Le Infezioni in Medicina, n. 4, 440-448, 2023
doi: 10.53854/liim-3104-3
REVIEWS
Outcomes of COVID-19 amongst patients with ongoing use of inhaled corticosteroids - a systematic review & meta-analysis
Syed Nazeer Mahmood1, Viraj Shah2, Urvish Patel3, Muhammad Umair Nawaz4, Narayana Varalakshmi Akula5, Irina Balan6, Divya Manivannan7, Yelena Pleshkova8, Shayaan Negit9, Prarthana Desai10, Richa Jaiswal11, Namratha Gurram12, Neel Patel13, Raghvendra Tirupathi14, Thoyaja Koritala15
1Department of Medicine, Section of Pulmonary/Critical Care, MedStar Washington Hospital Center,Washington, DC, USA;
2Department of Internal Medicine, Hackensack Meridian Health Ocean University Medical Center, NJ, USA;
3Department of Neurology and Public Health, Icahn School of Medicine at Mount Sinai, New York, NY, USA; 4Department of Internal Medicine, Griffin Hospital, Derby, CT, USA;
5Department of Internal Medicine, Carle Foundation Hospital, Champaign, IL, USA;
6Department of Family Medicine, Harnett Health, Campbell University, North Carolina, USA;
7Sri Ramachandra Institute of Higher Education and Research, India;
8Department of Internal Medicine, Adventhealth, Sebring, FL, USA;
9American University of the Caribbean, School of Medicine, Sint Marteen;
10Department of Internal Medicine, Danbury Hospital, Danbury, CT, USA;
11Department of Internal Medicine, Medical University of South Carolina, SC, USA;
12State University of New York, University at Albany, NY, USA;
13Department of Public Health, Icahn School of Medicine at Mount Sinai, New York, NY, USA;
14Department of Internal Medicine, Keystone Healwth, Chambersburg, PA, USA;
15Department of Internal Medicine, Mayo Clinic Health System, Mankato, MN, USA
Article received 17 August 2023; accepted 2 November 2023
Corresponding author
Urvish Patel
E-mail: dr.urvish.patel@gmail.com
SummaRY
Background: WHO quoted the numbers for the Coronavirus disease 2019 (COVID-19) pandemic as of August 2021 were 200 million cases with over 4 million deaths globally. COVID-19 is associated with several respiratory pathologies. Inhaled corticosteroids (ICS) are used to improve lung function by reducing inflammation, edema, mucus secretion, and inhibiting various cytokine activities. However, there is limited data on the effect of ICS usage in patients with COVID-19. In this study, we aim to evaluate the association between the use of ICS and the outcomes in COVID-19 patients compared to standard COVID-19 treatment.
Methods: We followed PRISMA guidelines and MOOSE protocol for conducting the systematic review and meta-analysis comparing ICS and standard COVID-19 therapy. A search on PubMed is conducted yielding 270 articles of which 6 manuscripts are finalized for inclusion in the study. Patients with COVID-19 are identified from the studies based on confirmed positive RT-PCR tests. Hospitalization, ICU admission, and mortality are selected as the outcomes of our study. Using RevMan 5.3, we performed random-effects models to estimate the pooled effect size (pooled odds ratio), 95% confidence interval (95% CI), and heterogeneity (I2). Forest plots are obtained and p <0.05 is considered statistically significant.
Results: Our study involves the comparison of ICS vs Non-ICS for mortality (N= 207,842 vs 166,217), ICU hospitalization (N= 1,084 vs 9,425), and the risk of hospitalization (N= 1,273 vs 1,676).Of the six studies, five reported mortality. We found a higher mortality rate in patients with asthma (60.88%, 107/160) and chronic obstructive pulmonary disease (COPD) (68.46%, 382/558) among ICS users. The overall mortality is 7.49% (107/1428). We found that ICS use was associated with higher odds of mortality (OR=1.45 95%CI: 1.10-1.91; p=0.009, I2= 68%) amongst COVID-19 patients. In subgroup analysis, higher odds of mortality among COPD patients using ICS was noted [pooled OR: 1.52 (1.24-1.86); p<0.0001; I2=0%]. However, no significant association between ICS and mortality was observed among asthma patients.
Conclusion: ICS is associated with increased mortality and risk for hospitalization in patients with COVID-19 as compared to standard non-steroid-based COVID-19 therapy. It is crucial for healthcare providers to carefully evaluate the potential risks and benefits of ICS usage in the context of COVID-19 management to optimize patient outcomes and safety.
Key words: COVID-19 outcomes, Inhaled corticosteroids, Mortality risk, Respiratory pathologies, Systematic review and meta-analysis.
INTRODUCTION
Coronavirus disease 2019 (COVID-19) is caused by the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) and has been declared a global pandemic by the World Health Organization (WHO) [1]. COVID-19 is the third outbreak of beta coronaviruses in the twenty-first century with over 600 million confirmed cases and over 6million deaths globally as of June 2023 [1]. Approximately 40-60% of chronic obstructive pulmonary disease (COPD) and up to 80% of asthma exacerbations are due to viral infections and include those caused by coronaviruses. Patients with underlying lung diseases like COPD, and asthma who develop COVID-19 have worse outcomes with a case fatality rate of 6.3% [2].
In humans, the SARS-CoV-2 virus targets an essential angiotensin-converting enzyme 2 (ACE2) receptor, which is highly expressed in the epithelial cells of the upper respiratory tract. As a result, it replicates within cells causing cellular injury or death with the release of pro-inflammatory cytokines. In addition, viral particles can stimulate an innate immune response, activating alveolar macrophages and the complement system. This, in turn, causes alveolar and vascular damage, vascular thrombosis, and ventilation-perfusion mismatch gradually involving other organs leading to multiorgan failure and death [3]. Pulmonary inflammation in some cases leads to Bronchiolitis, Acute Respiratory Distress Syndrome (ARDS) and pulmonary fibrosis [4].
Corticosteroids improve lung physiology by reducing inflammation, edema, mucus secretions, and extending inhibitory actions on the transcription and activity of various cytokines at any therapeutically relevant dose through classic genomic mechanisms [5]. In addition, they inhibit SARS-CoV-2 replication in infected epithelial cells by decreasing the expression of ACE2 receptors [6]. Five randomized controlled trials involving 7,692 patients showed that the overall mortality of patients treated with systemic corticosteroids was lower than those not receiving corticosteroids (26% vs. 28%; relative risk (RR)=0.89;95% confidence interval (CI) 0.82-0.96, p=0.003) [7].
Given the benefits of systemic corticosteroids, the use of inhaled corticosteroids (ICS) has been proposed. Yamaya et al. described an in-vitro study where pre-treatment of human respiratory epithelial cells with budesonide combined with long-acting beta-agonist (LABA) and long-acting muscarinic antagonist (LAMA), showed inhibition of human coronavirus HCV-229E replication and cytokine production [8]. Another study reported a 50% reduction in ARDS in at-risk patients using ICS before hospital admission, even after controlling for age, sex, and chronic respiratory diseases [6]. The Cleveland clinic’s COVID-19 registry analysis revealed that ICS therapy did not increase COVID-19 related healthcare utilization, ICU admission, need for intubation, or mortality outcome in patients with COPD. However, the PRINCIPLE trial in the UK noted that the use of Budesonide improves time to recovery and may reduce hospital admissions and death [9]. Currently, there is limited evidence in the literature when analyzing the safety and efficacy of ICS in patients with COVID-19. Therefore, we performed this meta-analysis to address the effectiveness of ICS along with the standard of care in COVID-19.
METHODS
Aim/Endpoint
The primary aim of the study is to evaluate outcomes of COVID-19 amongst patients with pre-existing use of ICS or use of ICS as part of COVID-19 therapy compared to standard COVID-19 treatment. Standard COVID-19 treatment in our study did not involve systemic corticosteroids.
We identified patients with COVID-19 from the studies based on confirmed positive RT-PCR tests. We selected hospitalization, ICU admission, need for mechanical ventilation, mortality, and poor composite outcome as the outcomes of our study.
Search strategy
We followed PRISMA guidelines and MOOSE protocol in conducting the systematic review and meta-analysis comparing the outcomes of ICS and standard COVID-19 therapy. We searched articles on PubMed with keywords: coronavirus disease 2019, COVID-19, SARS-CoV-2, severe acute respiratory syndrome coronavirus 2, and coronavirus to identify our study population; an inhaled corticosteroid, ICS, inhaled dexamethasone, nebulized dexamethasone, and nebulized corticosteroid to identify the intervention/comparison group from December 2019 to December 2021.
Inclusion Criteria: Observation studies and clinical trials describing COVID-19 positive cases and pre-existing use of ICS or ICS as a part of treatment for COVID-19 or clinical trial intervention were included in our meta-analysis.
Exclusion Criteria: Non-human, non-English, and studied pediatrics populations were excluded.
Study selection
Using keywords and selection criteria, we derived appropriate studies. We reviewed abstracts followed by full-length articles to obtain data from the studies for quantitative analysis. VS, MUN, and NA independently screened all of the identified studies and assessed full texts to determine eligibility. Any disagreements were resolved through consensus with UP and NG.
Figure 1 describes the literature search and study selection process.
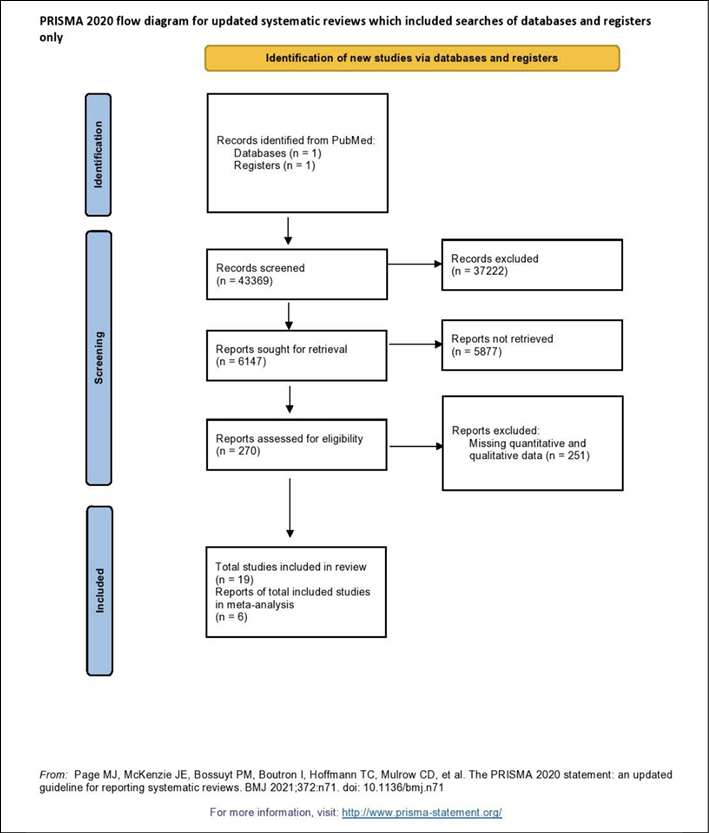
Figure 1 PRISMA flow chart describing the literature search and study selection process.
Data extraction
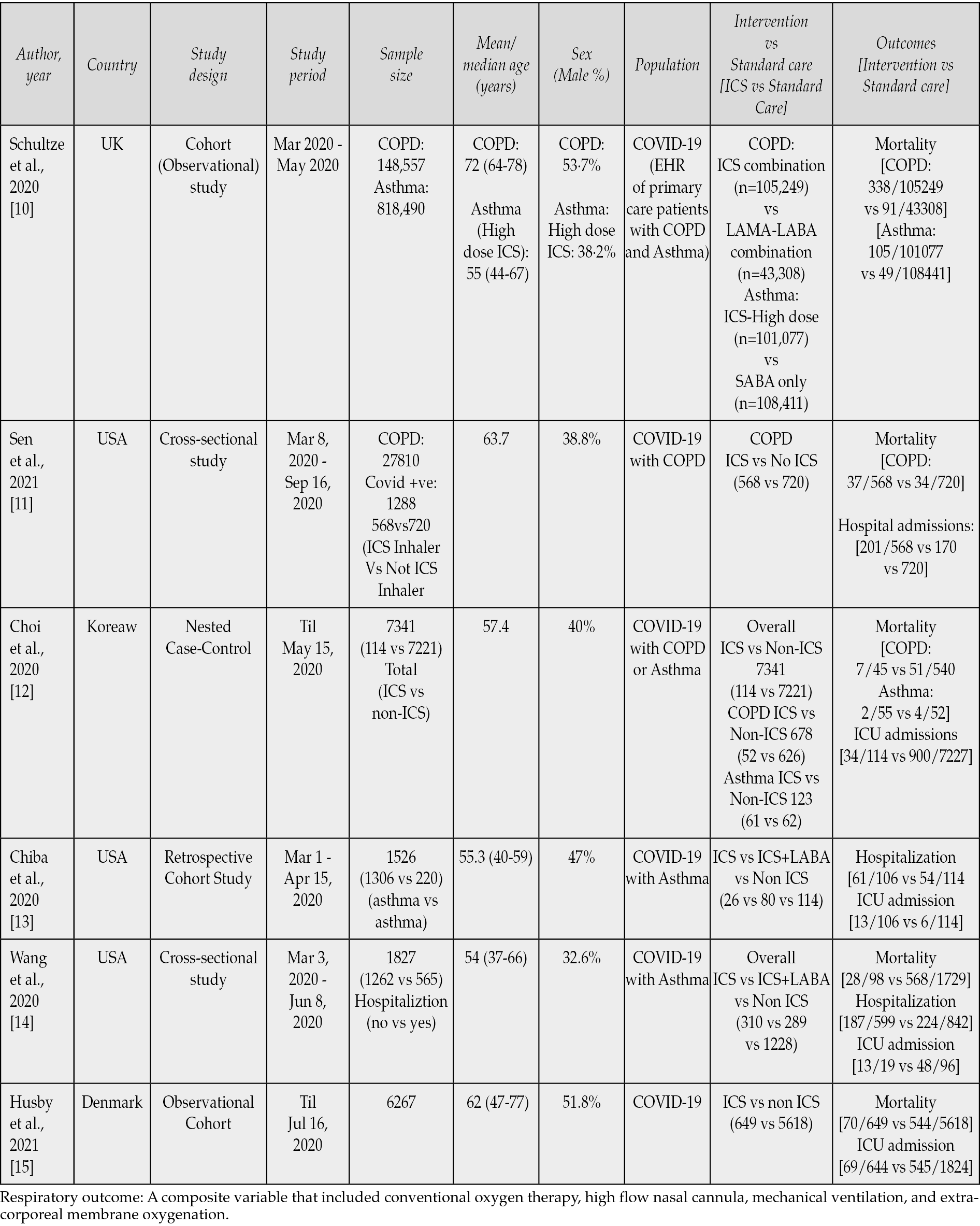
Data on study name, design, period, country, sample size, mean/median age, sex, population characteristics, type of intervention, and various outcomes were collected by IB, NA, RJ, VS using a standard template form, and any disagreements were resolved through consensus with UP and NG. Study characteristics and outcomes are described in Table 1.
Table 1 - Studies describing the use of ICS amongst COVID-19.
Statistical Analysis
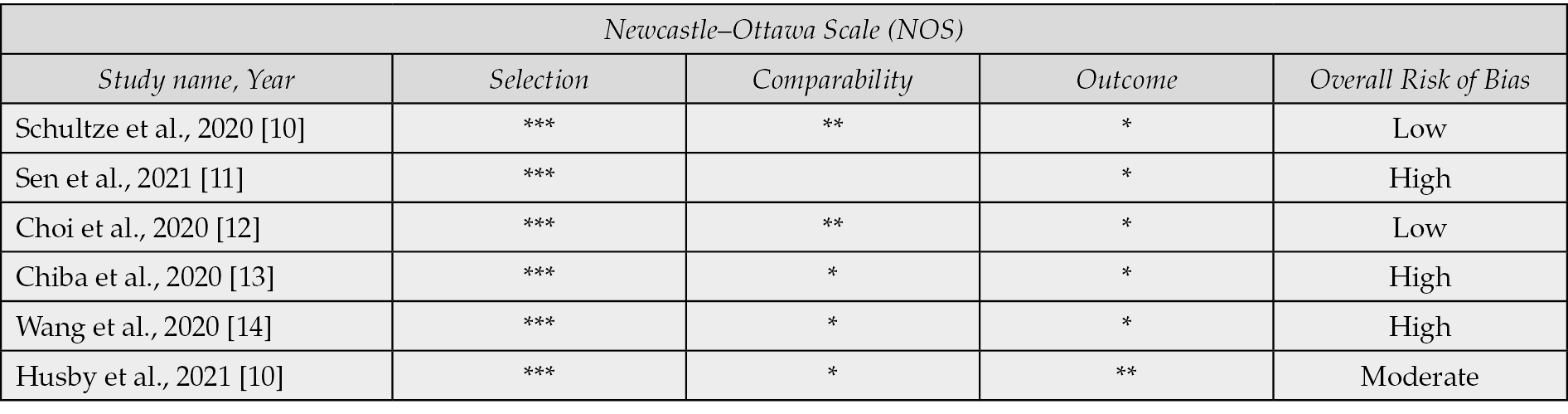
Excel sheet was used to collect the data and Review Manager version 5.3 software was used to analyze the data. We performed random-effects models to estimate the pooled effect size (pooled odds ratio) and 95% confidence interval (95% CI). p value <0.05 was considered statistically significant. To evaluate the heterogeneity, I2 statistics was used and I2>75% represented as high heterogeneity. In such circumstances, sensitivity analysis was performed and studies with higher variability were removed using a funnel plot. Risk of bias analysis was performed and described using the Newcastle-Ottawa Scale (NOS).
RESULTS
Based on eligibility criteria, we included 6 observation studies or clinical trials in our meta-analysis. Our study involved the comparison of ICS vs Non-ICS for mortality (N=207,842 vs 166,217), ICU hospitalization (N=1,084 vs 9,425), and the risk of hospitalization (N=1,273 vs 1,676).
COVID-19 specific mortality in ICS users vs non-ICS users
Amongst 5 studies those reported data on mortality, rate of mortality was high in patients with asthma (60.88% 107/160) and COPD (68.46% 382/558) among ICS users. All other patients had a mortality rate of 7.49% (107/1428). We found that use of ICS was associated with 1.45 higher odds of mortality (95%CI: 1.10-1.91; p=0.009) with 68% of heterogeneity (p=0.003). In the subgroup analysis, COPD patients had higher odds of death [pooled OR: 1.52 (1.24-1.86); p<0.001; I2 =0%; p for I2 = 0.89] while there was no significant increase in mortality in patients with asthma [pooled OR: 1.29 (0.28-5.93); p=0.74; I2 =69%; p for I2 = 0.07]. We did not conduct sensitivity analysis as I2 was <75% (Figure 2).
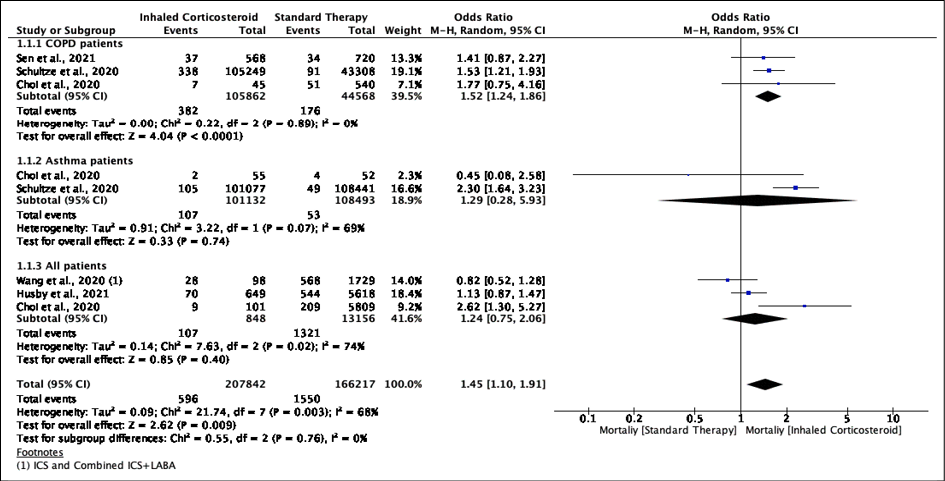
Figure 2 - COVID-19 specific mortality in ICS users vs non-ICS users.
COVID-19 specific ICU admission
In the 5 studies reporting the need for ICU admission, ICU admission among ICS users was higher 18.7% (203/1084) in comparison to those not receiving ICS 16.4% (1552/9425). This was however not statistically significant with pooled OR 1.37 (95%CI: .43-4.36; p=.59) with 96% of heterogeneity (p<.001) Figure 3.1. We conducted sensitivity analysis as I2 was >75%. In the sensitivity analysis, the odds ratio was noted to be significant at OR 2.06 (95% CI: 1.23-3.45; p=.05) (Figure 3.2) after removing the outlying study of Husby et al.

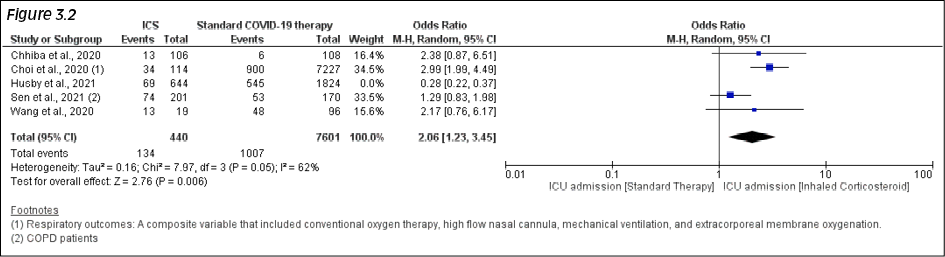
Figure 3.1 - 3.2 - COVID-19 specific ICU admission (Fig 3.1) with sensitivity analysis (Fig 3.2).
COVID-19 specific risk of hospitalization
Amongst the 5 studies, the prevalence of risk of hospitalization among ICS users was higher 35.27% (449/1273) in comparison to non-ICS users 26.73% (448/1676). This was also statistically significant with Pooled OR 1.49 (95%CI: 1.16-1.91; p=0.002) with 52% of heterogeneity (p.13) (Figure 4). We did not conduct sensitivity analysis as I2 was <75%.

Figure 4 - COVID-19 specific Risk of Hospitalization.
Our study has also assessed the risk of bias analysis using the Newcastle-Ottawa Scale and it shows moderate quality of evidence (Table 2).
Table 2 - Newcastle-Ottawa Scale for Risk of Bias Assessment.
DISCUSSION
ICS has been shown to lower ACE2 and TMPRSS2 gene expression in sputum cells and as SARS CoV-2 targets the ACE2 receptors, the protective effect of ICS has been theorized. In vitro studies by Jeon et al and Yamaya et al. also noted an antiviral effect of ICS [8,16,17]. These findings have prompted research into the effectiveness of ICS in COVID-19. Multiple studies have been published over the last year comparing the use of ICS in the management of COVID-19 to standard, non-corticosteroid-based management of the disease. However, the data has been conflicting and to our knowledge, no other meta-analysis has been performed to evaluate the effectiveness of ICS.
We found 6 studies that reported our primary endpoint of mortality and other endpoints of risk for hospitalization and need for ICU admission. In our study, the overall mortality was higher in ICS users with 1.45 higher odds of mortality (95%CI: 1.10-1.91; p=0.009). In the subgroup analysis focusing on mortality, we observed an elevated odds of death associated with the use of ICS in patients diagnosed with COPD. However, in patients with asthma, the use of ICS did not show a statistically significant association with mortality. [pooled OR: 1.29 (0.28-5.93); p=0.74]. The use of ICS is known to increase the risk of pneumonia in patients with COPD while the risk of pneumonia is not increased in patients with asthma. Furthermore, COPD is a progressive, irreversible condition with underlying damage to alveolar lining; ICS can improve symptoms in COPD but cannot reverse the structural damage already done [18, 19]. This may explain the higher mortality in the COPD subgroup.
Our study also found that the need for ICU admission was higher in patients using ICS as compared to those not using ICS (18.7% vs 16.4%) however this was not statistically significant. Husby et al. reported a higher need for ICU admission in patients not using ICS while all the other studies either reported no difference or higher rate of ICU admission in the ICS group. When this study was removed from our analysis, the odds ratio for the need for ICU admission was noted to be higher in the ICS group [pooled OR 2.06 (1.23-3.45); p=0.05].
The need for hospitalization was also much higher in the ICS group (35.27% vs 26.73%) with an OR of 1.49 (95%CI: 1.16-1.91; p=0.002). The higher mortality, need for hospitalization, and ICU admission with ICS use seen in our study is likely due to the increased burden of pulmonary complications in this population. ICS is currently used in patients for conditions that cause increased bronchial hyperreactivity like asthma and COPD conditions. Therefore, patients already on ICS who contract COVID-19 may be at a higher risk for worsening pulmonary disease and subsequent complications.
The strengths of this meta-analysis include lower overall heterogeneity due to the included clinical trials and observational studies being performed in different countries. The sub-group analysis of mortality amongst ICS users with COPD or asthma patients compared to non-ICS users augments the generalizability of our conclusions.
Our study is also not without limitations. Studies published in languages apart from English were not identified or included. We were unable to adjust risk with the inability to ascertain severity indexes. Data regarding pre-existing comorbidities was also lacking.
CONCLUSIONS
In conclusion, our study shows that inhaled corticosteroids are associated with increased mortality and risk for hospitalization in patients with COVID-19 as compared to non-steroid-based management of the disease. Future studies are needed to evaluate the potential risks and benefits of ICS usage in the context of COVID-19 management to optimize patient outcomes and safety More significant, high-quality randomized clinical trials on this topic are warranted before implementing this treatment worldwide.
Grant support/funding
The study had no internal or external funding source.
Disclosure of potential conflict of interest
Authors declare no conflict of interest.
Ethical approval
Though this article does not contain any studies with direct involvement of human participants or animals performed by any of the authors, all procedures performed in studies involving human participants were by the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The data used in this study is de-identified from national inpatient data thus informed consent or IRB approval was not needed for this study.
Availability of data and material
The data is collected from the studies published online, publicly available, and specific details related to data and/or analysis will be made available upon request.
Authors’ contributions
Conceptualization: Syed Nazeer Mahmood, Viraj Shah, Urvish Patel, Thoyaja Koritala.
Methodology: Urvish Patel, Richa Jaiswal, Namratha Gurram, Raghvendra Tirupathi, Thoyaja Koritala.
Acquisition of data: Syed Nazeer Mahmood, Viraj Shah, Muhammad Umair Nawaz, Yelena Pleshkova, Shayaan Negit, Prarthana Desai, Neel Patel.
Formal analysis and investigation: Narayana Varalakshmi Akula, Irina Balan, Divya Manivannan.
Writing - original draft preparation: Syed Nazeer Mahmood, Viraj Shah, Muhammad Umair Nawaz, Narayana Varalakshmi Akula, Irina Balan, Divya Manivannan, Neel Patel.
Writing - review, critical feedback, and editing: Yelena Pleshkova, Shayaan Negit, Prarthana Desai, Richa Jaiswal, Namratha Gurram, Neel Patel, Raghvendra Tirupathi, Thoyaja Koritala.
Resources: Neel Patel, Raghvendra Tirupathi, Thoyaja Koritala.
Supervision: Raghvendra Tirupathi, Thoyaja Koritala.
REFERENCES
[1] Jamaati H, Hashemian SM, Farzanegan B, et al. No clinical benefit of high dose corticosteroid administration in patients with COVID-19: A preliminary report of a randomized clinical trial. Eur J Pharmacol. 2021; 897: 173947. doi: 10.1016/j.ejphar.2021.173947.
[2] Halpin DMG, Singh D, Hadfield RM. Inhaled corticosteroids and COVID-19: a systematic review and clinical perspective. Eur Respir J. 2020; 55 (5): 2001009. doi: 10.1183/13993003.01009-2020.
[3] Mainous AG 3rd, Rooks BJ, Wu V, Orlando FA. COVID-19 post-acute sequelae among adults: 12 month mortality risk. Front Med. 2021; 8, 778434. https://doi.org/10.3389/fmed.2021.778434.
[4] Vasarmidi E, Tsitoura E, Spandidos D, Tzanakis N, Antoniou K. Pulmonary fibrosis in the aftermath of the Covid-19 era. Exp Ther Med. 2020; 20 (3): 2557-2560. doi: 10.3892/etm.2020.8980.
[5] Tang Y, Liu J, Zhang D, Xu Z, Ji J, Wen C. Cytokine Storm in COVID-19: the current evidence and treatment strategies. Front Immunol. 2020; 11: 1708. doi: 10.3389/fimmu.2020.01708.
[6] Nicolau DV, Bafadhel M. Inhaled corticosteroids in virus pandemics: a treatment for COVID-19? Lancet Respir Med. 2020; 8 (9): 846-847. doi: 10.1016/S2213-2600(20)30314-3.
[7] Pasin L, Navalesi P, Zangrillo A, et al. Corticosteroids for patients with Coronavirus Disease 2019 (COVID-19) with different disease severity: a meta-analysis of randomized clinical trials. J Cardiothorac Vasc Anesth. 2021; 35 (2): 578-584. doi: 10.1053/j.jvca.2020.11.057.
[8] Yamaya M, Nishimura H, Deng X, et al. Inhibitory effects of glycopyrronium, formoterol, and budesonide on coronavirus HCoV-229E replication and cytokine production by primary cultures of human nasal and tracheal epithelial cells. Respir Investig. 2020; 58 (3): 155-168. doi: 10.1016/j.resinv.2019.12.005.
[9] Yu LM, Bafadhel M, Dorward J, et al. Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial. The Lancet. 2021; 398 (10303): 843-855. doi: 10.1016/S0140-6736(21)01744-X.
[10] Schultze A, Walker AJ, MacKenna B, et al. Risk of COVID-19-related death among patients with chronic obstructive pulmonary disease or asthma prescribed inhaled corticosteroids: an observational cohort study using the OpenSAFELY platform. Lancet Respir Med. 2020; 8 (11): 1106-1120. doi: 10.1016/S2213-2600(20)30415-X.
[11] Sen P, Majumdar U, Zein J, Hatipoğlu U, Attaway AH. Inhaled corticosteroids do not adversely impact outcomes in COVID-19 positive patients with COPD: An analysis of Cleveland Clinic’s COVID-19 registry. Loukides S, ed. PLOS ONE. 2021; 16 (6): e0252576. doi:10.1371/journal.pone.0252576.
[12] Choi JC, Jung SY, Yoon UA, et al. Inhaled Corticosteroids and COVID-19 Risk and Mortality: A Nationwide Cohort Study. J Clin Med. 2020; 9 (11): 3406. doi:10.3390/jcm9113406.
[13] Chhiba KD, Patel GB, Vu THT, et al. Prevalence and characterization of asthma in hospitalized and nonhospitalized patients with COVID-19. J Allergy Clin Immunol. 2020; 146 (2): 307-314. e4. doi: 10.1016/j.jaci.2020.06.010.
[14] Wang L, Foer D, Bates DW, Boyce JA, Zhou L. Risk factors for hospitalization, intensive care, and mortality among patients with asthma and COVID-19. J Allergy Clin Immunol. 2020; 146 (4): 808-812. https://doi.org/10.1016/j.jaci.2020.07.018.
[15] Husby A, Pottegård A, Hviid A. Association between inhaled corticosteroid use and COVID-19 outcomes. Pharmacoepidemiol Drug Saf. 2021; 30 (11): 1486-1492. doi: 10.1002/pds.5345.
[16] Peters MC, Sajuthi S, Deford P, et al. COVID-19–related Genes in sputum cells in asthma. relationship to demographic features and corticosteroids. Am J Respir Crit Care Med. 2020; 202 (1): 83-90. doi: 10.1164/rccm.202003-0821OC.
[17] Jeon S, Ko M, Lee J, et al. Identification of antiviral drug candidates against SARS-CoV-2 from FDA-Approved Drugs. Antimicrob Agents Chemother. 2020; 64 (7): e00819-20. doi: 10.1128/AAC.00819-20.
[18] Yang M, Du Y, Chen H, Jiang D, Xu Z. Inhaled corticosteroids and risk of pneumonia in patients with chronic obstructive pulmonary disease: A meta-analysis of randomized controlled trials. Int Immunopharmacol. 2019; 77: 105950. doi: 10.1016/j.intimp.2019.105950.
[19] Kim SH. Risk of Pneumonia Associated With the Use of Inhaled Corticosteroids in Asthma. Allergy Asthma Immunol Res. 2019; 11 (6): 760. doi:10.4168/aair.2019.11.6.760.