y. Patil Vidyapeeth, Pune 411018, Maharashtra, India;
9 Department of Public Health Dentistry, Dr. D.Y. Patil Dental College and Hospital, Dr. D.Y. Patil Vidyapeeth, Pune 411018, Maharashtra, India;
10 Universidade Federal do Pará, Belém, Brazil;
11 Faculty of Health Sciences, Universidad Científica del Sur, Lima, Peru;
12 Gilbert and Rose-Marie Chagoury School of Medicine, Lebanese American University, Beirut P.O. Box 36, Lebanon;
13 Grupo de Investigación Biomedicina, Faculty of Medicine, Fundación Universitaria Autónoma de las Américas-Institución Universitaria Visión de las Américas, Pereira, Colombia
Article received 20 October 2024 and accepted 19 November 2024
Corresponding author
Alfonso J. Rodriguez-Morales
E-mail: arodriguezmo@cientifica.edu.pe
Arboviral infections have increasingly threatened public health throughout Latin America in recent years. The emergence of the Oropouche virus (OROV), a member of the Orthobunyavirus genus, is especially concerning. It was first isolated in Trinidad and Tobago in 1955 and endemic in Brazil, Panama, and Peru until 2000. In the last years, cases have been reported in several countries, including Bolivia, Brazil, Colombia, Cuba, Dominican Republic, Ecuador, Guyana, Panama, and Peru, and imported to European countries, Spain, Italy, and Germany; also to Canada and the United States. The primary vectorial transmission has been associated with Culicoides paraensis. The past few months have been especially troubling for Brazil, with an outbreak primarily affecting populations in Amazonas, Rondonia, and Pernambuco. The outbreaks have prompted research into the various clinical presentations linked to OROV, which encompass abrupt onset of fever, chills, headache, myalgia, and in rarer cases, exhibit neuroinvasive disease (meningitis, encephalitis), Guillain-Barre syndrome, and death. Sexual transmission is not yet known, but OROV was recently found in the semen of a returned traveller in Italy. Additionally, recent evidence suggests potential vertical transmission that could impact the fetus [1, 2]. The recent report on a series from Pernambuco highlights six cases of miscarriage among pregnant women infected with OROV [3]. These cases provide new evidence suggesting that OROV infection may lead to complications during pregnancy, specifically pregnancy loss [4]. As such, it is essential to draw attention to the implications of OROV in pregnancy and call for research into the maternal and neonatal consequences of OROV infection, focusing on enhanced public health surveillance and preventive measures in endemic regions.
As of epidemiological week 40 (EW40), 10,275 confirmed OROV cases have been reported across Bolivia, Brazil, Colombia, Cuba, Ecuador, Guyana, and Peru. Brazil has reported two fatalities, both young adults aged 21 and 24 (with rapid progression from symptoms to death), and significant complications in pregnancy [5]. In Pernambuco, Brazil, one fetal death of spontaneous abortion occurred at 30 weeks gestation, and four stillborns were recorded. OROV was confirmed in fetal tissues of the liver, kidneys, brain, spleen, heart, lungs, placenta and umbilical cord on October 9, 2024, raising concerns about OROV’s role in pregnancy complications. Although lab tests confirmed OROV in tissues from pregnancy loss, investigations continue to determine whether OROV directly caused fetal death. In addition to fetal death, one miscarriage of a pregnant female with OROV at eight weeks gestation was reported, and four newborns in Acre and Pará were born with microcephaly who were found to have IgM antibodies against OROV, in Pernambuco, potentially linked to vertical transmission [6]. As of EW40, cases under investigation include 12 cases of fetal death (Pernambuco=11, Ceará=1), 3 cases of congenital anomaly (Acre=2, Bahia=1), and three spontaneous miscarriages in Pernambuco [5]. A recent report of tested samples from 65 historical cases of congenital malformations from Brazil and 3 cases from 2024 confirmed OROV in all three cases in 2024 and 3 of the 65 historical cases [3]. Of these six positive cases, one died at 47 days (OROV positive by PCR), and one died in 2016 with no test samples available; from 5 available test samples, all 5 had IgM against OROV as well, and four maternal samples were IgM positive. While this data does not conclusively confirm vertical transmission or congenital malformations caused by OROV, it highlights the significant potential for fetal harm, warranting further investigation into these risks. Pernambuco has reported a further 156 OROV cases across municipalities, with investigations ongoing in Bom Jardim and Gravatá regarding fetal deaths [6, 7] (Figure 1).
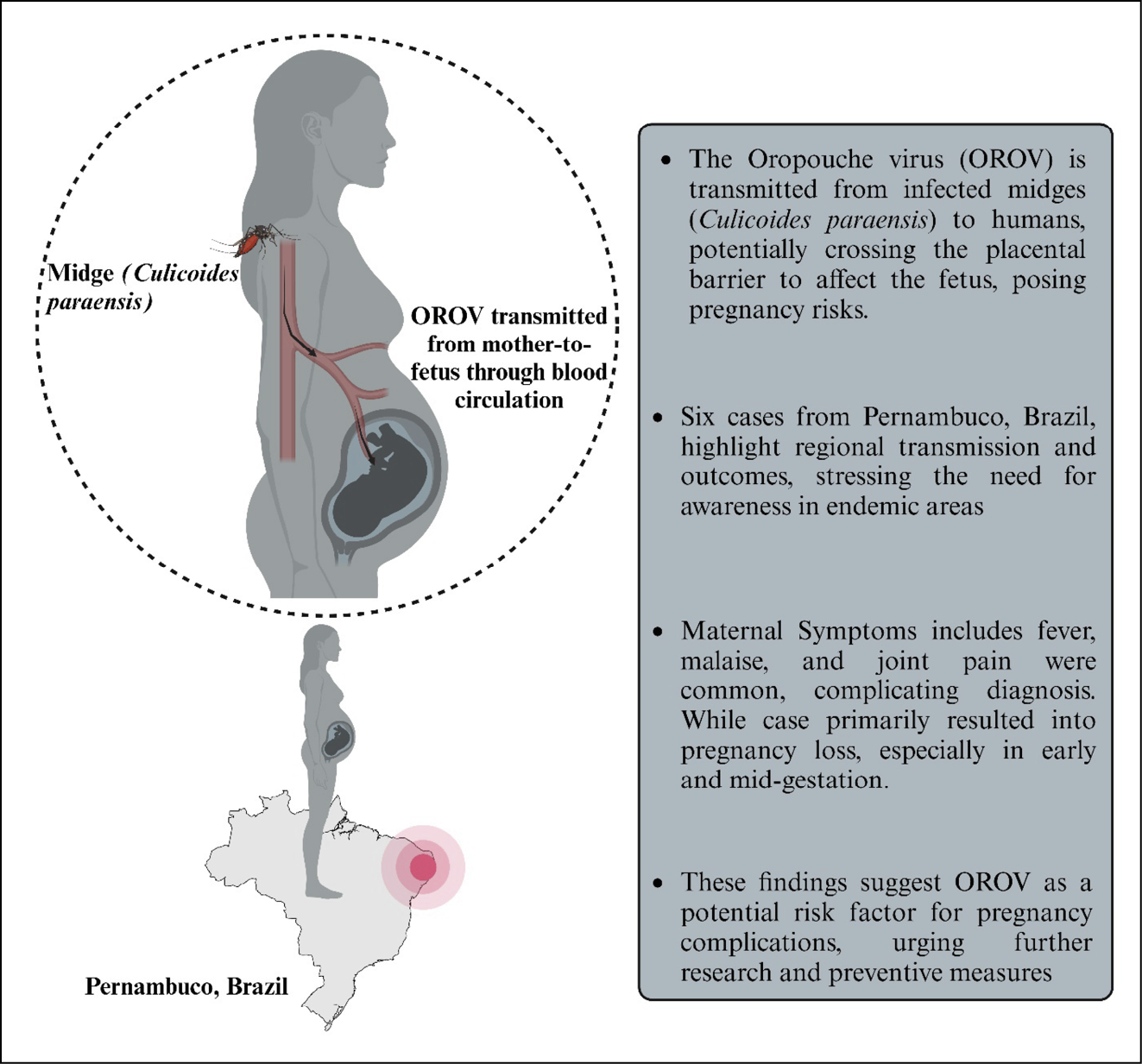
Figure 1 - Impact of Oropouche virus infection on pregnancy loss reported in Pernambuco, Brazil.
The burden among pregnant women is increased due to the difficulty in distinguishing symptoms between OROV and other co-endemic viruses, including Zika (ZIKV), dengue, and chikungunya. Therefore, there is a pressing need for a systematic clinical approach to managing exposed pregnant women to OROV risk. The first is advanced serological screening, early detection, and RT-PCR for confirmation. Such focused tests may help in differential diagnosis, especially in endemic regions with multiple arboviruses, facilitating early detection and possibly minimising adverse consequences of vertical transmission. Given OROV’s link to congenital anomalies, additional measures are recommended. Early prenatal imaging, including serial ultrasounds, can detect issues like microcephaly and growth restriction, providing more precise insight into OROV’s impact on fetal development from the first trimester [8]. Additionally, management of maternal-fetal infection should be strictly based on the established protocols for ZIKV that may involve structured counselling both for possible outcomes and coordination for neonatal care.
The Pernambuco cases raise critical gaps in our understanding of OROV and its impact on pregnancy, necessitating proactive public health approaches. In general, healthcare providers should especially consider OROV as part of the differential diagnosis of febrile illnesses among pregnant women in endemic areas. Because there are adverse outcomes such as miscarriage, fetal growth restriction, and possible neurological impact related to the virus, it is highly recommended that the clinical approach be highly structured. The patient should have regular ultrasounds, like that of ZIKV, where ultrasounds are usually conducted every 4-6 weeks to monitor fetal growth restriction, microcephaly or other developmental abnormalities [8, 9]. As there are no antiviral treatments available against OROV, symptom-alleviating medications are provided for symptoms such as fever, pain, and haemorrhagic phenomena while pregnant to manage associated complications and reduce inflammation. For high-risk fetal infection cases, especially after having an abnormal ultrasound, the test for OROV is done through amniocentesis in amniotic fluid, as commonly done in the case of ZIKV. If positive for OROV, pregnant women should consult neonatal units for potential complications. Newborns should undergo complete neurological and developmental assessments with ongoing follow-up for possible delays. Preventive counselling and education on using insect repellents, wearing protective clothing, using mosquito nets, and eliminating breeding sites are essential to reduce exposure and transmission risks, protecting both mother and fetus [8, 9].
The increasing number of documented pregnancy losses associated with OROV makes it mandatory to conduct intensive studies regarding the viral mechanism of maternal-fetal transmission, as well as its impacts on fetal development. Comparative studies in the mechanisms of infection pathways of the placenta and immune reactions might be enlightening between OROV and other viruses known to cause fetal issues, such as ZIKV. Longitudinal cohort studies that monitor the developmental profile of infants prenatally exposed to OROV could provide essential information regarding neurodevelopmental risks. These research efforts would focus on studying the neurotropism of OROV to identify potential neurological or developmental abnormalities in infants exposed to the virus during pregnancy, even in the absence of obvious congenital disabilities. This will establish key research milestones to understand better the dynamics involved in OROV and maternal-fetal transmission, leading to more effective clinical and public health responses [10].
The Pernambuco case series highlights the growing need for awareness and research on OROV’s maternal and fetal health risks. Evidence suggests possible vertical transmission and adverse pregnancy outcomes, but official guidance is still lacking [11]. A comprehensive approach should focus on improving maternal and neonatal health in OROV-affected areas with more robust surveillance, early diagnosis, and vector control. Further research is needed on multiple aspects of OROV, including transmission mechanisms, fetal impacts, and neurodevelopmental effects, which is crucial for developing targeted clinical and public health strategies to protect mothers and infants [12-14].
Conflict of interest
We declare no competing interests.
Funding
This study did not receive any funding.
Acknowledgments
This article has been registered in the Research Proposal Registration of the Coordination of Scientific Integrity and Surveillance of Universidad Cientifica del Sur, Lima, Peru, under the number PI-50-2024-0773.
REFERENCES
[1] Taylor L. Oropouche fever: Latin America on high alert for virus that can cause stillbirths. BMJ. 2024; 386: q1667.
[2] Sah R, Srivastava S, Kumar S, et al. Oropouche fever outbreak in Brazil: an emerging concern in Latin America. Lancet Microbe. 2024; 5(10): 100904.
[3] das Neves Martins FE, Chiang JO, Nunes BT, et al. Newborns with microcephaly in Brazil and potential vertical transmission of Oropouche virus: a case series. Lancet Infect Dis. 2024; S1473-3099(24)00617-0.
[4] Sah R, Srivastava S, Mehta R, et al. Oropouche fever fatalities and vertical transmission in South America: implications of a potential new mode of transmission. Lancet Reg Health Am. 2024; 38: 100896.
[5] PAHO. Epidemiological Update Oropouche in the Americas Region - 15 October 2024. Available at: https://www.paho.org/en/documents/epidemiological-update-oropouche-americas-region-15-october-2024 [accessed 6 November 2024].
[6] WHO. World Health Organisation. 23 August 2024. Oropouche virus disease - Region of the Americas. Available at: https://www.who.int/emergencies/disease-outbreak-news/item/2024-DON530. [accessed 4 November 2024].
[7] Castilletti C, Huits R, Mantovani RP, Accordini S, Alladio F, Gobbi F. Replication-Competent Oropouche Virus in Semen of Traveler Returning to Italy from Cuba, 2024. Emerg Infect Dis. 2024; 30(12).
[8] Schwartz DA, Dashraath P, Baud D. Oropouche Virus (OROV) in Pregnancy: An Emerging Cause of Placental and Fetal Infection Associated with Stillbirth and Microcephaly following Vertical Transmission. Viruses. 2024; 16(9): 1435.
[9] CDC. Interim Clinical Considerations for Pregnant People with Confirmed or Probable Oropouche Virus Disease. 04 November 2024. Available at: https://www.cdc.gov/oropouche/hcp/clinical-care/pregnancy.html [accessed 4 November 2024].
[10] Ginige S, Flower R, Viennet E. Neonatal outcomes from arboviruses in the perinatal period: a state-of-the-art review. Pediatrics. 2021; 147(4): e2020009720.
[11] Rodriguez-Morales AJ, Drexler JF. Re-emergence of Oropouche virus in Brazil and Latin America. Lancet Infect Dis. 2024; S1473-3099(24)00672-8.
[12] Escobedo AA, Rodríguez-Morales AJ. Oropouche fever rears its head in Cuba: What lies beneath the surface? New Microbes New Infect. 2024; 62: 101460.
[13] Al-Tawfiq JA, Rodriguez-Morales AJ, Johani SA. The emergence of Oropouche virus in Cuba - A wake-up call for global health. J Infect Public Health. 2024; 17(9): 102513.
[14] Culquichicón C, Cardona-Ospina JA, Patiño-Barbosa AM, Rodriguez-Morales AJ. Bibliometric analysis of Oropouche research: impact on the surveillance of emerging arboviruses in Latin America. F1000Res. 2017; 6: 194.