Le Infezioni in Medicina, n. 1, 86-92, 2023
doi: 10.53854/liim-3101-12
CASE REPORTS
Gingival presentation of mucormycosis post infection with COVID-19: series of 3 cases
Shruti Tandon, Anju Rana, Arundeep Kaur, Farrukh Faraz, Kamal Agarwal
Department of Periodontics, Maulana Azad Institute of Dental Sciences, New Delhi, India
Article received 10 October 2022, accepted 25 January 2023
Corresponding author
Anju Rana
E-mail: dranjurana13@gmail.com
SummaRY
Through the evolution of this global pandemic, it became increasingly clear that patients with pre-existing conditions such as diabetes mellitus, chronic kidney diseases (CKD), chronic cardiovascular disease, underlying malignancies, organ transplant recipients, and chronic liver disease are at increased risk for developing severe post COVID-19 complications. One such complication seen after COVID-19 is mucormycosis. Oral mucormycosis is a rare but highly fatal disease. Early diagnosis and timely treatment of rhino-maxillary mucormycosis are necessary to limit the spread of infection, which otherwise can lead to high morbidity and mortality. Dental healthcare practitioners should be familiar with its initial presentation, which if left undiagnosed or ignored can rapidly spread to the orbital and cerebral region leading to death. The case series describes the gingival clinical presentation in its initial stage, relevant basic clinical investigations, treatment, and the importance of an inter-professional team approach in the early management of patients with mucormycosis after post covid-19 infection.
Keywords: Mucormycosis, COVID-19, gingival disease.
INTRODUCTION
Mucormycosis was first described by Paultauf in 1885 [1]. It is a rare condition of the maxillofacial region caused by fungi belonging to the Mucorales order and the Mucoraceae family. Nearly 27 species under Mucorales are associated with human infections. Rhizopus arrhizus is the most common species causing mucormycosis across the globe. This fungus invades the arteries, forms thrombi within the blood vessels that reduce blood supply and cause necrosis of hard as well as soft tissues. It is a rapidly progressive, fatal infection, mainly occurring in immunocompromised patients with conditions such as diabetes mellitus, long-term steroid therapy, neutropenia, haematological malignancies (lymphoma and leukaemia), post-organ transplantation, malnutrition, burns, and immunosuppressive therapy [2].
Early diagnosis of mucormycosis is based on clinical, radiographic, and histopathological findings. Radiographic diagnosis includes plain radiographs, orthopantomogram (OPG), and cone-beam computed tomography (CBCT) view. Imaging technique includes computerized tomography of paranasal sinus (CT PNS view) with contrast and gadolinium-enhanced magnetic resonance imaging MRI. Mucorales are rapid growers (3-7 days) therefore culture is essential for the diagnosis of mucormycosis since it allows identification at genus and species levels. Nasal scrapings/deep nasal swab /pus swab is collected for KOH (potassium hydroxide) mount/ culture and biopsy is collected from denuded/necrosed bone. Histopathologically mucormycosis is confirmed by hematoxylin and eosin stain, Periodic Acid-Schiff (PAS) stain, and Grocott Gomori methamine silver stain. However, negative culture reports are often seen despite of positive histological findings.
Mucormycosis is rarely seen in healthy individuals and reported cases are scarce. But with the Covid-19 pandemic, there has been a surge in cases. India witnessed its second severe wave in March 2021, and as of 7th June 2021, the Indian Ministry of Health recorded 28,252 cases of mucormycosis [3]. The cases during this second wave became so prominent that many state governments had to declare this too, as an epidemic.
No case has been yet reported in the literature with initial gingival findings of mucormycosis in recovered patients of COVID-19 infection. In this paper, three cases of gingival/oral manifestations of mucormycosis are presented which were identified in their initial stage, along with a short review of the literature of gingival/periodontal mucormycosis before the pandemic. Early diagnosis and timely treatment in these cases prevented aggressive radical surgical resection like partial/total maxillectomy, and orbital exenteration.
Case 1
A 36-year-old male reported to the Department of Periodontics with a chief complaint of a small single painful boil on the gum of the left upper posterior tooth since last 10 days. He had a history of COVID-19 infection for which he was hospitalized 1 month back with high-grade fever with cough, generalised weakness and mild difficulty in breathing. Symptomatic supportive treatment was done with antiviral, IV antibiotic, IV steroid and inj. Remdesivir during his hospital admission. He was discharged after 15 days with a negative COVID-19 RT-PCR report and negative KOH (potassium hydroxide) nasal swab report. During discharge, nasal endoscopy and MRI PNS with contrast were normal. The patient had a history of intake of multivitamins, antacids, antibiotics and methylprednisolone 8 mg twice daily for three days after discharge along with seven days of home quarantine.
On clinical examination, an abscess was present in the attached gingiva of tooth # 24 which was tender on percussion. The tooth was examined to rule out fracture and trauma from occlusion. Intraoral periapical radiograph revealed no significant findings. The abscess was curetted and irrigated with povidone-iodine solution [Figure 1A]. After 5 days, the patient developed severe pain in the palatal region and mobility in the anterior left front teeth. On clinical examination, grade 2 mobility along with tenderness on percussion was present in teeth #21, 22, 23, and 24. Painful palatal swelling was also prominent in the left pre-maxillary region. As the symptoms of the patient had aggravated, emergent CBCT was advised avoiding the OPG view.
In CBCT, complete opacification of the left maxillary sinus with air entrapment was evident. Multiple ill-defined osteolytic lesions with irregular borders extending from the distal aspect of the nasopalatine canal with involvement of both the buccal and palatal cortices of the left maxillary region were seen. Destruction of the buccal and palatal alveolus was present from teeth # 21 to 26 giving it a moth-eaten appearance. A distal wall of the left nasal fossa was involved. The floor of the maxillary sinus was breached on the left side in teeth # 24, 25, and 26 regions, but intact on the right side. Perforation of the sinus floor in the 26 and 27 regions was also seen [Figure 1B]. Immediately without any delay, the patient was referred to the medical emergency ward with a suspected diagnosis of mucormycosis post-COVID-19. The pre and post-contrast multiplanar MR imaging of paranasal sinuses and the histopathological report confirmed the diagnosis of mucormycosis involving the left maxillary alveolus, perialveolar tissues and left middle turbinate.
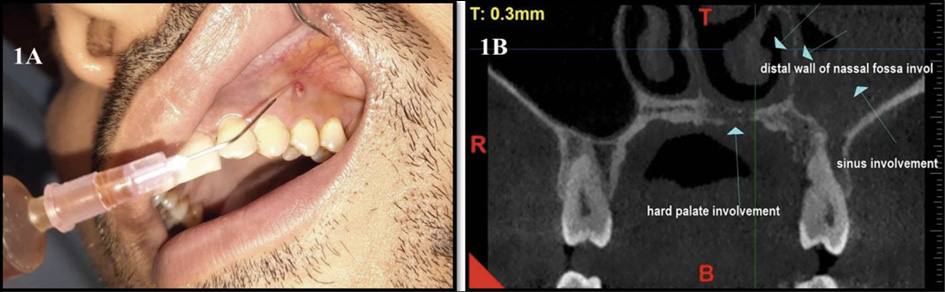
Figure 1A - Initial clinical presentation of mucormycosis with single periodontal abscess being irrigated with betadine; 1B) CBCT revealing hard palate, distal wall of nasal fossa and sinus involvement.
Case 2
A 58-year-old patient with controlled diabetes reported to the Department of Periodontics with a chief complaint of painful multiple boils on upper left teeth along with mobility since past 20 days. The patient had recovered from a COVID-19 infection two months back, for which he had taken steroids. Past dental history revealed extraction of carious 26 one month back. Clinical examination revealed generalized periodontal abscesses and periodontal pockets in relation to maxillary teeth, multiple pseudoclefts, grade III mobility wrt teeth #22 and 25 and grade II mobility of teeth #21,23 and 24 [Figure 2A]. Also, grade I mobility was present in teeth #11, 12, 13, 14, and 15. An unhealed socket was present in relation to tooth #26 and soft non-tender palatal swelling was present on the left maxillary arch. The patient was already on antibiotics and analgesics prescribed by the previous dentist. Mucormycosis was suspected and therefore a CBCT was advised. The findings were suggestive of the destruction of a maxillary buccal and palatal alveolus in different regions [Figure 2B]. Complete loss of cortical plates was noted in the alveolar bone of teeth #15 to 26 region resulting in the detached appearance of the cancellous bone from the peripheral margins. Also, opacification was seen in the maxillary sinuses bilaterally involving peripheral walls blocking the osteo-meatal complexes. Perforation of the nasal floor was noted on the left aspect of tooth # 24 region. The patient was immediately referred to the medical department where the case of mucormycosis was confirmed.

Figure 2A - Initial clinical presentation of mucormycosis with multiple periodontal abscesses; 2B) CBCT revealing complete loss of cortical plates wrt 15 to 26 with nasal floor perforations, haziness in the maxillary sinus can be appreciated.
Case 3
A 54 yr female patient with uncontrolled diabetes reported to the Department of Periodontics with a chief complaint of an unhealed extraction socket in relation to tooth #25 [Figure 3]. The patient had undergone an extraction of tooth #25, 15 days back. Since then, the patient noticed pain, multiple boil/s, and mobility in the adjacent teeth. The patient also had a history of COVID-19 infection two months back but was not hospitalised. CBCT findings revealed complete opacification of the left maxillary sinus with palatal perforation on the same side with respect to teeth #25 and 26 regions. The roof of the maxillary sinus was intact on both the left and right sides.
All three patients were immediately referred to the Department of emergency medicine where they confirmed the diagnosis as mucormycosis and started further management of the same.

Figure 3 - Clinical presentation of palatal perforation in mucormycosis with unhealed extraction socket wrt 25.
DISCUSSION
Mucormycosis is an angio-invasive fungal infection caused by the fungi belonging to the order Mucorales. The Mucorales can cross physical barriers and invade host defence mechanisms mainly in immunocompromised individuals. Human infection is said to be caused by asexual spore formation which then becomes airborne and resides on the oral and nasal mucosa of humans. In the vast majority of immunologically competent hosts, these spores are contained by the host phagocytic response. However, if the host defence fails, germination follows by hyphae development. It progresses as the hyphae begin to invade vessel walls and arteries causing thrombosis and ischemia of the affected tissues. Fungal hyphae produce a substance called Rhizoferrin (Siderophores), which binds to iron available in the blood circulatory system. This Iron-Rhizoferrin complex is responsible for the fungus growth and its intracellular processes. It has been documented that R. arrhizus adheres directly to the vascular endothelial cells through the endothelial cell surface receptor, human glucose-regulated protein 78 (GRP78) [4]. This invasion of the vascular system significantly increases acidosis and hyperglycaemic conditions. Patients with neutropenia have more extensive angioinvasion. Also, ischemic necrosis of the infected tissues caused by a fungus can prevent the delivery of leukocytes and antifungal agents to the foci of infection.
Various risk factors have been reported for mucormycosis which are listed in Table 1 [5, 6]. The difference in the prevalence of risk factors/underlying disease varies among different countries. Prakash H et al in 2019 published a review on the global epidemiology of mucormycosis and reported that in Asian countries, diabetes mellitus is the most common risk factor whereas transplantation and haematological disorders are more commonly seen as risk factors in European countries [7]. With the pandemic around the globe, there is an increase in rhinomaxillary mucormycosis in COVID-19 recovered patients. Acute lung damage in COVID-19 is controlled by administering corticosteroids. This led to transient hyperglycaemia, which coupled with low immunity served as an ideal environment for fungal growth and proliferation. Fungus once established in the paranasal sinuses can spread rapidly to the orbit and cerebral region via the nasolacrimal duct and medial orbit. So, it can be assumed that COVID-19 also can be one of the contributing factors/risk factors for rhinomaxillary mucormycosis.

Rhinomaxillary mucormycosis located in the periodontal tissue (gingival and alveolar bone) are rare. Most often, the clinical presentation is that of a non-healing extraction socket. Initial signs are non-specific necrotic ulcer, which later turns into a characteristic black necrotic lesion.
A literature search was performed in PubMed/Medline database using the terms “periodontal mucormycosis”, “rhinomaxillary mucormycosis”, and “gingival mucormycosis”. A total of 35 research papers have been retrieved. Only case reports in which cases showed a periodontal or gingival presentation of mucormycosis in the maxillary or mandibular arch were considered. A total of eleven case reports were included in the manuscript, whose main characteristics have been summarized in Table 2. None of these reported cases had a history of COVID-19. Nagalli et. al. in 2021 published a systematic review on “Mucormycosis in COVID-19” [6]. None of the cases in their review showed any gingival or periodontal clinical findings.

Roden MM et al. in 2005 conducted a review of 929 cases of zygomycosis [8]. The review showed that the fungal infection mainly occurred in the mean age group of 38.8 years with a higher prevalence seen in the males (65%) compared to females. Diabetes mellitus was the most common underlying condition seen in most patients. 154 patients presented with a malignant neoplasm, among which 147 (95%) were having haematological malignancies. However, Bonifaz A et al in 2020 in their literature review reported 11 cases of periodontal mucormycosis. The common risk factor in 10 of the patients was haematological malignancies and one was reported with diabetes [9]. In the present paper, the first case had transient hyperglycaemia due to steroids intake for COVID-19 treatment, the second case had controlled diabetes but had taken steroids for COVID-19 treatment, which again lead to transient hyperglycaemia during the treatment and the third patient had uncontrolled diabetes and was not taking any medications for the same. The common risk factor in all three patients was hyperglycaemia during COVID-19 infection. All three cases showed mucormycosis presentation within two months of COVID-19 recovery.
Out of the 3 reported cases, two were male and one female and all had a similar clinical gingival presentation. Palatal swelling was present in all three cases. It has been documented that along with angiotensin-converting enzyme (ACE2) receptors, the CoV-2 can bind to the glucose-regulating protein 78 (GRP78) receptors also to get entry into the host cells [20]. Therefore it can be hypothesized that as glucose-regulating protein 78 (GRP78) receptors are a common link for invasion into the host defence mechanism by mucormycosis as well as by SARS-CoV-2, the increased number of GRP78 receptors during COVID-19 infection, can be a reason leading to surge in mucormycosis cases after COVID-19 infection [21]. No case has been documented yet describing the gingival presentation of this silent and rare infection in Covid-19 recovered patients. This paper emphasizes the role of dental surgeons in recognizing the initial symptoms leading to early diagnosis. This otherwise neglect may later lead to a poor prognosis on extension into the rhino-orbital and rhino-cerebral region.
Mucormycosis is a medical emergency. Computed tomography (CT) is the cornerstone of modern medical radiology to diagnose the extension of the lesion in the rhinomaxillary region. Cone-beam Computed tomography (CBCT), which is advanced imaging technology in dentistry, provides images equivalent to medical CT at reduced costs and radiation doses. The radiation dose to the patient with CBCT is 40% lesser than that of the multi-slice CT dose. However, the usefulness of CBCT in the diagnosis of mucormycosis may be limited, especially if there are intracranial extensions. Due to the rapidity of invasive infection, CBCT should be immediately advised by dental professionals to monitor disease extension, if mucormycosis is suspected in COVID-19 recovered patients.
Prompt diagnosis, early management, and reversing the underlying risk factors, if possible may improve the prognosis and survival rate of the patient. In all three patients, CBCT was advised immediately on suspecting mucormycosis based on history and clinical findings. As all three CBCTs’ revealed unusually massive bone destruction, a provisional diagnosis of mucormycosis was made and the patients were immediately referred to the emergency medicine department. Further detailed investigations by the medicine and ENT department confirmed mucormycosis as the final diagnosis.
Surgical management also should be initiated early in the course of treatment. This should involve the debridement of all infected tissues. Debridement is commonly defined as the process of removing necrotic and devitalized tissue. The presence of necrotic tissue within a wound may impair wound repair processes by stimulating inflammation and delaying granulation and epithelialization. Several methods for debridement may be employed, including enzymatic, mechanical, autolytic, and surgical, along with systemic antifungals. Survival rates among patients with invasive sinus involvement without cerebral involvement may be as high as 50-80% [22]. Amphotericin B is the antifungal agent of choice. It is a polyene antifungal agent that acts by binding to sterols (primarily ergosterol) in the fungal cell membrane with a resultant change in membrane permeability. Combined treatment of thorough debridement and amphotericin B has a survival rate of 80% [14]. Lipid complex amphotericin B is a formulation designated to be less nephrotoxic than conventional amphotericin B [23]. Other antifungals used against Mucorales are posaconazole and isavuconazole [23-25]. Early surgical debridement and management due to timely diagnosis of mucormycosis prevented the further fungal spread and aggressive treatment in all three of the cases reported.
CONCLUSIONS
This case series presents 3 patients with rare clinical rhinomaxillary mucormycosis post-Covid-19 infection. The prompt and quick diagnosis followed by immediate referral, with a proper treatment plan prevented the rapid spreading of fungal infection to the orbital and cerebral region. Dental professionals play a critical role in spotting initial oral findings, advising timely CBCT, and early referral for further management. This can significantly reduce the morbidity of mucormycosis.
Declaration of competing interest
The authors declare that they have no known financial conflicts of interest.
Funding
None
REFERENCES
[1] Paltauf A. Mycosis mucorina. Virchow Arch Pathol Anat. 1885; 102, 543-564.
[2] Spellberg B, Edwards J J, Ibrahim A. Novel perspectives on mucormycosis: pathophysiology, presentation, and management. Clin Microbiol Rev. 2005; 18, 556-569.
[3] Dyer O. The excessive use of steroids in the treatment of COVID-19 and immunosuppression by the virus led to the emergence of this opportunistic fungal infection Covid-19: India sees record deaths as “black fungus” spreads fear. BMJ. 2021; 373, n1238.
[4] Liu M, Spellberg B, Phan Q, et al. The endothelial cell receptor GRP78 is required for mucormycosis pathogenesis in diabetic mice. J Clin Invest. 2010; 120, 1914-1924.
[5] Rincón CCA, Silva-Ramos CR, Arancibia JA, Prada-Avella MC, Suárez A. Rhino-orbito- cerebral mucormycosis in an acute lymphoblastic leukemia pediatric patient. Case report and review of literature. Infez Med. 2022; 30 (2), 298-303.
[6] Nagalli S, Kikkeri NS. Mucormycosis in COVID-19: A systematic review of literature. Infez Med. 2021; 29 (4), 504-512.
[7] Prakash H, Chakrabarti A. Global epidemiology of Mucormycosis. J Fungi (Basel). 2019; 5, 26.
[8] Roden MM, Zaoutis TE, Buchanan WL, et al. Epidemiology and outcome of zygomycosis: a review of 929 reported cases. Clin Infect Dis. 2005; 41, 634-653.
[9] Bonifaz A, Gabriela Fuentes-Nava A, Tirado-Sanchez A, et al. Gingival mucormycosis: case report and literature review. Our Dermatol Online. 2020; 11, 389-392.
[10] Pandilwar PK, Khan K, Shah K, Sanap M, K S A, Nerurkar S. Mucormycosis: A rare entity with rising clinical presentation in immunocompromised hosts. Int J Surg Case Rep. 2020; 77, 57-61.
[11] Kumar N, Singh AK, Pandey S, Singh S. Rhino-maxillary osteomyelitis due to mucormycosis in an immunocompromised geriatric patient: A case report with review of treatment options. Int J Health Allied Sci. 2015; 4, 160-164.
[12] Azarpira N, Ashraf MJ, Kazemi K, Khademi B. Rhinomaxillary mucormycosis in a renal transplant recipient: case report. Exp Clin Transplant. 2012; 10, 605-608.
[13] McDermott NE, Barrett J, Hipp J, et al. Successful treatment of periodontal mucormycosis: report of a case and literature review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010; 109, e64-9.
[14] Goel S, Palaskar S, Shetty VP, Bhushan A. Rhinomaxillary mucormycosis with cerebral extension. J Oral Maxillofac Pathol. 2009; 14-17.
[15] Dogan M, Leblebisatan G, Haytac M, Antmen B, Surmegozler O. Oral mucormycosis in children with leukemia: report of 2 cases. Quintessence Int. 2007; 38, 515-520.
[16] Auluck A. Maxillary necrosis by mucormycosis. A case report and literature review. Med Oral Patol Oral Cir Bucal. 2007; 12, E360-4.
[17] Jayachandran S, Krithika C. Mucormycosis presenting as palatal perforation. Indian J Dent Res. 2006; 17, 139.
[18] Salisbury PLr, Caloss R J, Cruz JM, Powell BL, Cole R, Kohut RI. Mucormycosis of the mandible after dental extractions in a patient with acute myelogenous leukemia. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1997; 83, 340-344.
[19] Jones AC, Bentsen TY, Freedman PD. Mucormycosis of the oral cavity. Oral Surg Oral Med Oral Pathol.1993; 75, 455-460.
[20] Allam L, Ghrifi F, Mohammed H, et al. Targeting the GRP78-Dependant SARS-CoV-2 Cell entry by peptides and small molecules. Bioinform Biol Insights. 2020; 14, 1177932220965505.
[21] Sabirli R, Koseler A, Goren T, Turkcuer I, Kurt O. High GRP78 levels in Covid-19 infection: A case-control study. Life Sci. 2021; 265, 118781.
[22] William P. Rhinocerebral mucormycosis.Available from: http://www.emedicine.com.
[23] Mastroianni A. Paranasal sinus mucormycosis in an immunocompetent host: efficacy and safety of combination therapy with Liposomal Amphotericin B and adjuvant rHuGM-CSF. Infez Med. 2004; 12 (4), 278-283.
[24] Marty F, Ostrosky-Zeichner L, Cornely OA, Mullane KM, Perfect JR, Thompson GRr, et al. Isavuconazole treatment for mucormycosis: a single-arm open-label trial and case-control analysis. Lancet Infect Dis. 2016; 16, 828-837.
[25] Nagappan V, Deresinski S. Reviews of anti-infective agents: posaconazole: a broad-spectrum triazole antifungal agent. Clin Infect Dis. 2007; 45, 1610-1617.