Le Infezioni in Medicina, n. 3, 306-322, 2023
doi: 10.53854/liim-3103-5
REVIEWS
The sources of antimicrobial peptides against Gram-positives and Gram-negatives: our research experience
Giulio Rizzetto1, Daisy Gambini1, Andrea Maurizi1, Elisa Molinelli1, Edoardo De Simoni1, Francesco Pallotta2, Lucia Brescini2, Oscar Cirioni2, Annamaria Offidani1, Oriana Simonetti1, and Andrea Giacometti2
1Clinic of Dermatology, Department of Clinical and Molecular Sciences, Polytechnic University of Marche, Ancona, Italy;
2Clinic of Infectious Diseases, Department of Biomedical Sciences and Public Health, Polytechnic University of Marche, Ancona, Italy
Article received 19 May 2023, accepted 3 July 2023
Corresponding author
Oriana Simonetti
E-mail: o.simonetti@staff.univpm.it
SummaRY
Antibiotic resistance of Gram-positive and Gram-negative bacteria is becoming increasingly prevalent. For this reason, the search for new molecules that can overcome current resistance and also recover antibiotics that are no longer effective is becoming increasingly urgent. Our research group at the ‘Polytechnic University of Marche’ managed to study the effectiveness of certain antimicrobial peptides (AMPs). We decided to review our experience with AMPs by classifying them according to their origin and evaluating their effect on Gram-negative and Gram-positive bacteria. AMPs can derive from mammals, amphibians, microorganisms, and insects. In conclusion, our research experience shows that the richest source of AMPs are amphibians. However, the studies done are mainly in vitro or in animal models, requiring further human studies to assess the efficacy and safety of these molecules. AMPs may be a new therapeutic option for infections sustained by multi-resistant micro-organisms and for overcoming the mechanisms of resistance to antibiotics currently used.
In particular, combining AMPs with antibiotics, including those with limited antimicrobial activity due to antimicrobial resistance, has often shown a synergistic effect, increasing or restoring their efficacy. The possibility of using manageable and relatively safe antibiotics again is crucial, considering the widespread increase in bacterial resistance in hospitals and the community. Despite a plethora of research on AMPs and their application as potential treatment on infectious diseases, this area needs further exploration. There is evidence that the characteristics of AMPs can seriously improve through structural chemical modifications and different delivery systems to become alternatives drugs to conventional antibiotics. The aim is to provide an overview of the possible sources from which AMPs are extracted, evaluating their action exclusively on Gram-positive and negative bacteria. This is to determine, based on our experience, which might be the most promising sources of AMPs for future research as well.
Keywords: AMPs, antibiotic resistance, Gram positive bacteria, Gram negative bacteria.
INTRODUCTION
Antibiotic resistance of Gram-positive and Gram-negative bacteria is becoming increasingly prevalent, partly due to the widespread use of antibiotics not only in the medical field, but also in agriculture and animal husbandry [1]. For this reason, the search for new molecules that can overcome current resistance and also enable the recovery of antibiotics that are no longer effective is becoming increasingly urgent. Consequently, our research group at the ‘Polytechnic University of Marche’ managed to study the action of certain antimicrobial peptides (AMPs), evaluating their action against different microorganism in vitro or in animal models of sepsis, prosthetic/device infections, and skin wound infections. Our experience regards mainly in vitro studies and studies conducted on animals, not observational clinical studies. We decided to review our experience with AMPs by subdividing and classifying them according to their origin and evaluating their effect on Gram-negative and Gram-positive bacteria. AMPs can derive from mammals (especially human defense peptides), amphibians, microorganisms, and insects, as inferred from the antimicrobial peptide database [2]. It is important to note that AMPs are active not only against bacteria, but also against fungi, allowing resistance to traditional therapies to be overcome [3-9]. This is why the research field of AMPs is very promising for the future, especially considering that associations with traditional antibiotics have often proved to be synergic. A limitation of our experience is that no studies have been conducted on humans, but only on animal models or in vitro. However, we believe that the potential of some AMPs may also be useful in humans, offering an overview of available molecules that could in the near future also be used in clinical studies in patients.
MATERIAL AND METHODS
We performed a narrative review of studies from our research group at the Università Politecnica delle Marche, consisting of the dermatology clinic and the infectious diseases clinic. We searched on Pubmed for the following keywords in combination: sources, AMPs, Gram-positive, Gram-negative, mammalian, insects, amphibians, and synthetic peptides. Only English-language studies, produced by our research group, and in which AMPs were evaluated in vivo in an animal model or in vitro, were included. The aim was to provide an overview of the possible sources from which AMPs are extracted, evaluating their action exclusively on Gram-positive and Gram negative bacteria. This is to determine, based on our experience, which might be the most promising sources of AMPs for future research as well.
AMPS DERIVED FROM MAMMALS (Table 1)
Mammalian-derived AMPs are components of the innate immune system, mainly represented by cathelicidins and denfensins. Most of these AMPs have been isolated from humans, but also from cattle, sheep, and other vertebrates [10]. Furthermore, cathelicidins and denfensins can act directly against pathogens, but also appear to be able to regulate the immune response, apoptosis and wound healing [11].
Table 1 - Mammalian-derived AMPs, summary of main studies.
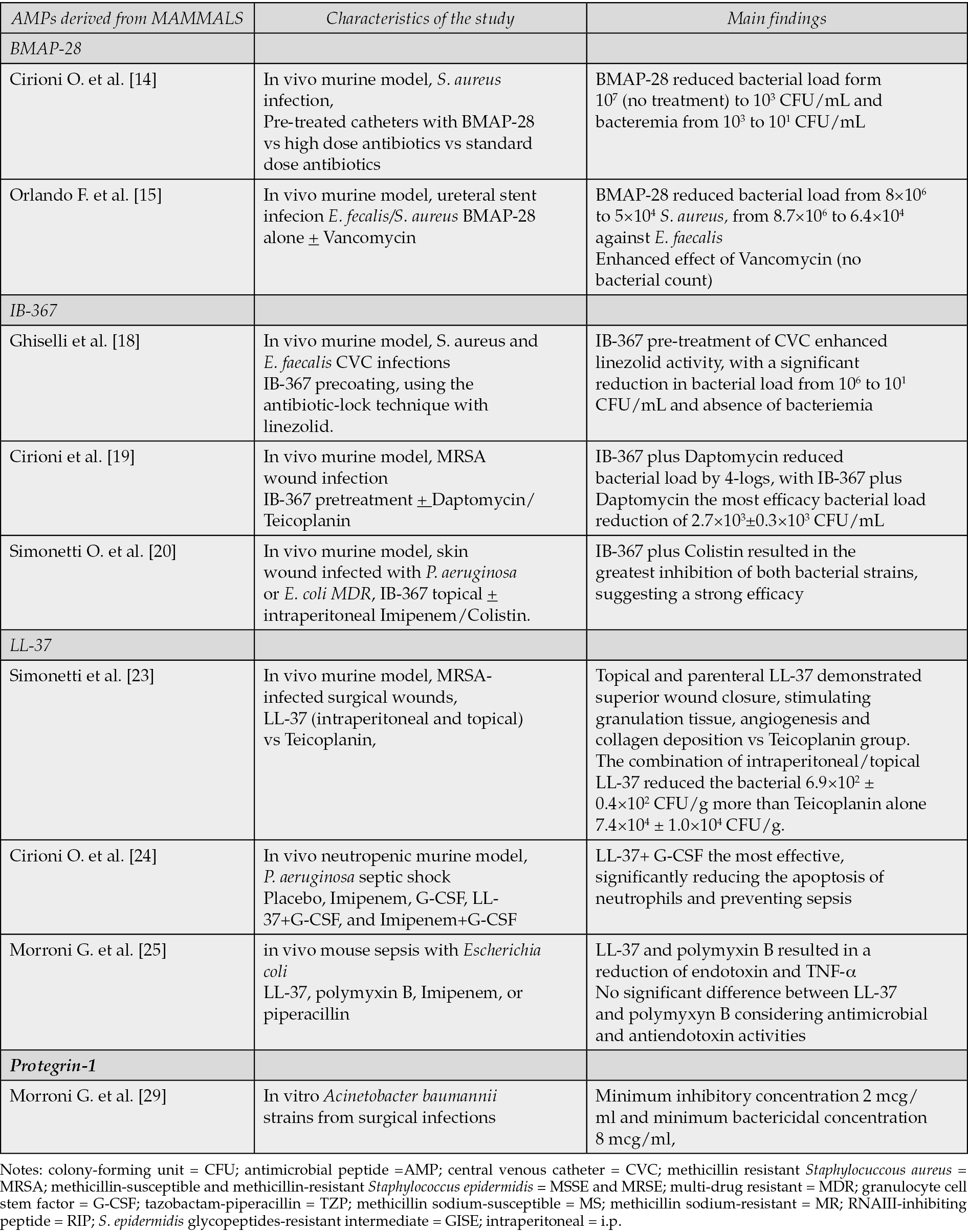
Bovine myeloid antimicrobial peptide (BMAP)-28
BMAP-28 is a bovine antimicrobial peptide of the cathelicidin family, showing the ability to kill antibiotic resistant bacteria in vitro. Moreover recent in vivo studies have demonstrated BMAP-28 efficacy in reducing mortality in different infections [12, 13].
The efficacy of BMAP-28 was assessed on both central venous catheter (CVC) and uretheral stent in two different studies. Some bacteria tend to develop biofilms on the surfaces of medical devices such as long-term silicone catheters, such as CVCs. Biofilms are structured microbial communities that occur as surface-attached communities or suspended aggregates which are able to start the production of an extracellular polysaccharide matrix. Antibiotic penetration within the biofilm is scarce and various components work together within a biofilm to reduce, or fully prevent, antibiotic effectiveness. In addition sessile bacteria living in a biofilm can exhibit a 10 to 1,000-fold increase in antibiotic resistance compared to planktonic bacteria.
Cirioni et al. described the potential of BMAP-28 pre-coating in the treatment of S. aureus central venous catheter-associated infections [14]. The authors demonstrated, using an in vivo murine model, that catheters pre-treated with BMAP-28 or high-dose antibiotics have a lower bacterial load compared to catheters with standard-dose antibiotics or without BMAP-28 (from 107 to 103 colony-forming unit (CFU)/mL and bacteremia from 103 to 101 CFU/mL). A further significant reduction in bacterial load (from 107 to 101 CFU/mL) was observed when catheters were impregnated with BMAP-28 and then treated with a higher dose of antibiotics. In vitro studies confirm these results [14].
The efficacy of BMAP-28 alone and in combination with Vancomycin was assessed in the treatment of animal models of uretheral stent infection induced by Enterococcus faecalis and Staphylococcus aureus. The study suggests that the BMAP-28- impregnated uretheral stent has a lower rates of infection, in fact in vivo model revealed that BMAP-28 reduced bacterial load (from 8 × 106 to 5 × 104 against S. aureus and from 8.7 × 106 to 6.4 × 104 against E. faecalis) and enhanced the effect of Vancomycin (no bacterial count); and in vitro studies support these results [15].
Finally, BMAP-28 showed to reduce nitric oxide (NO) production and tumor necrosis factor (TNF)-alpha release. In murine models, similar lethality to antibiotics as Clarithromycin and Imipenem against Staphylococcus aureus was reported. Furthermore, it was superior to neutralize bacterial products of Gram-positives, improving severe staphylococcal infections when used in combination with other antibiotics [16].
IB-367
IB-367, known also as Iseganan, is a synthetic protegrin, cysteine-rich AMP, with bactericidal and fungicidal activity, isolated from porcine leukocytes [17].
Ghiselli et al. assessed the efficacy of IB-367 pre-coating in the treatment of CVC infections using the antibiotic-lock technique with linezolid in murine model. The study showed that IB-367 pre-treatment of CVC enhanced linezolid activity against S. aureus and E. faecalis, causing a significant reduction in bacterial load on CVC (from 106 to 101 CFU/mL) and absence of bacteremia [18].
Cirioni and colleagues investigated the activity of IB-367, exploring whether IB-367 pre-treatment could improve the efficacy Daptomycin and Teicoplanin against methicillin resistant Staphylococcus aureus (MRSA) wound infection in a mouse model. The study’s primary outcomes were quantitative bacterial culture and analysis of natural killer (NK) cytotoxicity and leukocyte phenotype. While antibiotics alone showed comparable antimicrobial efficacy, their combination with IB-367 significantly enhanced their activity. IB-367 plus Daptomycin reduced bacterial load by 4-logs, demonstrating the most efficacy (bacterial load reduction of 2.7×103 ± 0.3×103 CFU/mL) [19].
In conclusion, IB-367 could be considered an interesting association with conventional antibiotics for the treatment of CVC and other medical devices and for the treatment of MRSA wound infection.
Considering Gram-negative bacteria, topical IB-367 showed interesting activity against multi-drug resistant (MDR) P. aeruginosa or E. coli [20]. In a mouse model of skin wound infection, IB-367 was administered topically alone and associated with intra-peritoneal Imipenem/Colistin. IB-367 plus Colistin resulted in the greatest inhibition of both bacterial strains, suggesting a strong efficacy [20]. In conclusion, IB-367 may be an excellent candidate for topical therapy of Gram-negative infected wounds in the future.
LL-37
LL-37 is a human antimicrobial peptide categorized as a cathelicidin, showing a broad-spectrum activity against a variety of pathogens, including Gram-positive and Gram-negative bacteria, viruses, and fungi [21]. Additionally, LL-37 demonstrated other biological activities, such as the regulation of inflammation responses and an important role in wound closure and angiogenesis [22].
Simonetti et al. compared the efficacy of LL-37 with that of Teicoplanin in MRSA-infected surgical wounds in mice. LL-37 had a greater effect than Teicoplanin on wound healing processes in MRSA-infected mice, although it had a lesser effect on bacterial culture growth. The administration of LL-37 alone, intra-peritoneally, reduced the bacterial count to 7.1×105 ± 0.6×105CFU/g. The combination of intra-peritoneal and topical LL-37 further reduced the bacterial count to 6.9×102 ± 0.4×102 CFU/g. In comparison, Teicoplanin, when administered intra-peritoneally alone, produced a bacterial count of 7.4×104 ± 1.0×104 CFU/g. Furthermore, the administration of LL-37, both topically and parenterally, demonstrated superior wound closure by stimulating the formation of granulation tissue and promoting angiogenesis and collagen deposition, resulting in better organized epithelial reconstitution compared to the Teicoplanin group [23].
Considering Gram-negatives, LL-37 may be useful against P. aeruginosa. In a neutropenic murine model with P. aeruginosa septic shock, all therapy groups (Imipenem, granulocyte cell stem factor (G-CSF), LL-37+G-CSF, and Imipenem+G-CSF) were superior to placebo. LL-37+ G-CSF association was the most effective by significantly reducing the apoptosis of neutrophils and preventing sepsis [24]. Similar results were reported in mouse sepsis with Escherichia coli [25]. All treatments (LL-37, polymyxin B, Imipenem, or piperacillin) reduced the death of mice, but exclusively LL-37 and polymyxin B resulted in a reduction of endotoxin and TNF-α plasma levels. Moreover, there was no significant difference between LL-37 and polymyxin B considering antimicrobial and antiendotoxin activities. In the light of its anti-inflammatory and immunomodulatory effect, LL-37 emerges as a candidate for the treatment of Gram-negative sepsis. Finally, LL-37 showed a good action against biofilm formation of Escherichia coli when combined with Colistin [26].
Similarly, another cathelicidin with activity against P. aeruginosa MDR is tritrpticin, showing a complete inhibition of the procoagulant activity of lipopolysaccharides [27].
Protegrin-1
Protegrin-1 is a18-amino-acid beta-hairpin AMP that belongs to the cathelicidin family. It showed a strong bactericidal action and synergy with Colistin as well in a murine model. On the other hand, no effect on biofilm was reported. Furthermore, resistance to PG-1 was not detected. For this reasons, protegrin-1 may be an interesting future option for the treatment of Gram-negative MDR infections [28]. In particular, Acinetobacter baumannii represents a Gram-negative associated with multiple antibiotic resistance. The minimum inhibitory concentration and minimum bactericidal concentration of protegrin-1 were 2 mcg/ml and 8 mcg/ml, respectively, considering in vitro cultures of A. baumannii MDR [29].
AMPS DERIVED FROM AMPHIBIANS (Table 2)
Amphibians are a very important source of AMPs. In particular, frogs are the amphibians from which the most AMPs are derived. The skin secretion of frogs of the family Pipidae, including Silurana, Xenopus, Hymenochirus, and Pseudhymenochirus, is very rich in peptides that help to protect against infection [30]. In our research experience, amphibian-derived AMPs are in fact the most numerous and most studied.
Table 2 - AMPs derived from amphibians, summary of main studies.
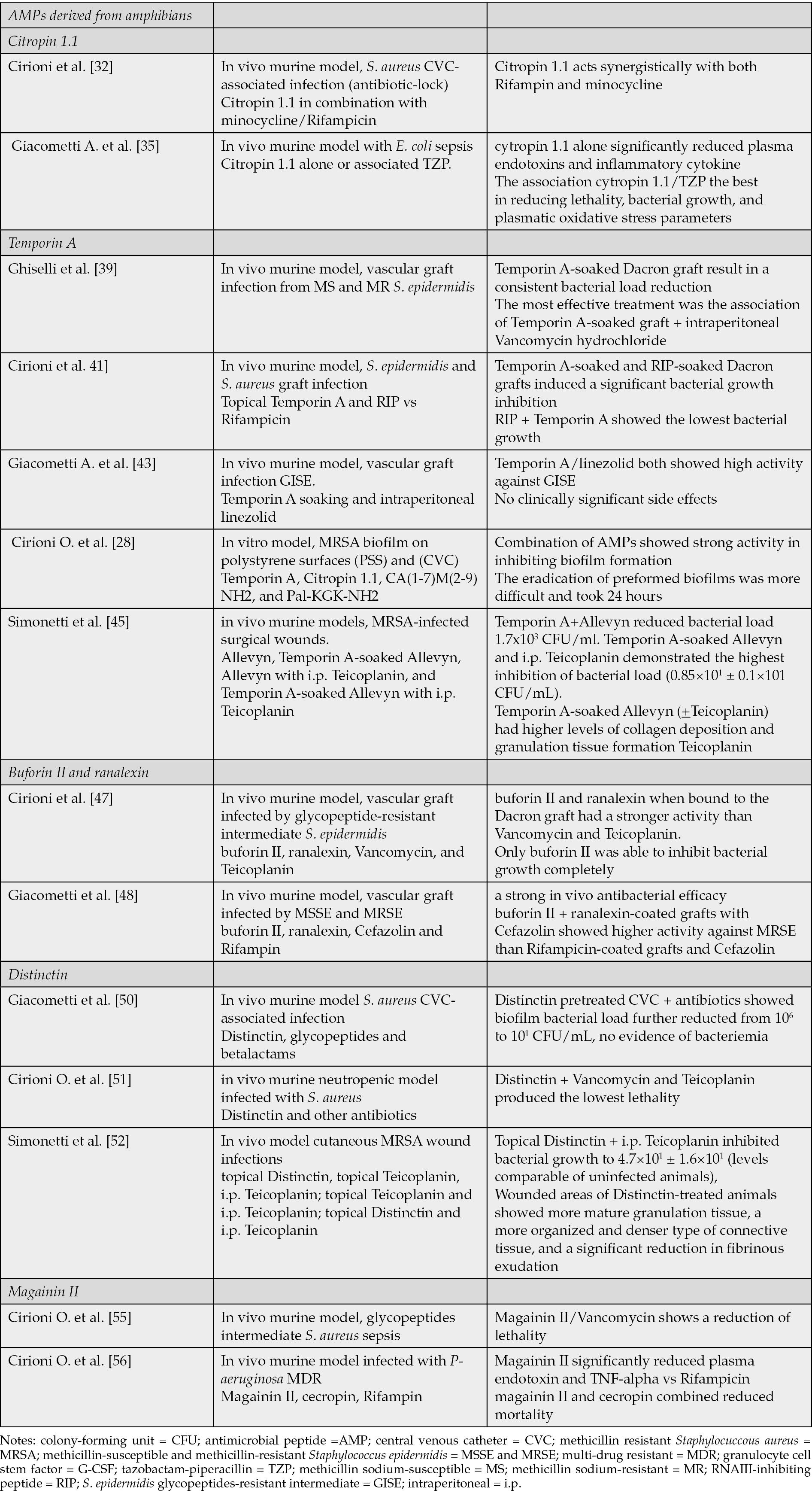
Citropin 1.1
Citropin 1.1 is the major AMP produced by the green tree frog, Litoria citropa. It is a wide-spectrum amphibian antimicrobial peptide [31].
Cirioni et al. assessed the efficacy of Citropin 1.1 in combination with minocycline and Rifampicin, in the treatment and prevention of S. aureus CVC-associated infection using the antibiotic-lock technique. In vitro studies show that biofilms were strongly affected by the presence of Citropin 1.1. Furthermore, this study demonstrated that Citropin 1.1 acts synergistically with both Rifampin and minocycline. In fact Citropin 1.1 acts synergistically with hydrophobic antibiotics, probably inducing a damage of the membrane allowing a maximal entry of the hydrophobic substrates [32]. Finally, Citropin 1.1 showed its efficacy against staphylococci and streptococci with a concentration ranging from 1 to 16 mg/L. When Citropin 1.1 was associated with antibiotics, such as Clarithromycin and doxyxline, synergy was reported [33, 34].
Regarding Gram-negative bacteria, in mice with E. coli sepsis Citropin 1.1 was administered alone or associated with tazobactam-piperacillin (TZP). Although all the treatment groups reduced the lethality compared with controls, cytropin 1.1 alone significantly reduced plasma endotoxins and inflammatory cytokines, while TZP exerted an opposite effect. The association of cytropin 1.1 and TZP was the best in reducing lethality, bacterial growth, and plasmatic oxidative stress parameters. Therefore, citroprin 1.1 has not only antimicrobial but also immunomodulatory activity and may be an interesting option in case of severe Gram-negative infection [35]. The role of this molecule could be interesting in case of infections in patients with increased oxidative stress also due to other concomitant pathologies, such as psoriasis [36, 37].
Uperine 3.6
Uperine 3.6, a broad-spectrum AMP, was isolated from the amphibian Uperoleia mjobergii. Only 17 amino acids are included in its structure, and so it represents the smallest antibiotic peptide isolated from amphibians. Although most of the antibiotics tested were more effective than uperine 3.6, it was effective against both susceptible and multiresistant germs [38].
Temporin A
Temporin A is a peptide amide with an antimicrobial activity against a wide spectrum of microorganisms, including antibiotic-resistant Gram-positive cocci, found in the skin of the European red frog, Rana temporaria [39]. The mechanism of action of Temporin A is still unclear, however there are different hypotheses: Temporin A could act inserting into the hydrophobic core of the cell membrane, troughs interaction with bacterial phospholipids, altering enzyme activities, binding to DNA [40].
Different studies investigated the role of Temporin A in the treatment of medical devices infections (vascular graft, polystyrene surfaces and central venous catheters) and of infected wounds.
Ghiselli et al. evaluated the efficacy of Temporin A as a prophylactic agent in a rat model of vascular graft infection from methicillin sodium-susceptible (MS) and methicillin sodium-resistant (MR) Staphylococcus epidermidis. According to in vitro studies both MR and MS were similarly susceptible to Temporin A. In vivo studies showed that the use of a Temporin A-soaked Dacron graft in vascular surgery can result in a consistent bacterial load reduction. The most effective treatment was the association of a Temporin A-soaked graft and intraperitoneal Vancomycin hydrochloride, which inhibited bacterial growth for both the methicillin-resistant (MR) and methicillin-sensitive (MS) strains [39].
Cirioni et al. tested the efficacy of topical Temporin A and RNAIII-inhibiting peptide (RIP) compared to Rifampicin in preventing S. epidermidis and S. aureus graft infection in a subcutaneous rat pouch model [41].
RIP is a heptapeptide isolated from culture supernatants of S. xylous and has a strong activity against S. aureus and S. epidermidis. RIP inhibits cell-cell communication, also known as quorum sensing, preventing bacterial adhesion and virulence [42]. This study showed that the use of Temporin A-soaked and RIP-soaked Dacron grafts induced a significant bacterial growth inhibition. The combination of RIP and Temporin A showed the lowest bacterial growth (negative quantitative cultures for Vancomycin intermediate S. epidermidis (VISE)4 and from 6 × 107 to 6.9 × 101 CFU/mL for VISA4). More specifically, Temporin A had a high antistaphylococcal activity, independent of the level of resistance shown by the isolates. RIP was more effective against staphylococcal strains when used alone than Temporin A or Rifampicin alone. In conclusion these molecules appear potentially useful for antimicrobial chemoprophylaxis in vascular surgery [41].
Another study investigated the ability of Temporin A soaking and intraperitoneal linezolid to prevents vascular graft infection in a rat model with S. epidermidis glycopeptides-resistant intermediate (GISE). Temporin A and linezolid both showed high activity against the GISE clinical strain without clinically significant side effects, representing a valid opportunity of prophylaxis in vascular surgery [43].
Temporin A was also studied in combination with Citropin 1.1, CA(1-7)M(2-9)NH2, and Pal-KGK-NH2 for their synergistic activity against MRSA biofilms developed on polystyrene surfaces (PSS) and central venous catheters (CVC). Combination of AMPs showed strong activity in inhibiting biofilm formation on PSS and even on CVC where is common the formation of MRSA biofilm. The eradication of preformed biofilms, on the other hand, was more difficult and took 24 hours [28].
In addition, Temporin A inhibited the production of NO, TNF-alpha, and IL-6 by macrophages in mouse models, and was effective against antibiotic-resistant staphylococci and streptococci. In particular, it demonstrated particularly high efficacy against S. aureus 6 hours after injection. The most effective antibiotic used in combination was Imipenem (lethality rates of 25% Temporin A, 20% Imipenem, 10% Temporin A+Imipenem). Temporin A is able to facilitate the passage of Imipenem through the bacterial membrane by destructuring it while both go on to act on peptidoglycan [44].
Simonetti and colleagues conducted a study to examine the impact of using Temporin A topically in murine models with MRSA-infected surgical wounds. The mice were divided into several groups: those treated with drug-free Allevyn, Temporin A-soaked Allevyn, drug-free Allevyn with daily intraperitoneal Teicoplanin, and Temporin A-soaked Allevyn with daily intraperitoneal Teicoplanin. Results indicated that the combination of Temporin A and Allevyn reduced bacterial load to 1.7x103 CFU/ml. However, the group that received Temporin A-soaked Allevyn and i.p. Teicoplanin demonstrated the highest inhibition of bacterial load (0.85×101 ± 0.1×101 CFU/mL). Furthermore, histological examination revealed that infected mice treated with Temporin A-soaked Allevyn (+Teicoplanin) had higher levels of collagen deposition and granulation tissue formation compared to the other groups. There was also a significant increase in serum VEGF expression observed in mice receiving Temporin A topically with or without intraperitoneal Teicoplanin [45].
Buforin II and ranalexin
Amphibian tissues are the source from which Buforin II and Ranalexin are derived. They are polycationic peptides with antimicrobial activity on the cytoplasmatic membrane. Buforin II was derived from Buforin I, a peptide from the stomach of a toad (Bufo bufo gargarizans), while Ranalexin was derived from the skin of a bullfrog (Rana catasbeiana) [46].
These two peptides were evaluated by Cirioni et al. to prevent infection of the vascular prosthetic graft by glycopeptide-resistant intermediate Staphylococcus epidermidis.
In vivo studies demonstrated that Buforin II and Ranalexin when bound to the Dacron graft had a stronger activity (from 4.9×106 to 1.9×102 CFU/mL) than Vancomycin (from 4.9×106 to 6.2×103 CFU/mL) and Teicoplanin (from 4.9×106 to 5.1×104 CFU/mL). In particular, only buforin II was able to inhibit bacterial growth completely [47].
Giacometti et al. investigated the efficacies of the same polycationic peptides comparing their activity to that of Rifampicin in the prevention of methicillin-susceptible and methicillin-resistant Staphylococcus epidermidis vascular prosthetic graft infections. This study found that these peptides have a strong in vivo antibacterial efficacy, in fact their polycationic activities against Staphylococcus epidermidis were not significantly different from that of Rifampin. The combinations of buforin II and ranalexin-coated grafts with Cefazolin showed higher activity against the methicillin-resistant strain (no infection detected) than that of the combination of Rifampicin-coated grafts and Cefazolin [48].
Policationic peptides appear potentially useful for future topical antimicrobial treatments, such as peri and preoperative chemoprophylaxis in prosthetic surgery.
Distinctin
Distinctin is an AMP derived from Phyllomedusa distincta with a heterodimeric structure consisting of two different polypeptide chains linked by a disulfide bond. It has a lytic activity on unilamellar vesicles, suggesting their possible action on bacterial membranes [49].
Giacometti et al. assessed the efficacy of Distinctin in the treatment of Staphylococcus aureus CVC-associated infection. The study evaluated the ability of Distinctin in inhibiting the attachment of S. aureus to CVCs and in increasing its susceptibility to glycopeptides and betalactams once it is adherent. The in vitro study showed a strong activity of Distinctin on the biofilm and the ability to enhance the efficacy of antibiotics when used in combination. When antibiotics where used in Distinctin pretreated CVC, biofilm bacterial load was further reduced from 106 to 101 CFU/mL with no evidence of bacteriemia [50].
Furthermore, in neutropenic mice infected with Staphylococcus aureus, this molecule demonstrated efficacy when administered intravenously, alone or in combination with other antibiotics. Specifically, the combination with Vancomycin and Teicoplanin produced the lowest lethality rate in this murine model [51]. Distinctin displays potential as an adjunctive agent to antibiotics in the treatment of CVC-related infections.
Simonetti et al. studied the efficacy of Distinctin in the management of cutaneous MRSA wound infections. Mice were treated with topical Distinctin, topical Teicoplanin, intraperitoneal Teicoplanin; topical Teicoplanin and daily intraperitoneal Teicoplanin; topical Distinctin and daily intraperitoneal Teicoplanin. Topical Distinctin combined with i.p. Teicoplanin inhibited bacterial growth to 4.7×101 ± 1.6×101 (levels comparable with those observed in uninfected animals), but the combination of topical and i.p Teicoplanin proved to be the most effective treatment in reducing bacterial counts. In addition, wounded areas of Distinctin-treated animals showed more mature granulation tissue, a more organized and denser type of connective tissue, and a significant reduction in fibrinous exudation [52].
Aurein 1.2
Aurein 1.2 is a 13 amino acid, amphipathic peptide. its action against Gram-positive cocci occurs at concentrations between 1 and 16 mg/litre. Clarithromycin and minocycline have shown synergistic activity with aurein 1.2. In particular, the latter promotes the entry of hydrophobic substrates by increasing membrane permeability and altering membrane organisation [53].
Magainin II
Magainin II is an alpha-helical AMP with in vitro activity and in vivo efficacy against glycopeptides intermediate Staphylococcus aureus. More specifically, the combination of Magainin II/Vancomycin shows a reduction of lethality in murine models with staphylococcal sepsis (death of 1/20 vs 6/20 Vancomycin vs 10/20 Magainin II). This peptide inserts itself into the cytoplasmic membrane, activating murine bacterial peptidoglycan hydrolases and so destroying the pathogen [54].
Magainin II was also effective against Gram-negative bacteria. In a mouse model infected with P- aeruginosa MDR, Magainin II significantly reduced plasma endotoxin and TNF-alpha levels compared to the control group and those treated with Rifampicin. In addition, Magainin II and cecropin A were combined in this study, showing a synergistic effect against P. aeruginosa MDR, reducing mortality [55].
AMPS DERIVED FROM INSECTS
Antimicrobial peptides are mainly synthesized in fat bodies and blood cells of insects, which is one of the main reasons for insects’ strong adaptability to survival [56]. Cecropin, an alpha-helical antimicrobial peptides, is the most famous family of AMPs from insects, and it can be found in guppy silkworm, bees, Drosophila. Cecropin A shows activity against different inflammatory diseases and cancers. Furthermore, it demonstrated in vitro and in vivo efficacy against Staphylococcus aureus in comparison with Vancomycin and Magainin II in murine models [57].
Tachyplesin III
Tachyplesin III is a peptide that shows activity against P. aeruginosa MDR when associated in vitro with Colistin and beta lactams [58]. In a murine model with P. aeruginosa MDR sepsi tachyplesin III, Colistin, and Imipenem were evaluated[59]. Combination therapy of Tachyplesin III and Imipenem resulted in the highest significantly lower levels of bacteremia than groups with a single agent, or other combinations. Furthermore, the efficacy was evaluated considering plasma levels of lipopolysaccharide, tumour necrosis factor alpha, and interleukin-6, showing the best reduction in Tachyplesin III and Imipenem group. From this study we can highlight that combination therapy with AMPs and traditional antibiotics may be a new therapeutic option. Similarly, mice treated with Tachyplesin III in combination with Piperacillin/tazobactam had a reduction of P.aeruginosa growth by 1000-fold compared to monotherapy [60].
Finally, Tachyplesin III and Clarithromycin were evaluated against Escherichia coli sepsis in a murine model. Tachyplesin III, administered 1 mg/kg intraperitoneally alone, obtained a greater reduction in bacterial growth and a lower level of endotoxin and TNF-alpha plasma concentration compared to the control and Clarithromycin alone. The association of Tachyplesin III and Clarithromycin was the most effective for all considered parameters [61].
SYNTHETIC ANTIMICROBIAL PEPTIDES
6.1 Pal-Lys-NH2 and Pal-Lys-Lys
Pal-Lys-NH2 and Pal-Lys-Lys are short bactericidal lipopeptides with a strong antimicrobial activity against a wide spectrum of Gram-positive cocci [62]. Lipopeptides are monomeric in solution while longer ones form oligomers and this feature can potentiate bacterial killing.
A study investigated their action alone or combined to Vancomycin in preventing prosthesis biofilm in a subcutaneous rat pouch model of staphylococcal vascular graft infection. The results of this study showed that Vancomycin (from 6.94 log to 3.65 log CFU/mL) and lipopeptides (from 6.94 log to 3.87 log CFU/mL for Pal-Lys-Lys NH2 and from 6.94 log to 4.080 log CFU/mL for Pal-Lys-Lys) when used alone had similar significant bacterial growth inhibition. All combinations showed efficacies significantly higher than that of each single compound. The combination of Vancomycin with Pal-Lys-Lys-NH2 had the strongest efficacy (from 6.94 log to 1 log CFU/mL). The in vitro study globally confirms the in vivo one [63].
Daptomycin
Daptomycin is a lipopetide with a rapid bactericidal activity against staphylococci. The efficacy of this peptide was assessed in different studies to evaluate its action on biofilm, wound infections and enterococcal infections [64].
Cirioni et al. investigated the efficacy of Daptomycin and Rifampicin either alone or in combination in preventing vascular graft biofilm formation in a rat pouch model of Staphylococcal infection. The results of this study showed that when tested alone both Rifampicin and Daptomycin have good efficacies without any toxicity and drug-related adverse effects.
Their combination showed higher efficacies than that of each single compound, this effect could be due to the Daptomycin mechanism of action, in fact, it binds and open channels that can allow specific entry of Rifampicin [65].
In vivo efficacy of Daptomycin in the treatment of burn wound infections caused by MRSA was evaluated, in comparison to Teicoplanin, assessing the wound healing process through immunohistochemical and morphological analysis. The results showed greater antimicrobial activity of Daptomycin compared with Teicoplanin. In addition, there was better overall healing with significantly higher epithelialization and collagen scores than the other groups; these results were also confirmed by immunohistochemical data on EGFR and FGF-2[66]. Moreover, in a previous studies, Daptomycin showed synergy in its effect against MRSA when combined with other antibiotics such as tigecycline [67, 68].
Teicoplanin
Teicoplanin is a glycopeptide antibiotic structurally related to Vancomycin. It is produced from Actinoplanes teichomyceticus. Its spectrum of activity includes Gram positive bacteria [69].
Giacometti et al. investigated the efficacy of Levoxacin, Cefazolin, and Teicoplanin in preventing vascular prosthetic graft infection induced by methicillin-susceptible and methicillin-resistant Staphylococcus epidermidis. The efficacy of Levoxacin against the methicillin-susceptible strain did not differ from that of Cefazolin or Teicoplanin, but Levoxacin (from 106 to 103 CFU/mL) showed slightly less efficacy than Teicoplanin (from 106 to 102 CFU/mL) against the methicillin-resistant strain. The Levoxacin-Rifampin combination proved to be similarly effective to the Rifampin-Teicoplanin combination and more effective than the Rifampin-Cefazolin combination against both strains. The most useful combination for the prevention of late-appearing vascular graft infections caused by S. epidermidis is Rifampicin-Levoxacin (no infection detected), because it takes advantage of the good antistaphylococcal activity of both drugs [70].
Ghiselli et al. wanted to study the efficacy of Teicoplanin for the treatment of wound infection with Staphylococcus aureus in a mouse model, comparing topical vs systemic treatment. Results showed a strong inhibition of bacterial growth in all groups treated with intraperitoneal Teicoplanin. However, the highest inhibition was obtained with Teicoplanin-soaked Allevyn and intraperitoneal Teicoplanin. Histologic examination showed that each treatment improved wound repair, but the best results were obtained with Teicoplanin-soaked Allevyn, associated with wound remodeling similar to that in uninfected mice, assessing microvessel density, VEGF expression, and granulation tissue formation [71]. VEGF is an endothelial cell-specific mitogen and an angiogenic inducer active in growth and development, in wound healing, and in various pathologic conditions [72-75].
Dalbavancin
Dalbavancin is a semi-synthetic lipoglycopeptide that exhibits activity against Gram-positive bacteria. It has been approved for treating acute bacterial skin and skin structure infections (ABSSSI) [76]. Simonetti et al. conducted a study to investigate the effect of Dalbavancin on wound healing compared to Vancomycin. They also determined the potential involvement of MMP-1, MMP-9, EGFR, and VEGF in Dalbavancin’s therapeutic mechanisms. It was established a mouse model of MRSA skin infection, and mice were treated daily with Vancomycin or weekly with Dalbavancin on days 1 and 8. Both drugs effectively reduced the bacterial load (8.71×105 ± 9.02×105 CFU/mL for Dalbavancin vs. 8.04×106 ± 7.96×106 CFU/mL for Vancomycin). Wounds treated with Dalbavancin exhibited well-organized granulation tissue with several blood vessels, though slightly fewer than those in the uninfected group. Immunohistochemical staining showed elevated EGFR and VEGF expression in both treated groups (more pronounced in Dalbavancin-treated mice), decreased MMP-1 and MMP-9 levels in uninfected tissue, and in both treated tissues compared to untreated infected wounds [77].
Laur-CKK-NH2
Laur-CKK-NH2 is a new compound synthetized by conjunction of lauric acid with the amino acids cysteine-lysine-lysine.
An experimental study assessed both the in vitro and in vivo interaction between the Laur-CKK-NH2 lipopeptide and Daptomycin using two Enterococcus faecalis strains with different patterns of susceptibilities. The in vitro studies demonstrated that the Laur-CKK-NH2 dimer prevents the emergence of Daptomycin resistance. The in vivo study showed the presence of a synergistic effect between the Laur-CKK-NH2 dimer and Daptomycin exhibiting the highest efficacy in measuring lethality and bacteremia. In conclusion this AMP could be a potential therapeutic option for severe enterococcal infections [78].
Pexiganan
Pexiganan is a 22-amino acid synthetic lysine-rich peptide, derived from magainin. In a mouse model with A. baumannii sepsis, it was reported effective. Considering the bacterial count, Pexiganan plus Colistin association resulted in the best reduction, with 90% of survival rate [79]. For this reason, Pexiganan may also be a future option to overcome MDR Gram-negative. Furthermore, Pexiganan showed synergic activity with tigecycline against P. aeruginosa in a mouse model, revealing a possible antibiotic that would not normally be effective against Gram-negatives. In fact, Pseudomonas aeruginosa is not reliably inhibited by tigecycline [80]. Considering P. aeruginosa urethral stents infections in a murine model, Pexiganan and Imipenem at sub-MIC concentration resulted in a marked reduction of adhesion and biofilm expression compared to untreated controls. The average reduction was of 34+8% and 27+4%, respectively [81].
AMPs derived from microorganisms
Some famous peptides obtained from microorganisms are nisin and Gramicidin from Lactococcus lactis, Bacillus subtilis, and Bacillus brevis [82].
Interestingly, there are some neoplastic conditions, in particular cutaneous lymphomas, in which it appears to be a dysregulation of the production of cutaneous AMPs. In these patients the risk of developing infections, even lethal, is higher [83-85]. However, some AMPs such as nisin showed an immunomodulatory action, enhancing the cytotoxic and apoptotic action of Rituximab in Burkitt’s lymphoma [86].
Due to the high price of chemical synthesis of AMPs, the biological expression has attracted the increase of attention. Specific yeast species like Pichia pastoris, Saccharomyces cerevisiae, and bacteria like Escherichia coli, B. subtilis, and plants have been used for expression systems [87], but it should be noticed that because of the toxicity, proteolytic degradation, and purification, AMPs are difficult to be produced in E. coli, which is necessary to take advantage of fusion tags [88].
Colistin
Colistin, derived from Bacillus Colistinus, allows the permeabilization of the A. baumannii outer membrane, facilitating the action of large size molecule as glycopeptide and lipopeptide, and achieving an improvement of the patients with A. baumannii MDR severe infections [89]. In fact, the bacterial count was 6.7×104 ± 1.1×104 Colistin alone, 5.0×109 ± 1.6×109 Daptomycin alone, 7.3×109±1.8×109 Teicoplanin alone, 2.9×102±0.4×102 Colistin + Daptomycin, and 3.1×102±0.2×102 Colistin + Teicoplanin, respectively [90]. Furthermore, the association with Colistin and Pexiganan was more effective against Gram negative bacterial infection than single treatment [91]. This is an example of how an antibiotic not normally effective against Gram-negative bacteria can become effective with the help of an antimicrobial peptide [92].
CONCLUSIONS
In this narrative review of our research experience, as already anticipated in the study, we decided to focus more specifically on the sources of AMPs, with the aim of assessing which might be the best sources from which to draw new AMPs [93]. In conclusion, our research experience shows that the richest source of AMPs are amphibians. However, the studies done are mainly in vitro or in animal models, making further human studies necessary to assess the efficacy and safety of these molecules. A further limitation is the cost of some AMPs, although the possibility of selecting some extremely promising ones could encourage the search for manufacturing solutions to reduce their cost and make them more easily available.
However, AMPs may be a new therapeutic option for infections sustained by multi-resistant micro-organisms and for overcoming the mechanisms of resistance to antibiotics currently used.
In particular, combining AMPs with antibiotics, including those with limited antimicrobial activity due to aantimicrobial resistance, has often shown a synergistic effect, increasing or restoring their efficacy. The possibility of using manageable and relatively safe antibiotics again is crucial, considering the widespread increase in bacterial resistance in hospitals and the community.
In the literature there are cases of AMP used on humans [94-98]. Specifically, with regard to Gram-negative infections, the most promising molecules are IB-367 and LL-37, showing a synergistic action with traditional antibiotic therapy. Regarding IB-367 (Iseganan), a phase I clinical study on prophylaxis of oral mucositis post cytotoxic therapy showed that oral topical IB-367 was not absorbed and was well tolerated. As early as one hour after administration of 9 mg of IB-367, the density of Gram-positive flora was reduced 1000-fold, and Gram-negative bacteria had also been significantly reduced [94]. Some phase III studies have also been conducted. In particular, Iseganan was studied in patients receiving stomatotoxic chemotherapy, underlining that topical Iseganan HCl significantly reduces the total oral aerobic bacterial, streptococcal, and yeast load [95]. In addition, Elad et al. conducted a multicentre, double-blind versus placebo study on 2025 patients undergoing chemotherapy, mainly myeloablative. Iseganan HCl 9 mg/3 ml was used topically 6 times daily for mucosal prevention. After one month, there was a statistically significant reduction in bacterial load and an unchanged MIC, confirming its efficacy in this type of patient [96]. Specific studies on MDR microorganisms are currently lacking, but the data reported on humans are promising. Furthermore, LL-37 showed in humans its action on wound marginalization of venous leg ulcers, proving to be safe and thus making it an excellent candidate for further studies [97-99].
Despite a plethora of research on AMPs and their application as potential treatment on infectious diseases, this area needs further exploration. There is evidence that the characteristics of AMPs can seriously improve through structural chemical modifications and different delivery systems to become alternatives drugs to conventional antibiotics.
Funding sources statement
The work was not supported by founding sources.
Conflict of interest disclosures
The authors have no competing interests to declare.
REFERENCES
[1] Ayukekbong JA, Ntemgwa M, Atabe AN. The threat of antimicrobial resistance in developing countries: causes and control strategies. Antimicrob Resist Infect Control. 2017; 6, 47.
[2] Wang G, Li X, Wang Z, APD3. The antimicrobial peptide database as a tool for research and education. Nucleic Acids Res. 2016; 44, D1087-D1093.
[3] Simonetti O, Silvestri C, Arzeni D, et al. In vitro activity of the protegrin IB-367 alone and in combination compared with conventional antifungal agents against dermatophytes. Mycoses. 2014; 57 (4), 233-239.
[4] Kamysz E, Simonetti O, Cirioni O, et al. In vitro activity of the lipopeptide PAL-Lys-Lys-NH2, alone and in combination with antifungal agents, against clinical isolates of Candida spp. Peptides. 2011; 32 (1), 99-103.
[5] Barchiesi F, Silvestri C, Arzeni D, et al. In vitro susceptibility of dermatophytes to conventional and alternative antifungal agents. Med Mycol. 2009; 47 (3), 321-326.
[6] Simonetti O, Arzeni D, Ganzetti G, et al. In vitro activity of the lipopeptide derivative (Pal-Lys-Lys-NH), alone and in combination with antifungal agents, against clinical isolates of dermatophytes. Br J Dermatol. 2009; 161 (2), 249-252.
[7] Simonetti O, Ganzetti G, Arzeni D, et al. In vitro activity of Tachyplesin III alone and in combination with terbinafine against clinical isolates of dermatophytes. Peptides. 2009; 30 (10), 1794-1797.
[8] Fioriti S, Cirioni O, Simonetti O, et al. In Vitro Activity of Novel Lipopeptides against Triazole-Resistant Aspergillus fumigatus. J Fungi (Basel). 2022; 18 (8), 872.
[9] Lazzaro BP, Zasloff M, Rolff J. Antimicrobial peptides: Application informed by evolution. Science. 2020; 368 (6490), eaau5480.
[10] Reddy KVR, Yedery RD, Aranha C. Antimicrobial peptides: premises and promises. Int J Antimicrob Agents. 2004; 24, 536-547.
[11] Wang G. Human antimicrobial peptides and proteins. Pharmaceuticals. 2014; 7, 545-594.
[12] Benincasa M, Skerlavaj B, Gennaro R, Pellegrini A, Zanetti M. In vitro and in vivo antimicrobial activity of two alpha-helical cathelicidin peptides and of their synthetic analogs. Peptides. 2003; 24,1723-1731.
[13] Skerlavaj B, Gennaro R, Bagella L, et al. Biological characterization of two novel cathelicidin-derived peptides and identification of structural requirements for their antimicrobial and cell lytic activities. J Biol Chem. 1996; 271, 28375-28381.
[14] Cirioni O, Giacometti A, Ghiselli R, et al. Pre-treatment of central venous catheters with the cathelicidin BMAP-28 enhances the efficacy of antistaphylococcal agents in the treatment of experimental catheter-related infection. Peptides. 2006; 27, 2104-2110.
[15] Orlando F, Ghiselli R, Cirioni O. et al. BMAP-28 improves the efficacy of Vancomycin in rat models of Gram-positive cocci ureteral stent infection. Peptides. 2008; 29, 1118-1123.
[16] Giacometti A, Cirioni O, Ghiselli R, et al. The antimicrobial peptide BMAP-28 reduces lethality in mouse models of staphylococcal sepsis. Crit Care Med. 2004; 32, 2485-2490.
[17] Chen J, Falla TJ, Liu H, et al. Development of protegrins for the treatment and prevention of oral mucositis: Structure-activity relationships of synthetic protegrin analogues. Biopolymers. 2000; 55, 88-98. doi: 10.1002/1097-0282(2000)55:1<88::AID-BIP80>3.0.CO;2-K.
[18] Ghiselli R, Giacometti A, Cirioni O, et al. Pretreatment with the protegrin IB-367 affects Gram-positive biofilm and enhances the therapeutic efficacy of linezolid in animal models of central venous catheter infection. JPEN J Parenter Enteral Nutr. 2007; 31, 463-448. doi: 10.1177/0148607107031006463.
[19] Cirioni O, Silvestri C, Pierpaoli E, et al. IB-367 pre-treatment improves the in vivo efficacy of Teicoplanin and Daptomycin in an animal model of wounds infected with meticillin-resistant Staphylococcus aureus. J Med Microbiol. 2013; 62, 1552-1558. doi: 10.1099/jmm.0.057414-0.
[20] Simonetti O, Cirioni O, Ghiselli R, et al. In vitro activity and in vivo animal model efficacy of IB-367 alone and in combination with Imipenem and Colistin against Gram-negative bacteria. Peptides. 2014; 55, 17-22. doi: 10.1016/j.peptides.2014.01.029.
[21] Bucki R, Leszczyńska K, Namiot A, Sokołowski W. Cathelicidin LL-37: A multitask antimicrobial peptide. Arch Immunol Ther Exp. 2010; 58, 15-25. doi: 10.1007/s00005-009-0057-2.
[22] Nagaoka I, Tamura H, Reich J. Therapeutic potential of Cathelicidin Peptide LL-37, an antimicrobial agent, in a murine sepsis model. Int J Mol Sci. 2020; 21, 5973. doi: 10.3390/ijms21175973.
[23] Simonetti O, Cirioni O, Goteri G, et al. Efficacy of Cathelicidin LL-37 in an MRSA wound infection mouse model. Antibiotics. 2021; 10, 1210. doi: 10.3390/antibiotics10101210.
[24] Cirioni O, Giacometti A, Ghiselli R, et al. LL-37 protects rats against lethal sepsis caused by Gram-negative bacteria. Antimicrob Agents Chemother. 2006; 50, 1672-1679. doi: 10.1128/AAC.50.5.1672-1679.2006.
[25] Morroni G, Sante LD, Simonetti O, et al. Synergistic effect of antimicrobial peptide LL-37 and Colistin combination against multidrug-resistant Escherichia coli isolates. Future Microbiol. 2021; 16, 221-227. doi: 10.2217/fmb-2020-0204.
[26] Cirioni O, Giacometti A, Silvestri C, et al. In vitro activities of tritrpticin alone and in combination with other antimicrobial agents against Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2006; 50, 3923-3925. doi: 10.1128/AAC.00652-06.
[27] Simonetti O, Cirioni O, Ghiselli R, et al. In vitro activity and in vivo animal model efficacy of IB-367 alone and in combination with Imipenem and Colistin against Gram-negative bacteria. Peptides. 2014; 55, 17-22. doi: 10.1016/j.peptides.2014.01.029.
[28] Cirioni O, Simonetti O, Pierpaoli E, et al. Enhanced efficacy of combinations of Pexiganan with Colistin versus Acinetobacter baumannii in experimental sepsis. Shock. 2016; 46, 219-25. doi: 10.1097/SHK.0000000000000584.
[29] Morroni G, Simonetti O, Brenciani A, et al. In vitro activity of Protegrin-1, alone and in combination with clinically useful antibiotics, against Acinetobacter baumannii strains isolated from surgical wounds. Med Microbiol Immunol. 2019; 208, 877-883. doi: 10.1007/s00430-019-00624-7.
[30] Conlon JM, Mechkarska M. Host-defense peptides with therapeutic potential from skin secretions of frogs from the family pipidae. Pharmaceuticals (Basel). 2014; 7(1), 58-77. doi: 10.3390/ph7010058.
[31] Doyle J, Brinkworth CS, Wegener K.L., et al. nNOS inhibition, antimicrobial and anticancer activity of the amphibian skin peptide, Citropin 1.1 and synthetic modifications. The solution structure of a modified Citropin 1.1. Eur J Biochem. 2003; 270, 1141-1153. doi: 10.1046/j.1432-1033.2003.03462.x
[32] Cirioni O, Giacometti A, Ghiselli R, et al. Citropin 1.1-treated central venous catheters improve the efficacy of hydrophobic antibiotics in the treatment of experimental staphylococcal catheter-related infection. Peptides. 2006; 27, 1210-1216.
[33] Giacometti A, Cirioni O, Kamysz W, et al. In vitro activity of Citropin 1.1 alone and in combination with clinically used antimicrobial agents against Rhodococcus equi. J Antimicrob Chemother. 2005; 56, 410-412. doi: 10.1093/jac/dki236.
[34] Cirioni O, Silvestri C, Pierpaoli E, et al. IB-367 pre-treatment improves the in vivo efficacy of Teicoplanin and Daptomycin in an animal model of wounds infected with meticillin-resistant Staphylococcus aureus. J Med Microbiol. 2013; 62, 1552-1558. doi: 10.1099/jmm.0.057414-0.
[35] Giacometti A, Cirioni O, Del Prete MS, et al. Combination studies between polycationic peptides and clinically used antibiotics against Gram-positive and Gram-negative bacteria. Peptides. 2000; 21, 1155-60. doi: 10.1016/S0196-9781(00)00254-0.
[36] Simonetti O, Lucarini G, Campanati A, et al. VEGF, survivin and NOS overexpression in psoriatic skin: critical role of nitric oxide syntheses. J Dermatol Sci. 2009; 54 (3), 205-208. doi: 10.1016/j.jdermsci.2008.12.012.
[37] Giacometti A, Cirioni O, Kamysz W, et al. In vitro activity and killing effect of Citropin 1.1 against Gram-positive pathogens causing skin and soft tissue infections. Antimicrob Agents Chemother. 2005; 49, 2507-2509. doi: 10.1128/AAC.49.6.2507-2509.2005.
[38] Giacometti A; Cirioni O; Kamysz W, et al. In vitro activity and killing effect of uperin 3.6 against Gram-positive cocci isolated from immunocompromised patients. Antimicrob Agents Chemother. 2005; 49, 3933-3936.
[39] Ghiselli R, Giacometti A, Cirioni O, et al. Temporin A as a prophylactic agent against methicillin sodium-susceptible and methicillin sodium-resistant Staphylococcus epidermidis vascular graft infection. J Vasc Surg. 2002; 36, 1027-1030. doi: 10.1067/mva.2002.127530.
[40] Harjunpää I, Kuusela P, Smoluch MT, et al. Comparison of synthesis and antibacterial activity of Temporin A. FEBS Lett. 1999; 449, 187-90. doi: 10.1016/S0014-5793(99)00406-8.
[41] Cirioni O, Giacometti A, Ghiselli R, et al. Prophylactic efficacy of topical Temporin A and RNAIII-inhibiting peptide in a subcutaneous rat Pouch model of graft infection attributable to staphylococci with intermediate resistance to glycopeptides. Circulation. 2003; 108, 767-771. doi: 10.1161/01.CIR.0000083717.85060.16
[42] Balaban N, Gov Y, Bitler A, Boelaert J. Prevention of Staphylococcus aureus biofilm on dialysis catheters and adherence to human cells. Kidney Int. 2003; 63, 340-345. doi: 10.1046/j.1523-1755.2003.00733.x.
[43] Giacometti A, Cirioni O, Ghiselli R, et al. Temporin A soaking in combination with intraperitoneal linezolid prevents vascular graft infection in a subcutaneous rat pouch model of infection with Staphylococcus epidermidis with intermediate resistance to glycopeptides. Antimicrob Agents Chemother. 2004; 48, 3162-3164. doi: 10.1128/AAC.48.8.3162-3164.2004.
[44] Cirioni O, Giacometti A, Ghiselli R, et al. Temporin A alone and in combination with Imipenem reduces lethality in a mouse model of staphylococcal sepsis. J Infect Dis. 2005; 192, 1613-1620. doi: 10.1086/496888.
[45] Simonetti O, Cirioni O, Goteri G, et al. Temporin A is effective in MRSA-infected wounds through bactericidal activity and acceleration of wound repair in a murine model. Peptides. 2008; 29, 520-528. doi: 10.1016/j.peptides.2007.12.011.
[46] Giacometti A, Cirioni O, Barchiesi F, Del Prete MS, Scalise G. Antimicrobial activity of polycationic peptides. Peptides. 1999; 20 (11), 1265-73. doi: 10.1016/s0196-9781(99)00131-x.
[47] Giacometti A, Cirioni O, Ghiselli R, et al. Efficacy of polycationic peptides in preventing vascular graft infection due to Staphylococcus epidermidis. J Antimicrob Chemother. 2000; 46, 751-756. doi: 10.1093/jac/46.5.751.
[48] Giacometti A, Cirioni O, Ghiselli R, et al. Polycationic peptides as prophylactic agents against methicillin-susceptible or methicillin-resistant Staphylococcus epidermidis vascular graft infection. Antimicrob Agents Chemother. 2000; 44, 3306-3309. doi: 10.1128/AAC.44.12.3306-3309.2000.
[49] Batista CV, Scaloni A, Rigden DJ, et al. A novel heterodimeric antimicrobial peptide from the tree-frog Phyllomedusa distincta. FEBS Lett. 2001; 494, 85-89. doi: 10.1016/S0014-5793(01)02324-9.
[50] Giacometti A, Cirioni O, Ghiselli R, et al. Distinctin improves the efficacies of glycopeptides and betalactams against staphylococcal biofilm in an experimental model of central venous catheter infection. J Biomed Mater Res A. 2007; 81, 233-239. doi: 10.1002/jbm.a.30982.
[51] Cirioni O, Ghiselli R, Orlando F, et al. Efficacy of the amphibian peptide Distinctin in a neutropenic mouse model of staphylococcal sepsis. Crit Care Med. 2008; 36, 2629-2633. doi: 10.1097/CCM.0b013e318184430d.
[52] Simonetti O, Cirioni O, Ghiselli R, et al. Antimicrobial properties of Distinctin in an experimental model of MRSA-infected wounds. Eur J Clin Microbiol Infect Dis. 2012; 31, 3047-3055. doi: 10.1007/s10096-012-1663-1.
[53] Giacometti A, Cirioni O, Riva A, et al. In vitro activity of aurein 1.2 alone and in combination with antibiotics against Gram-positive nosocomial cocci. Antimicrob Agents Chemother. 2007; 51, 1494-1496. doi: 10.1128/AAC.00666-06.
[55] Cirioni O, Silvestri C, Ghiselli R, et al. Experimental study on the efficacy of combination of alpha-helical antimicrobial peptides and Vancomycin against Staphylococcus aureus with intermediate resistance to glycopeptides. Peptides. 2006; 27, 2600-2606. doi: 10.1016/j.peptides.2006.05.004.
[56] Cirioni O, Giacometti A, Kamysz W, et al. In vitro activities of tachyplesin III against Pseudomonas aeruginosa. Peptides. 2007; 28, 747-751. doi: 10.1016/j.peptides.2007.01.001.
[57] Vilcinskas A. Evolutionary plasticity of insect immunity. J Insect Physiol. 2013; 59, 123-129.
[58] Cirioni O, Silvestri C, Ghiselli R, et al. Experimental study on the efficacy of combination of alpha-helical antimicrobial peptides and Vancomycin against Staphylococcus aureus with intermediate resistance to glycopeptides. Peptides. 2006; 27, 2600-2606. doi: 10.1016/j.peptides.2006.05.004.
[59] Cirioni O, Ghiselli R, Silvestri C, et al. Efficacy of tachyplesin III, Colistin, and Imipenem against a multiresistant Pseudomonas aeruginosa strain. Antimicrob Agents Chemother. 2007; 51, 2005-2010. doi: 10.1128/AAC.01576-06.
[60] Minardi D, Ghiselli R, Cirioni O, et al. The antimicrobial peptide tachyplesin III coated alone and in combination with intraperitoneal piperacillin-tazobactam prevents ureteral stent Pseudomonas infection in a rat subcutaneous pouch model. Peptides. 2007; 28, 2293-2298. doi: 10.1016/j.peptides.2007.10.001.
[61] Cirioni O, Ghiselli R, Silvestri C, et al. Efficacy of the combination of tachyplesin III and Clarithromycin in rat models of Escherichia coli sepsis. Antimicrob Agents Chemother. 2008; 52, 4351-4355. doi: 10.1128/AAC.00086-08.
[62] Cirioni O, Ghiselli R, Tomasinsig L, et al. Efficacy of LL-37 and granulocyte colony-stimulating factor in a neutropenic murine sepsis due to Pseudomonas aeruginosa. Shock. 2008; 30, 443-448. doi: 10.1097/SHK.0b013e31816d2269.
[63] Kamysz W, Silvestri C, Cirioni O, et al. In vitro activities of the lipopeptides palmitoyl (Pal)-Lys-Lys-NH(2) and Pal-Lys-Lys alone and in combination with antimicrobial agents against multiresistant Gram-positive cocci. Antimicrob Agents Chemother. 2007; 51, 354-358. doi: 10.1128/AAC.00344-06.
[64] Cirioni O, Giacometti A, Ghiselli R, et al. The lipopeptides Pal-Lys-Lys-NH(2) and Pal-Lys-Lys soaking alone and in combination with intraperitoneal Vancomycin prevent vascular graft biofilm in a subcutaneous rat pouch model of staphylococcal infection. Peptides. 2007; 28, 1299-1303. doi: 10.1016/j.peptides.2007.03.017.
[65] Rizzetto G, Molinelli E, Radi G, et al. Role of Daptomycin in cutaneous wound healing: a narrative review. Antibiotics. 2022; 11, 944. doi: 10.3390/antibiotics11070944.
[66] Cirioni O, Mocchegiani F, Ghiselli R, et al. Daptomycin and Rifampin alone and in combination prevent vascular graft biofilm formation and emergence of antibiotic resistance in a subcutaneous rat pouch model of staphylococcal infection. Eur J Vasc Endovasc Surg. 2010; 40, 817-822. doi: 10.1016/j.ejvs.2010.08.009.
[67] Simonetti O, Lucarini G, Orlando F, et al. Role of Daptomycin on burn wound healing in an animal Methicillin-Resistant Staphylococcus aureus infection model. Antimicrob Agents Chemother. 2017; 61, 606-617. doi: 10.1128/AAC.00606-17.
[68] Silvestri C, Cirioni O, Arzeni D, et al. In vitro activity and in vivo efficacy of tigecycline alone and in combination with Daptomycin and Rifampin against Gram-positive cocci isolated from surgical wound infection. Eur J Clin Microbiol Infect Dis. 2012; 31, 1759-1564. doi: 10.1007/s10096-011-1498-1.
[69] Giacometti A, Cirioni O, Kamysz W, et al. In vitro activity and killing effect of Temporin A on nosocomial isolates of Enterococcus faecalis and interactions with clinically used antibiotics. J. Antimicrob. Chemother. 2005; 55, 272-274. doi: 10.1093/jac/dkh54.
[70] Fanos V, Kacet N, Mosconi G. A review of Teicoplanin in the treatment of serious neonatal infections. Eur J Pediatr. 1997; 156 (6), 423-427. doi: 10.1007/s004310050629. PMID: 9208233.
[71] Giacometti A, Cirioni O, Ghiselli R, et al. Vascular graft infection by Staphylococcus epidermidis: Efficacy of various perioperative prophylaxis protocols in an animal model. Infez Med. 2001; 9, 13-18.
[72] Ghiselli R, Cirioni O, Giacometti A, et al. Comparative efficacy of topical versus systemic Teicoplanin in experimental model of wound infections. J Surg Res. 2008; 144, 74-81. doi: 10.1016/j.jss.2007.02.051.
[73] Ferrara N, Gerber HP. The role of vascular endothelial growth factor in angiogenesis. Acta Haematol. 2001; 106 (4), 148-156. doi: 10.1159/000046610.
[74] Simonetti O, Lucarini G, Rubini C, et al. Clinical and prognostic significance of survivin, AKT and VEGF in primary mucosal oral melanoma. Anticancer Res. 2015; 35 (4), 2113-2120.
[75] Simonetti O, Goteri G, Lucarini G, et al. In melanoma changes of immature and mature dendritic cell expression correlate with tumor thickness: an immunohistochemical study. Int J Immunopathol Pharmacol. 2007; 20 (2), 325-333. doi: 10.1177/039463200702000212.
[76] Simonetti O, Lucarini G, Rubini C, et al. Microvessel density and VEGF, HIF-1α expression in primary oral melanoma: correlation with prognosis. Oral Dis. 2013; 19 (6), 620-627. doi: 10.1111/odi.12048.
[77] Simonetti O, Rizzetto G, Molinelli E, Cirioni O, Offidani A. Review: a safety profile of Dalbavancin for on- and off-label utilization. Ther Clin Risk Manag. 2021; 17, 223-232. doi: 10.2147/TCRM.S271445.
[78] Simonetti O, Lucarini G, Morroni G, et al. New evidence and insights on Dalbavancin and wound healing in a mouse model of skin infection. Antimicrob Agents Chemother. 2020; 64, 2062-2019. doi: 10.1128/AAC.02062-19.
[79] Cirioni O, Kamysz E, Ghiselli R, et al. Lipopeptide Laur-CKK-NH2 dimer preserves Daptomycin susceptibility and enhances its activity against Enterococcus faecalis. J Antimicrob Chemother. 2011; 66, 859-862. doi: 10.1093/jac/dkr001.
[80] Cirioni O, Simonetti O, Morroni G, et al. Efficacy of Pexiganan combination with tigecycline in a mouse model of Pseudomonas aeruginosa sepsis. Curr Top Med Chem. 2018; 18, 2127-2132. doi: 10.2174/1568026619666181219123431.
[81] Cirioni O, Silvestri C, Ghiselli R. et al. In vitro and in vivo effects of sub-MICs of Pexiganan and Imipenem on Pseudomonas aeruginosa adhesion and biofilm development. Infez Med. 2013; 21, 287-295.
[82] Cirioni O, Silvestri C, Ghiselli R, et al. Protective effects of the combination of alpha-helical antimicrobial peptides and Rifampicin in three rat models of Pseudomonas aeruginosa infection. J Antimicrob Chemother. 2008; 62, 1332-1338. doi: 10.1093/jac/dkn393.
[83] Cao J, de la Fuente-Nunez C, Ou RW, et al. Yeast-based synthetic biology platform for antimicrobial peptide production. ACS Synthet Biol. 2018; 7, 896-902.
[84] Wehkamp U, Jost M, Wehkamp K, Harder J. Dysregulated expression of antimicrobial peptides in skin lesions of patients with cutaneous T-cell lymphoma. Acta Derm Venereol. 2020; 7, 100. doi: 10.2340/00015555-3372.
[85] Fanok MH, Sun A, Fogli LK, et al.. Role of dysregulated cytokine signaling and bacterial triggers in the pathogenesis of cutaneous T-cell lymphoma. J Invest Dermatol. 2018; 138, 1116-1125.
[86] Goteri G, Ranaldi R, Simonetti O, et al. Clinico-pathological features of primary cutaneous B-cell lymphomas from an academic regional hospital in central Italy: no evidence of Borrelia burgdorferi association. Leuk Lymphoma. 2007; 48 (11), 2184-2188. doi: 10.1080/10428190701618250.
[87] Mohammadi P, Zangeneh M, Mohammadi-Motlagh HR, Khademi F. The antimicrobial peptide, nisin, synergistically enhances the cytotoxic and apoptotic effects of Rituximab treatment on Human Burkitt’s lymphoma cell lines. Rep Biochem Mol Biol. 2020; 9 (3), 250-256. doi: 10.29252/rbmb.9.3.250.
[88] Parachin NS, Mulder KC, Viana AAB, Dias SC, Franco OL. Expression systems for heterologous production of antimicrobial peptides. Peptides. 2012; 38, 446-456.
[89] Yu H, Li H, Gao D, Gao C, Qi Q. Secretory production of antimicrobial peptides in Escherichia coli using the catalytic domain of a cellulase as fusion partner. J Biotechnol. 2015; 214, 77-82.
[90] Falagas ME, Rafailidis PI. Re-emergence of Colistin in today’s world of multidrug-resistant organisms: personal perspectives. Expert Opin Investig Drugs. 2008; 17 (7), 973-981. doi: 10.1517/13543784.17.7.973.
[91] Cirioni O, Simonetti O, Pierpaoli E, et al. Colistin enhances therapeutic efficacy of Daptomycin or Teicoplanin in a murine model of multiresistant Acinetobacter baumannii sepsis. Diagn Microbiol Infect Dis. 2016; 86 (4), 392-398. doi: 10.1016/j.diagmicrobio.2016.09.010.
[92] Cirioni O, Simonetti O, Pierpaoli E, et al. Enhanced efficacy of combinations of Pexiganan with Colistin versus Acinetobacter baumannii in experimental sepsis. Shock. 2016; 46, 219-225. doi: 10.1097/SHK.0000000000000584.
[93] Randall CP, Mariner KR, Chopra I, et al. The target of Daptomycin is absent from Escherichia coli and other gram-negative pathogens. Antimicrob Agents Chemother. 2013; 57(1), 637-639. doi:10.1128/AAC.02005-12.
[94] Rizzetto G, Gambini D, Maurizi A, et al. Our Experience over 20 years: antimicrobial peptides against Gram positives, Gram negatives, and fungi. Pharmaceutics. 2022; 15(1), 40. doi: 10.3390/pharmaceutics15010040.
[95] Mosca DA, Hurst MA, So W, Viajar BS, Fujii CA, Falla TJ. IB-367, a protegrin peptide with in vitro and in vivo activities against the microflora associated with oral mucositis. Antimicrob Agents Chemother. 2000; 44(7), 1803-1808. doi: 10.1128/AAC.44.7.1803-1808.2000.
[96] Giles FJ, Rodriguez R, Weisdorf D, et al. A phase III, randomized, double-blind, placebo-controlled, study of iseganan for the reduction of stomatitis in patients receiving stomatotoxic chemotherapy. Leuk Res. 2004; 28(6), 559-565. doi: 10.1016/j.leukres.2003.10.021.
[97] Elad S, Epstein JB, Raber-Durlacher J, Donnelly P, Strahilevitz J. The antimicrobial effect of Iseganan HCl oral solution in patients receiving stomatotoxic chemotherapy: analysis from a multicenter, double-blind, placebo-controlled, randomized, phase III clinical trial. J Oral Pathol Med. 2012; 41 (3), 229-234.
[98] Grönberg A, Mahlapuu M, Ståhle M, Whately-Smith C, Rollman O. Treatment with LL-37 is safe and effective in enhancing healing of hard-to-heal venous leg ulcers: a randomized, placebo-controlled clinical trial. Wound Repair Regen. 2014; 22 (5): 613-621. doi: 10.1111/wrr.12211.
[99] Mahlapuu M, Sidorowicz A, Mikosinski J, et al. Evaluation of LL-37 in healing of hard-to-heal venous leg ulcers: A multicentric prospective randomized placebo-controlled clinical trial. Wound Repair Regen. 2021; 29 (6), 938-950. doi: 10.1111/wrr.12977.