Le Infezioni in Medicina, n. 1, 103-112, 2024
doi: 10.53854/liim-3201-14
CASE REPORTS
Presumptive pulmonary toxocariasis in a patient affected by acute myeloid leukemia and Hodgkin lymphoma: case report and review of the literature in immunocompromised hosts
Tommaso Lupia1, Elena Crisà2, Valentina Sangiorgio3, Roberta Bosio4, Giacomo Stroffolini5, Elena Staffilano4, Vanesa Gregorc2, Silvia Corcione4,6, Francesco Giuseppe De Rosa1,4
1Unit of Infectious Diseases, Cardinal Massaia, Asti, Italy;
2Unit of Oncology and Haematology, Candiolo Cancer Institute, Candiolo, Italy;
3Pathological Anatomy Unit, Candiolo Cancer Institute, FPO-IRCCS, Candiolo, Italy;
4Department of Medical Sciences, Infectious Diseases, University of Turin, Turin, Italy;
5Department of Infectious-Tropical Diseases and Microbiology, IRCCS Sacro Cuore Don Calabria Hospital,
Negrar di Valpolicella, Verona, Italy;
6School of Medicine, Infectious Disease & Geographic Medicine, Tufts University, Boston, MA, USA.
Article received 10 October 2023, accepted 26 January 2024
Corresponding author
Tommaso Lupia
E-mail: tommaso.lupia89@gmail.com
SummaRY
Toxocariasis is a zoonosis transmitted by the nematode Toxocara spp. Immunocompromised hosts are more susceptible than general population to bacterial, viral, fungal and parasitic infections. In this population toxocariasis may present as exacerbation or reactivation and could have severe or atypical manifestations being a diagnostic challenge for healthcare providers. We report a case of a presumptive pulmonary toxocariasis during chemotherapy in a patient affected by acute myeloid leukaemia (AML) and Hodgkin lymphoma and we summarize current evidence of pulmonary involvement in immunocompromised population with Toxocara spp infection in a narrative review. The aim of this work is also to revise the current literature on pulmonary involvement during Toxocara spp infection in immunocompromised hosts to improve knowledge on clinical presentation, treatment and outcome. A 66 years old man who had undergone to a cytarabine and idarubicin chemotherapy induction scheme for AML, complained of febrile neutropenia and dry cought. At the chest computed tomography (CT) there were multiple nodular pulmonary lesions with subpleural consolidations. The lung biopsy revealed inflammatory infiltration with diffuse small granulomas with minor eosinophil component. The laboratory analysis showed high immunoglobulin E (IgE) count with normal peripherical eosinophils, among the extended parasitological analysis, Toxocara immunoblot assay resulted positive. In the most accepted hypothesis of a polmunary toxocariasis infection, the patient was treated with a combination of albendazole plus corticosteroids for four weeks, with a positive outcome. Infection complications during chemotherapy are not uncommon, however, this is the first reported case of pulmonary toxocariasis during cytarabine and idarubicin treatment in AML. The revised literature shows male gender and younger age as possible risk factors, nevertheless the majority of cases of seropositivity for Toxocara was reported in solid organ malignancies. In this case, the suspect was mainly based on laboratory total elevated IgE, confirmed by serological, anatomo-pathological and radiological findings. Hypereosinophilia is often not present in chronic infection. In conclusion, pulmonary toxocariasis should be ruled out in patients with pulmonary involvement and high IgE titre, with or without peripheral eosinophilia, especially in those with known immunocompromised status.
Keywords: Toxocara, pulmonary toxocariasis, haematological diseases, pneumonia, immunoglobulin E.
INTRODUCTION
Toxocara canis and Toxocara cati are the two main helminthic species that cause human toxocariasis in tropical and subtropical latitudes [1]. The global seroprevalence for this anthropozoonosis is around 19% and varies widely among countries and between rural or urban areas [2]. Infection occurs when a person accidentally ingests the eggs of Toxocara, usually through contaminated soil, water, or food. Once ingested, the eggs hatch in the intestine and the larvae can migrate to various organs and tissues in the body, including the liver, lungs, eyes, and central nervous system. The larvae do not develop into adult worms in humans, but their presence and movement can cause tissue damage and provoke an immune response. Therefore, humans are considered non-definitive hosts and the parasite larvae are unable to mature into adults and complete their life cycles after ingestion of viable eggs from a contaminated source (especially dog and cat faeces) [3-6]. Instead, the larvae of Toxocara spp. are capable of invading almost all organs in humans, especially the liver, lungs, spleen, brain and eyes [3-6].
Toxocariasis can present unique challenges in immunocompromised patients, as exacerbation or reactivation during chemotherapy in immunocompromised hosts has been reported rarely in the literature and most cases have occurred in non-haematological malignancies [7]. Generally speaking, individuals with weakened immune systems, such as those with HIV/AIDS, organ transplant recipients, or individuals undergoing chemotherapy, may be more susceptible to severe and disseminated forms of toxocariasis. Importantly, it has to be considered that immunocompromised patients are more susceptible to a range of infectious and parasitic diseases, and that host-immune-parasite interactions are unique [8, 9]. In particular, it has been reported that solid organ and bone marrow transplantations, blood transfusions and immuno-suppressive treatment are associated with a small but real risk of parasitic infections in Europe [10]. Immunocompromised individuals may include those with HIV/AIDS, organ transplant recipients, cancer patients undergoing chemotherapy, individuals on immunosuppressive medications, and those with congenital immune deficiencies. Parasitic infections that can pose specific significant risks in immunocompromised patients include Toxoplasmosis, Cryptosporidiosis, Strongyloidiasis, Leishmaniasis, Trypanosomiasis (i.e., African sleeping sickness and Chagas disease), and Paragonimiasis among others. Amidst these, Strongyloidiasis has been more extensively described and present peculiar features such as an auto-infective cycle [11, 12].
Broadly, global climate change and demographic changes are modifying the natural eco-system, and travel across continents have resulted in an increase in the transmission of parasites to human beings. Specifically, parasitic lung infections are being increasingly recognized and patients with immunodeficiency syndromes and acute or subacute chest symptoms should be evaluated for early detection of these pathogens [13].
As already pointed out, in immunocompromised individuals these infections can have severe and atypical manifestations, and clinicians may lack awareness in diagnosis. It is important for healthcare providers to be vigilant in assessing and managing parasitic infections in immunocompromised patients before any new therapy to be introduced, as the range of biological therapies is increasing, and a widespread use of newer immunosuppressive therapies, the growing population of individuals with immuno-compromised states as well as the prolonged survival of these patients have altered the pattern of parasitic infection [8, 9].
In immunocompromised patients, the larvae of Toxocara can migrate more extensively throughout the body, leading to widespread infection and potentially affecting multiple organs. Laboratory tests such as serological assays to detect specific antibodies may be helpful, although cross-reactivity with other nematodes (specifically Ancylostoma sp.) is possible. In some cases, a biopsy of affected tissues may be necessary to confirm the presence of larvae [3-6].
Special consideration should be made for immunocompromised returning travellers, or when immunocompromised are traveling to foreign regions where there are greater health risks than their home country [14, 15]. Moreover, transplant tourism, travel with the intent of receiving or donating a transplanted organ, has grown immensely in the past decade and appropriate epidemiological screening and diagnostic testing, may help to reduce the risk of transmission and disease of endemic pathogens [16]. An increase in donors who have emigrated from tropical areas and more transplant recipients traveling to endemic areas have led to a rise in parasitic infections reported among SOT recipients [17].
Here, we report the case of a presumptive pulmonary toxocariasis exacerbation during chemotherapy in a patient recently diagnosed with acute myeloid leukaemia (AML). Moreover, we have revised the literature describing cases of pulmonary toxocariasis in the immunocompromised population affected by onco-haematological diseases.
MATERIALS AND METHODS
The current narrative review followed the Scale for the Assessment of Narrative Review Articles (SANRA) flow-chart (Figure 1) [18].
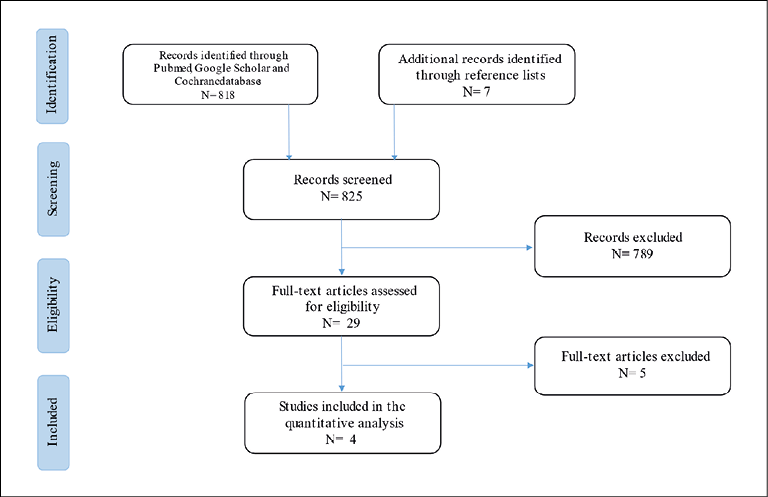
Figure 1 - Flow-chart of the studies revised in the narrative review.
The main aim of this work was to summarize current evidence on pulmonary involvement during Toxocara spp. infections in immunosuppressed adults patients to understand clinical characteristics, treatment, and outcome.
A search was run on Cochrane, PubMed, and Google Scholar using the terms (‘Toxoxara’ [Mesh]) AND (‘Lung’ [Mesh]) OR (‘Pulmonary’ [Mesh]) OR (‘Pneumonia’), (‘Toxocaras’ [Mesh]) AND (‘Immunocompromised’ [Mesh]), (‘Toxocara’ [Mesh]) AND (‘Malignancy’ [Mesh]), (‘Toxocara’ [Mesh]) AND (‘Tumor’ [Mesh]) and (‘Toxocara’ [Mesh]) AND (‘Haematological’ [Mesh]) in English. Results were limited to those published between 1 January 1990 and 1 July 2023. Studies were filtered for practice guidelines, guidelines, meta-analyses, systematic reviews, narrative reviews, case series, and case reports. Therefore we have filtered results including only humans and adults patients.
Our search strategy permitted the identification of 818 papers, of which 789 were excluded by title and abstract evaluation. Then, the reviewers studied titles and abstracts. Subsequently, 29 papers were included. Finally, quality assessment of full-text studies was performed by two independent reviewers (EC and TL). Researchers reviewed the summary of all articles sought and ultimately used data from full articles to compile this review paper. Researchers assessed the inclusion of all titles and abstracts without language limitations in English. We duplicated other studies previously included and excluded papers with no methods described, along with papers not strictly related to the aim of the study and according to journal importance and the number of references.
Full-text papers were then assessed for eligibility according to the above criteria, and the results are included in Table 1.
Table 1 - Manuscripts collected for the narrative review.

We performed descriptive statistics on the entire study population. Data were analyzed using standard statistical methods. Variables were described with medians, absolute values, and rates.
CASE REPORT
A 66-year-old male patient, living in an urban area with no history of recent travel, was admitted to hospital for a “7-3” cytarabine and idarubicin chemotherapy induction scheme for AML. His medical history included a bowel resection for acute appendicitis three months earlier, arterial hypertension, insulin-dependent diabetes mellitus, chronic pulmonary obstructive syndrome and concomitant classic Hodgkin lymphoma diagnosed three years prior. After chemotherapy induction, the patient presented with a long-lasting neutropenia of 18 days complicated by febrile neutropenia and a bloodstream infection (BSI) due to Stenotrophomonas maltophilia, which was treated effectively with intravenous cotrimoxazole and cefiderocol. Four weeks after the BSI, the patient developed a second episode of febrile neutropenia complicated by a new-onset dry cough. During this febrile episode, chest computed tomography (CT) was done, revealing multiple nodular pulmonary lesions and sub-pleural consolidations, without cavitation and characterized in part by surrounding ground-glass halos (Figure 2).

Figure 2 - Chest computed tomography revealing pulmonary involvement during the second episode of febrile neutropenia.
At the time of evaluation, the patient did not have fever and pulse oximetry was 96% while breathing room air. Laboratory examination revealed a white blood cell count of 3,950 cells/uL with a normal eosinophil count (300 cells/uL). Other results included haemoglobin (8.2 g/dL), platelets (12,000), lactate dehydrogenase (330 U/L) and C-reactive protein (47 g/dL) (Table 2).
Table 2 - Laboratory Features from diagnosis to clinical and radiological resolution.

Broncho-alveolar lavage was also performed; bacterial, viral, fungal and mycobacterial microbiological analyses were negative. After a pulmonary and radiological consult, a lung biopsy was carried out, which revealed diffuse inflammation infiltrated with small diffuse granulomas with a minor eosinophil component. In addition, bacterial, fungal and mycobacterial microscopy and cultures on biopsy samples were negative. Serological analysis showed a high immunoglobulin E (IgE) count (5,677 kUA/L; normal value <0.5) with a normal eosinophil count. After an infectious disease consultation, a Strongyloides spp., Trichinella spp., HTLV-1 serological analysis and an extended parasitological analysis of faeces on three consecutive samples were performed, with negative results. In addition, an immunoblot assay (Toxocara western blot IgG [LDBIO Diagnostics, Lyon, France]) was performed and had a strongly positive result. The biopsy was revised, but no larvae were seen in the sample. After an eye examination to exclude ocular involvement and due to the compatible CT findings, treatment with albendazole 400 mg every day for four weeks in combination with 50 mg of prednisone progressively de-escalated during the first week was started. The CT scan was repeated four weeks after the end of anti-helminthic treatment and revealed a complete regression of lung involvement (Figure 3).

Figure 3 - Chest computed tomography after four weeks from the end of albendazole treatment.
Interestingly, the total IgE count had a descent up to 3,740 kUA/L after anti-helminthic treatment. Because of the persistence of a detectable IgE count, an immunological consultation was made that excluded a Hyper IgE syndrome but revealed a concomitantly high anti-staphylococcal toxin A IgE count (1.84 kUA/L; normal value <0.1). We conclude that the patient had a multifactorial high IgE count due to haematological disease (i.e., Hodgkin lymphoma and AML), recent anthropozoonosis and the staphylococcal IgE component.
DISCUSSION
Pulmonary toxocariasis is an uncommon presentation of Toxocara spp. infestation in the haematological population and this is, to our knowledge, the first case reported in an AML/LH patient. Therefore we have reviewed the previously reported cases of pulmonary toxocariasis in immuncompromised patients presented in the literature (Table 3).
Table 3 - Cases collected from the narrative review of the literature performed.
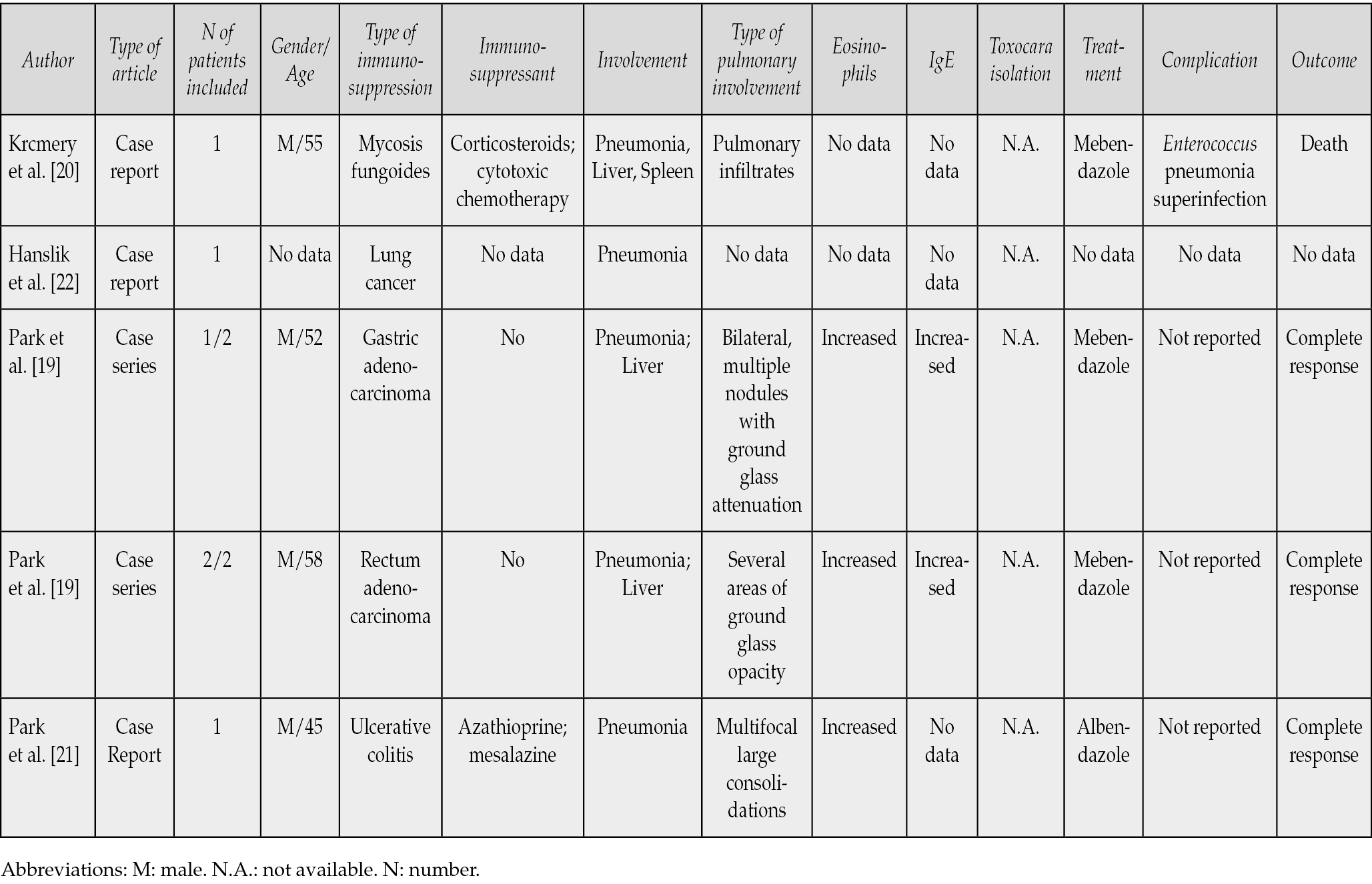
Most of the cases presented in this review occurred in males (Table 3) as in our patient. Raissi and colleagues reported an interesting sero-epidemiological study on human toxocariasis in individuals with blood disorders and cancer patients in Iran. Raissi and colleagues have founded an higher prevalence of seropositivity in female patients in all the groups (i.e., white cell disorders, blood cell disorders and cancer from all types), despite the lack of statistical significance between differences [7]. According to a review of studies on toxocariasis in the United States, Lee et al. discovered that 73% of the studies showed a higher prevalence in males compared to females, whereas 27% of the studies revealed no significant differences between genders [24]. The prevalence of Toxocara infection was not observed to be substantially greater in females compared to males in any study. Interestingly, Rostami and colleagues in a global meta-analysis have showed that being male (odds ratio [OR], 1.27; 95% CI, 1.17-1.39) represent a possible risk factors for seropositivity to Toxocara worldwide [2]. Corresponding to the larger collections of pulmonary toxocariasis cases reported by Ranasuriya et al., most of the patients (8/12) were males [25].
Moreover, Ranasuriya and colleagues in their review on pulmonary toxocariasis without mention of immunocompromising reported younger ages than reported in our review with a median of 38 vs 53 but with a wider range of ages (20 to 75 vs 45 to 58) of patients included [25]. Therefore, in the global meta-analysis of Rostami et al., younger age was reported among possible risk factors for positive serology of Toxocara, despite that patients aged between 41-60 vs ≥60 presented similar prevalence (17.4 vs 18.1) in their analysis [2].
Immunocompromised cohort presented in this review showed a single patient affected by haematological disorders (i.e., mycosis fungoides). Moreover, most of patients presented solid organ malignancies. Interestingly, Raissi and colleagues in their epidemiological analysis in immuncompromised hosts reported out of 101 individuals an higher seropositivity for Toxocara in cancer patients (11.06%) respect to white blood cell disorders (5.94%) [7].
Interestingly, we reported a case of pulmonary toxocariasis that occurred and was exacerbated after chemotherapy induction. Moreover, only two patients among five collected had undergone immunosuppressive regimens before diagnosis of pulmonary toxocariasis. At our knowledge no cases of toxocariasis were reported after or during cytarabine and idarubicin treatment for AML, on the other hand infections events are not uncommon during this regimen in AML patients with 186 events among 810 patients (22.9%) in randomized clinical trials according to Wang and colleagues meta-analysis [26]. Therefore, other helminthic infection reactivations during anti-neoplastic regimens, such as Strongyloides infections, were reported.
In our patients radiological suspected involvement by toxocariasis was uniquely pulmonary. Despite that three patients of five reported other organ involvement, notably hepatic. Moreover, we reported the CT pattern of pulmonary involvement with sub-pleural nodules and consolidations at diagnosis and after treatment (Figures 2 and 3), respectively. Lee et al. reported a more comprehensive collection of CT findings in pulmonary toxocariasis (N=63 patients) [27]. In this report, sub-pleural lesions were reported in 81% of cases. Moreover, solid nodules and patchy consolidations on CT were reported in 29% and 21% of cases, respectively [27]. These CT patterns were also reported in pneumonia involving of patients reviewed in this work (Table 3).
As a specific feature, migratory nodular shadows with halos are important chest computed tomographic findings in human toxocariasis, and can also be unexpectedly found in other parasitic disease in specific vulnerable population [28]. Projects exist to implement integrated multidisciplinary programs to prevent, diagnose and treat zoonotic or parasitic infections in the immunocompromised host [29]. Awareness is increasing regarding the different parasitic infections that may be evaluated prior to immune-modulatory therapies and guidelines have been produced accordingly [30, 31].
We therefore reported data regarding presence of hyper-eosinophilia and IgE. Contrary to our case with isolated hyper IgE most of patients collected reported both higher eosinophils and IgE. According to Park and colleagues, 20-30% of patients with toxocariasis do not have eosinophilia. In general, eosinophilia in helminth infection is more frequent and more pronounced in acute orearly infection and in the paediatric population [3-6, 32-34]. In fact, in chronic infection, chemotactic stimulation is reduced, and the migration of eosinophils presenting as peripheral blood eosinophilia is diminished [3-6, 32, 33]. For these reasons, the role of eosinophilia in subacute or chronic toxocariasis diagnosis may be less consistent [3-6, 32, 33]. Interestingly, in our patient the diagnosis was driven by the high total IgE count and confirmed by compatible serological, anatomo-pathological and radiological patterns paired with the exclusion of other microbial aetiologies. The serum IgE count correlates well with pulmonary infiltration and the level of toxocariasis activity, and this trend could be a useful surrogate to evaluate treatment efficacy [35].
In addition, Takamoto and colleagues [36] described the production of eosinophils and IgE in mutant mice deficient in CD4’ T cells and their normal and heterozygous littermates after T. canis infection. Eosinophil counts slowly decreased after a peak on day 10; in contrast, total IgE counts increased rapidly on days 7-14 and less rapidly on days 14-42 after T. canis infection [36]. Furthermore, Boldiš and colleagues reported that high IgE counts in human toxocariasis were more frequent in patients coming from urban areas, as was the case with our patient [32].
Histopathological findings of larvae in lung biopsies are rarely reported in humans and biopsies are performed in only a small percentage of suspected pulmonary toxocariasis cases [4, 5, 32]. Moreover, cases collected from the literature did not report histopathological isolation of Toxocara spp. as happened in our experience.
Albendazole treatment was continued for up to 4 weeks according to extent of lung involvement, immunological status of the patients and accordingly previous reported cases. In their literature review, Ranasuriya et al. reported a treatment duration for pulmonary toxocariasis between 5 and 56 days [26]. The treatment was well tolerated for the entire duration of therapy.
Treatment of toxocariasis in immunocompromised patients usually involves a combination of anthelmintic drugs, such as albendazole or mebendazole, and close monitoring for potential drug interactions with other medications. The treatment duration may be prolonged compared to immunocompetent individuals, and the response to therapy may vary based on the degree of immunosuppression [6]. Moreover, alongside anthelmintics, corticosteroids are currently are indicated for patients with severe symptoms. Corticosteroids are effective in decreasing inflammation and managing hypersensitivity responses induced by deteriorated larvae after nthelmintics therapy [37].
Pulmonary toxocariasis should be ruled out in patients with pulmonary involvement and high IgE counts with or without eosinophilia, and in patients from urban areas, especially those with known immunocompromised status.
This narrative review presents different limitations. First, this is a narrative revision of the literature and lacks a systematic methodology or meta-analysis of the data. Secondly, cases reported are uncommon in literature and the whole population is small respect to other more frequent pulmonary parasitic infections (i.e., Strongyloidiasis) in haematological population.
This review highlights the importance of continuing raising awareness and surveillance. In fact, increased awareness among healthcare professionals about pulmonary toxocariasis, particularly in immunocompromised populations, can lead to earlier diagnosis and appropriate management. Surveillance systems can help public health authorities monitor the prevalence and trends of this infection, enabling them to take timely preventive measures. In this perspective, we should not forget the zoonotic transmission nature of this infection. Therefore, public health efforts should focus on educating the public about the risks of exposure to contaminated soil and the importance of good hygiene practices, such as handwashing after contact with animals, to prevent infection. As clinicians we should promote integrated locally led multidisciplinary programs. This would enhance the prevention, diagnosis, and treatment of parasitic infections in immunocompromised hosts at the capillary level. These programs can also bring together healthcare providers, public health officials, and researchers to collaboratively address the challenges associated with these infections, providing an update body of literature for patient care. Particularly, at a global scale data we retrieved are suggesting possible risk factors for Toxocara seropositivity, including gender and age, and highlights the need for international collaboration in studying and addressing parasitic infections. Public health efforts should extend beyond national borders to tackle zoonotic infections effectively. By understanding and addressing the factors influencing pulmonary toxocariasis in immunocompromised individuals, healthcare professionals and public health authorities can improve patient outcomes, enhance surveillance and monitoring, and implement preventive measures to protect vulnerable populations. Possibly, enhanced diagnostic methods, both immune and parasite-based assays, coupled with imaging software, could overcome the difficulties in diagnosing and screening immune-compromised patients. Characterization and discussion of these challenges can be informed by data presented in the present work.
Author contributions
Each authors contributed equally to this work respect to study drafting and critical revision of the manuscript intellectual content.
Conflict of interest disclosure
All the authors declare no conflict of interest.
Financial support
No financial support for this work.
REFERENCES
[1] Chen J, Liu Q, Liu GH, et al. Toxocariasis: a silent threat with a progressive public health impact. Infect Dis Poverty. 2018; 7(1): 59.
[2] Rostami A, Riahi SM, Holland CV, et al. Seroprevalence estimates for toxocariasis in people worldwide: A systematic review and meta-analysis. PLoS Negl Trop Dis. 2019; 13(12): e0007809. https://doi.org/10.1371/journal.pntd.0007809.
[3] Wu TK, Bowman DD. Toxocara canis. Trends Parasitol. 2022; 38(8): 709-710. https://doi.org/10.1016/j.pt.2022.01.002.
[4] Moreira GM, De Lima Telmo P, Mendoça M et al. Human toxocariasis: current advances in diagnostics, treatment, and interventions. Trends Parasitol. 2014; 30(9): 456-564. https://doi.org/10.1016/j.pt.2014.07.003.
[5] Despommier D. Toxocariasis: clinical aspects, epidemiology, medical ecology, and molecular aspects. Clin Microbiol Rev. 2003; 16(2): 265-272. https://doi.org/10.1128/cmr.16.2.265-272.2003.
[6] Neumayr, Andreas. Antiparasitic Treatment Recommendations - A practical guide to clinical parasitology. 2018. ISBN: 978-3746950020.
[7] Raissi V, Sohrabi N, Bayat F, et al. Human Toxocariasis in individuals with blood disorders and cancer patients: the first seroepidemiological study in Iran. J Parasit Dis. 2021; 45(3): 643-650. https://doi.org/10.1007/s12639-021-01347-4.
[8] Evering T, Weiss LM. The immunology of parasite infections in immunocompromised hosts. Parasite Immunol. 2006; 28(11): 549-565. doi: 10.1111/j.1365-3024. 2006.00886.x.
[9] Fabiani S, Bruschi F. Rheumatological patients undergoing immunosuppressive treatments and parasitic diseases: a review of the literature of clinical cases and perspectives to screen and follow-up active and latent chronic infections. Clin Exp Rheumatol. 2014; 32(4): 587-585.
[10] Wołyniec W, Sulima M, Renke M, et al. Parasitic infections associated with unfavourable outcomes in transplant recipients. Medicina (Kaunas). 2018; 54(2): 27. doi: 10.3390/medicina54020027.
[11] Keiser PB, Nutman TB. Strongyloides stercoralis in the immunocompromised population. Clin Microbiol Rev. 2004; 17(1): 208-217. doi: 10.1128/CMR.17.1.208-217.2004.
[12] Lupia T, Crisà E, Gaviraghi A, et al. Overlapping Infection by Strongyloides spp. and Cytomegalovirus in the immunocompromised host: a comprehensive review of the literature. Trop Med Infect Dis. 2023; 8(7): 358. doi: 10.3390/tropicalmed8070358.
[13] Vijayan VK. Parasitic lung infections. Curr Opin Pulm Med. 2009; 15(3): 274-282. doi: 10.1097/MCP.0b013e328326f3f8.
[14] Rello J, Manuel O, Eggimann P, et al. Management of infections in critically ill returning travellers in the intensive care unit-II: clinical syndromes and special considerations in immunocompromised patients. Int J Infect Dis. 2016; 48: 104-112. doi: 10.1016/j.ijid.2016.04.020.
[15] Rosen J. Travel medicine and the solid-organ transplant recipient. Infect Dis Clin North Am. 2013; 27(2): 429-457. doi: 10.1016/j.idc.2013.02.009.
[16] Kotton CN. Transplant tourism and donor-derived parasitic infections. Transplant Proc. 2011; 43(6): 2448-2449. doi: 10.1016/j.transproceed.2011.06.022.
[17] Muñoz P, Valerio M, Puga D, et al. Parasitic infections in solid organ transplant recipients. Infect Dis Clin North Am. 2010; 24(2): 461-495. doi: 10.1016/j.idc.2010.01.009.
[18] Baethge C, Goldbeck-Wood S, Mertens S. SANRA - A scale for the quality assessment of narrative review articles. Res. Integr. Peer Rev. 2019; 4: 5. https://doi.org/10.1186/s41073-019-0064-8.
[19] Park BM, Jeong SO, Park HS, et al. Differences in the clinical and radiological characteristics of lung-involved toxocariasis between toxocariasis with eosinophilia and those without eosinophilia. J Thorac Dis, 2014; 6(12): 1757-1764. https://doi.org/10.3978/j.issn.2072-1439.2014.12.24.
[20] Krcméry V Jr, Gould I, Sobota K, et al. Two cases of disseminated toxocariasis in compromised hosts successfully treated with mebendazole. Chemotherapy. 1992; 38(5): 367-368. doi: 10.1159/000239027.
[21] Park S, Kim YS, Kim YJ, et al. Toxocariasis masquerading as liver and lung metastatic nodules in patents with gastrointestinal cancer: clinicopathologic study of five cases. Dig dis sci, 2012; 57(1): 155-160. https://doi.org/10.1007/s10620-011-1833-5.
[22] Hanslik T, Bougnoux ME, Kirch C,et al. Metastasis or visceral larva migrans? Ann Med Interne. 1998; 149(8): 533-535.
[23] Janczar S, Bulas M, Walenciak J, et al. Pulmonary exacerbation of undiagnosed Toxocariasis in intensively-treated high-risk neuroblastoma patients. Children (Basel). 2020; 7(10): 169. https://doi.org/10.3390/children7100169.
[24] Lee RM, Moore LB, Bottazzi ME, et al. Toxocariasis in North America: a systematic review. PLoS Negl Trop Dis. 2014; 8(8). https://doi.org/10.1371/journal.pntd.0003116.
[25] Ranasuriya G, Mian A, Boujaoude Z, et al. Pulmonary toxocariasis: a case report and literature review. Infection. 2014; 42(3): 575-578. https://doi.org/10.1007/s15010-014-0587-3.
[26] Wang H, Xiao X, Xiao Q, et al. The efficacy and safety of daunorubicin versus idarubicin combined with cytarabine for induction therapy in acute myeloid leukemia: A meta-analysis of randomized clinical trials. Medicine. 2020; 99(24): e20094. https://doi.org/10.1097/MD.0000000000020094.
[27] Lee KH, Kim TJ, Lee KW. Pulmonary Toxocariasis: initial and follow-up CT findings in 63 patients. AJR Am J roentgenol. 2015; 204(6): 1203-1211. https://doi.org/10.2214/AJR.14.13700.
[28] Gobbi F, Buonfrate D, Angheben A, et al. Pulmonary nodules in African migrants caused by chronic schistosomiasis. Lancet Infect Dis. 2017; 17(5): 159-165. doi: 10.1016/S1473-3099(16)30530-8.
[29] Peña A, Abarca K, Weitzel T, et al. One health in practice: a pilot project for integrated care of zoonotic infections in immunocompromised children and their pets in Chile. Zoonoses Public Health. 2016; 63(5): 403-409. doi: 10.1111/zph.12241.
[30] La Hoz RM, Morris MI. Infectious Diseases Community of Practice of the American Society of Transplantation. Tissue and blood protozoa including toxoplasmosis, Chagas disease, leishmaniasis, Babesia, Acanthamoeba, Balamuthia, and Naegleria in solid organ transplant recipients- Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant. 2019; 33(9). doi: 10.1111/ctr.13546.
[31] La Hoz RM, Morris MI, AST Infectious Diseases Community of Practice. Intestinal parasites including Cryptosporidium, Cyclospora, Giardia, and Microsporidia, Entamoeba histolytica, Strongyloides, Schistosomiasis, and Echinococcus: Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant. 2019; 33(9). doi: 10.1111/ctr.13618.
[32] Boldiš V, Ondriska F, Špitalská E, et al. Immunodiagnostic approaches for the detection of human toxocarosis. Exp Parasitol. 2015; 159: 252-258. https://doi.org/10.1016/j.exppara.2015.10.006.
[33] Ramirez GA, Yacoub MR, Ripa M, et al. Eosinophils from Physiology to Disease: A Comprehensive Review. Biomed Res Int. 2018. https://doi.org/10.1155/2018/9095275.
[34] Mazur-Melewska K, Jończyk-Potoczna K, Kemnitz P, et al. Pulmonary presentation of Toxocara sp. infection in children. Pneumonol Alergol Pol. 2015; 83(4): 250-255. https://doi.org/10.5603/PiAP.a2015.0043.
[35] Obwaller A, Jensen-Jarolim E, Auer H, et al. Toxocara infestations in humans: symptomatic course of toxocarosis correlates significantly with levels of IgE/anti-IgE immune complexes. Parasite Immunol. 1998; 20(7): 311-317. https://doi.org/10.1046/j.1365-3024. 1998.00146.x.
[36] Takamoto M, Sugane K. Mechanisms of eosinophilia in Toxocara canis infected mice: in vitro production of interleukin 5 by lung cells of both normal and congenitally athymic nude mice. Parasite Immunol. 1993; 15(9): 493-500. https://doi.org/10.1111/j.1365-3024.1993.tb00636.x.
[37] Chen J, Liu Q, Liu GH et al. Toxocariasis: a silent threat with a progressive public health impact. Infect Dis Poverty. 2018; 7(1): 59. https://doi.org/10.1186/s40249-018-0437-0.