Le Infezioni in Medicina, n. 2, 272-276, 2022
doi: 10.53854/liim-3002-13
ORIGINAL ARTICLE
Predictors of lethality in severe leptospirosis in Transcarpathian region of Ukraine
Pavlo Petakh1,2, Andriy Nykyforuk1
1Department of Biochemistry and Pharmacology, Uzhhorod National University, Uzhhorod, Ukraine;
2Department of Microbiology, Virology, and Immunology, I. Horbachevsky Ternopil National Medical University, Ternopil, Ukraine
Article received 2 February, accepted 3 April 2022
Corresponding author
Pavlo Pethakh
E-mail: pavlo.petakh@uzhnu.edu.ua
SummaRY
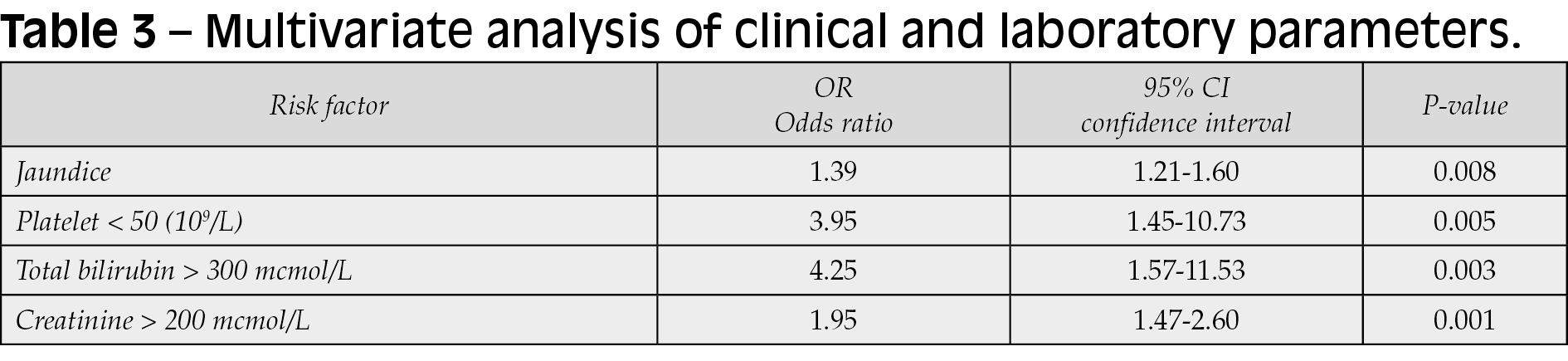
Leptospirosis is one of the most widespread zoonoses in the Transcarpathian region, with an average lethality of 12.5%. To determine the predictors of lethality, a retrospective study of 97 medical records of patients with leptospirosis in the period from 2009 to 2018 was conducted. Quantitative variables in the presence of normal distribution were compared using a paired Student’s t-test, and in the case of an abnormal distribution, the Mann-Whitney U test was used. The criterion χ2 was used for qualitative variables. Multivariate analysis was used for the calculation of the Odds ratio. The following factors that are associated with death from leptospirosis have been identified: total bilirubin greater than 300 mcmol/L (OR, 4.25; 95% confidence interval [CI], 1.57-11.53), platelets less than 50 x (109/L) (OR, 3.95; 95% confidence interval [CI], 1.45-10.73), creatinine above 200 mcmol/L (OR, 1.95; 95% confidence interval [CI], 1.47-2.60) and jaundice (OR, 1.39; 95% confidence interval [CI], 1.21-1.60). Detection of these predictors will help to quickly identify a patient at risk of severe course of the disease and death, which will allow deciding on the use of early intensive care.
Keywords: zoonoses, lethality, leptospirosis.
INTRODUCTION
Leptospirosis is one of the world’s most significant and extensively distributed zoonoses, found on all continents except Antarctica, and has posed a severe dilemma for epidemiologists and physicians around the world [1, 2].
Leptospirosis affects over one million people worldwide each year, with 58,000 of them dying [3]. This disease causes an annual loss of 2.9 million disability-adjusted life years (DALY) [4].
Ukraine is one of the regions with a high incidence of leptospirosis due to its climatic and geographical characteristics [5]. The highest incidence of leptospirosis in the last two decades was in the Transcarpathian, Kyiv, Kropyvnytskyi, Mykolaiv, Chernihiv, and Chernivtsi regions with maximum rates of 12.77 and 12.65 per 100,000 population in 1997 in Kropyvnytskyi and Transcarpathian regions, respectively [6].
Although leptospirosis is often an acute febrile illness (AFI), around 10% of patients may develop severe leptospirosis, which includes acute renal failure, jaundice, and/or pulmonary haemorrhage [7, 8]. Even with the best therapy, lethality from severe leptospirosis vary from 5 to 20%. [9]. Predictors of lethal outcomes should be evaluated taking into account local characteristics [10].
Early identification of severe or potentially severe leptospirosis patients may help to reduce the disease’s lethality, which is still rather significant [11, 12].
To determine the requirement for intensive care unit admission and more aggressive treatment procedures, prognostic markers that contribute to a severe course and lethality from leptospirosis must be identified [11, 12].
The problem of determining predictors of leptospirosis severity and lethality was studied by Dupont et al. (1997), who identified the main 5 factors associated with lethality, namely: dyspnoea, oliguria, white blood cell count over 12.900/mm3, repolarization abnormalities in ECG, alveolar infiltrates on chest radiographs [11]. Elizabeth De Francesco Daher et al. identified hypotension, tachypnoea, and acute kidney damage as risk factors for hospitalization in the intensive care unit (ICU) [12]. Hochedez P et al. recommend the use of qPCR to assess the severity of the disease [13].
In the Transcarpathian area, leptospirosis is still a very frequent zoonotic disease. In Transcarpathia, the case fatality ratio (CFR) for leptospirosis is 12.5%, compared to 9.8% nationally [14].
The study aimed to determine the factors associated with fatalities in leptospirosis in the Transcarpathian region.
PATIENTS AND METHODS
Data collection and definitions
A retrospective case-control study was conducted at the Transcarpathian Regional Clinical Infectious Diseases Hospital, Ukraine. The study protocol included a review of 97 medical records of patients who were hospitalized between 2009 and 2018. In this study, patients were divided into two groups: survivors (control) and non-survivors (cases). Leptospirosis was determined according to the criteria of the World Health Organization [15]. Each case was confirmed in the Especially Dangerous Infections (EDIs) of the State Institution Transcarpathian Region Center for Disease Control and Prevention of the Ministry of Health of Ukraine where a microscopic agglutination test (MAT) was conducted. Individuals who did not have laboratory confirmation of their diagnosis were excluded from the research.
Demographic (age, gender, place of residence (village or city)), clinical (jaundice), and laboratory data (alanine aminotransferase, total and conjugated (direct) bilirubin, creatinine, urea; granulocytes and platelets; erythrocyte sedimentation rate; the presence of icterohaemorrhagiae serogroup) were collected. On the first day of admission, all laboratory parameters were determined.
Statistical analysis
Statistical data were processed using IBM SPSS Statistics 23 software. Quantitative variables in the presence of normal distribution were compared using a paired Student’s t-test, and in the case of an abnormal distribution, the Mann–Whitney U test was used. The criterion χ2 was used for qualitative variables.
Adjusted odds ratios (OR) and 95% confidence intervals (CI) were calculated using multivariate analysis. The value of P ≤ 0.05 was considered significant.
Ethics
The protocol of this study was approved by the Ethics Committee of the Transcarpathian Regional Clinical Infectious Hospital.
RESULTS
Among the 97 leptospirosis patients included in the study, 75 patients (77%) survived and 22 (23%) died.
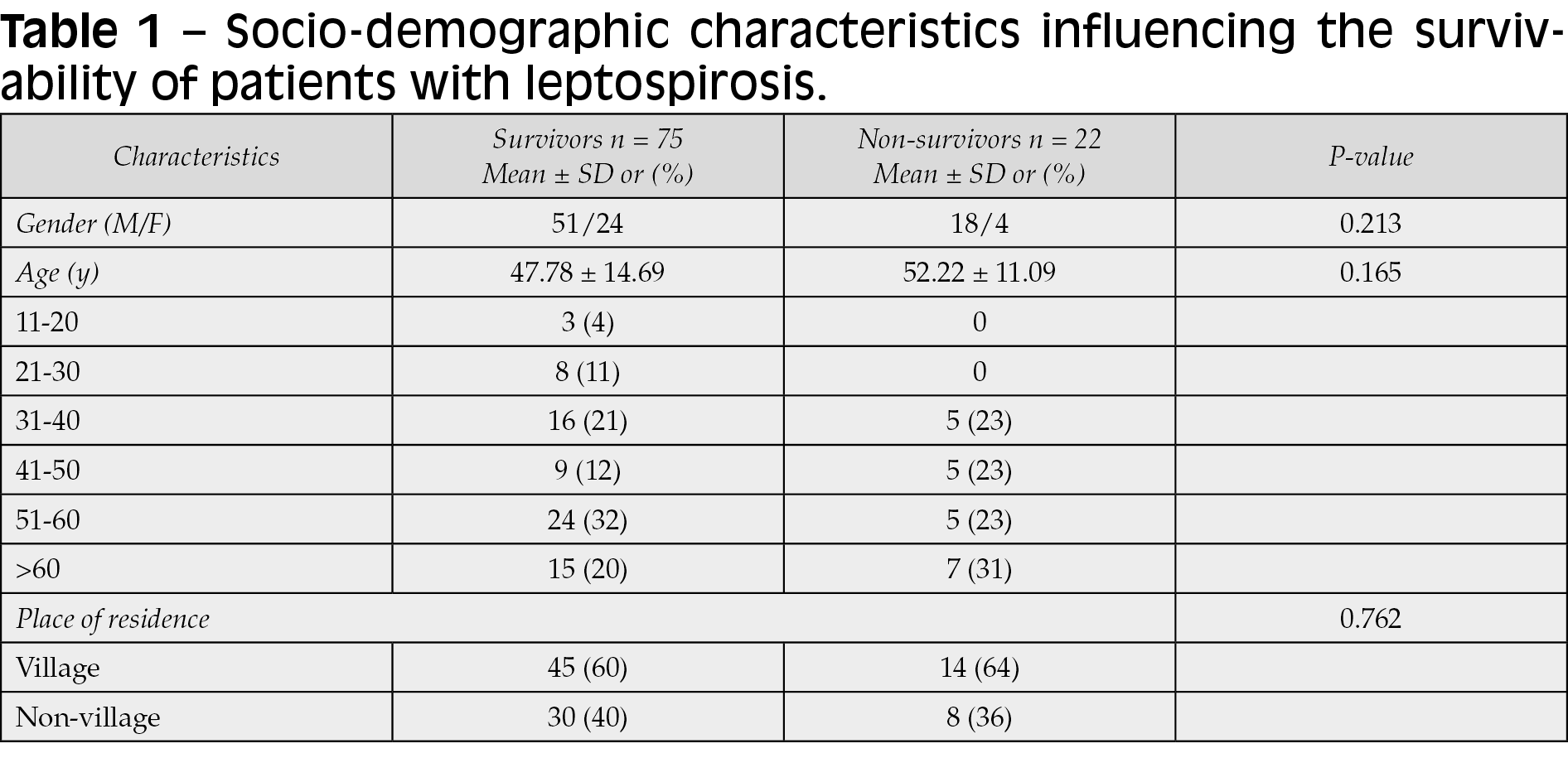
Demographic data, such as age, gender, and place of residence (urban or rural), shown in Table 1, are not related to death from leptospirosis (P>0.05).
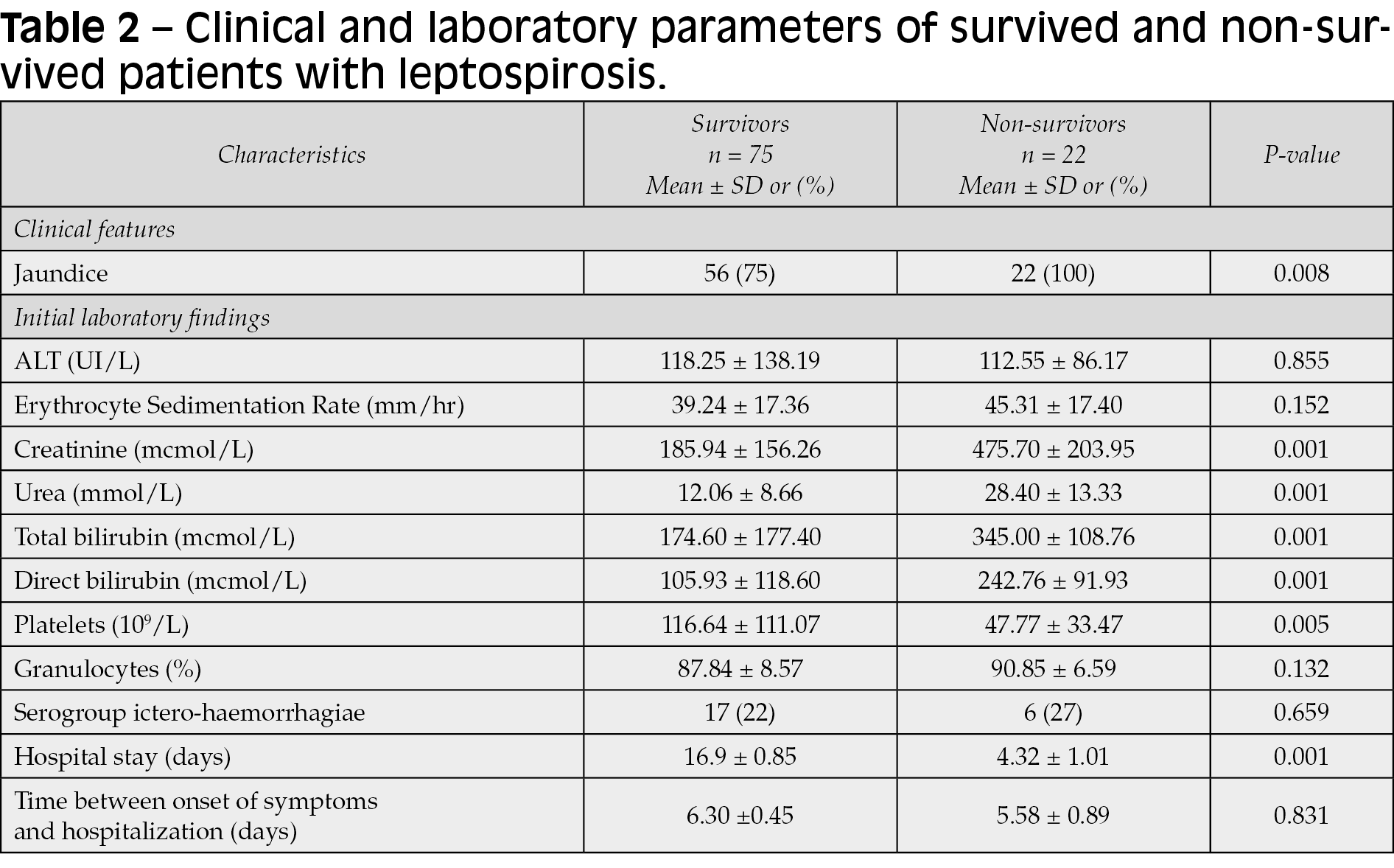
As shown in Table 2, patients who died from leptospirosis had higher serum creatinine levels compared with those who survived, namely 475.70±203.95 mcmol/L versus 185.94±156.26 mcmol/L, respectively (P=0.001). Statistically significant changes were also found in the levels of laboratory parameters such as urea (P=0.001), total bilirubin (P=0.001), direct bilirubin (P=0.001) and platelet level (P=0.005). Jaundice, as the clinical symptom of leptospirosis, was also associated with lethality (P=0.008). Alanine aminotransferase (ALT), erythrocyte sedimentation rate, granulocyte percentage in the blood, and the presence of icterohaemorrhagiae serogroup were not associated with leptospirosis lethality (P>0.05).
Jaundice, thrombocytopenia, hyperbilirubinemia, and increased serum creatinine were identified to be probable predictors of lethality in leptospirosis in a multivariate study (Table 3).
The strongest predictive risk factor for leptospirosis lethality is total bilirubin > 300 mcmol/L (OR, 4.25; 95% confidence interval [CI], 1.57-11.53).
DISCUSSION
For the healthcare system, leptospirosis is a major issue. This zoonotic disease has the potential for a severe course and a high case fatality rate. As a result, finding predictors of lethality and the requirement for intensive care to detect possibly fatalities early in the disease for better diagnosis and treatment is critical [16].
Leptospirosis is more frequent and lethal in the Transcarpathian region than in the rest of Ukraine. From 2005 to 2015, 420 cases of leptospirosis were recorded in this region, which is three times higher than the average incidence in Ukraine [14].
Patients who died in this research had higher total and direct bilirubin levels than those who recovered. Several scientific articles have proven the importance of hyperbilirubinemia as a predictor of lethality and severe leptospirosis [10-12]. In leptospirosis, liver enzymes such as ALT and AST are moderately increased, suggesting that liver impairment is generally mild and resolves with time [17]. The role of hepatic transaminases as a factor in disease severity or lethality needs to be further investigated, as there is evidence that confirms or refutes the role of these biochemical parameters in patients with leptospirosis. An aspartate aminotransferase–alanine aminotransferase ratio of >3 may indicate a poorer prognosis [12, 13, 18,19].
In our research, jaundice was found to be a risk factor for a more severe course of leptospirosis and lethality. Hepatic capillary injury occurs without hepatocellular necrosis, resulting in jaundice. There are retrospective studies that confirm, and deny the role of jaundice as a predictor of death in this infectious disease [10, 12, 20, 21].
Thrombocytopenia, which is frequent in leptospirosis, is another key risk factor for a severe course of the disease and lethality [22]. One potential explanation for thrombocytopenia in leptospirosis is that certain strains of Leptospira directly activate platelets [23]. Thrombocytopenia in the acute phase of the disease may play a role in hemorrhagic disorders. In many studies, thrombocytopenia has been identified as one of the most common causes of severe course and death [10, 11, 24, 25]. Another factor that contributes to bleeding in the acute phase of the disease may be uraemia. The pathophysiology of bleeding in uraemia is multifaceted, nevertheless, alterations in platelet-platelet and platelet-vessel wall interaction play a crucial role. Platelet dysfunction is caused in part by uremic toxins found in circulating blood [26].
Increased blood creatinine and urea levels indicate kidney damage and the potential for acute renal failure, one of the most common and crucial predictors of death in leptospirosis [24]. The presence of renal failure in leptospirosis patients should be given special attention when it is accompanied by jaundice (Weil’s syndrome) because it is the clinical syndrome most associated with the risk of death [25].
According to some authors, the icterohaemorrhagiae serogroup is more prevalent in individuals with a more severe course of the disease and may be linked to renal failure, however, in our study, the link between this serogroup with the severe course and death was not found, as in the study of Spichler et al [10, 11, 13, 27].
The age, which did not differ between the two groups in our study (P > 0.05), but did in certain studies, is one of the demographic variables that can be discussed, however, patients who died from leptospirosis were older than those who recovered [10, 11]. Most studies, including ours, have discovered that while gender is not a major determinant in leptospirosis lethality, the number of cases has been higher in men than in women [10, 12].
In conclusion, in the Transcarpathian region of Ukraine, leptospirosis remains as a common zoonotic disease that can cause acute renal failure, haemorrhage, and death.
In order to reduce lethality, we have proposed 4 important predictors of lethality in leptospirosis: the presence of jaundice, platelets less than 50 (109/L), total bilirubin more than 300 mcmol/L and creatinine of more than 200 mcmol/L. These “red flag” laboratory and clinical characteristics will aid medical personnel in rapidly identifying a patient at risk of death, which is critical in determining the severity of the condition and the need for early intensive care and therapy adjustment.
Acknowledgments
The authors thank the director of the Transcarpathian Regional Clinical Infectious Diseases Hospital, Mykhailo Polyak, for his assistance in conducting this study.
Funding
There was no specific funding for this project
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
REFERENCES
[1] Adler B, de la Peña Moctezuma A. Leptospira and leptospirosis. Vet Microbiol. 2010; 140 (3-4), 287-96.
[2] Perry RJ, Peng L, Barry NA, et al. Acetate mediates a microbiome–brain-β-cell axis to promote metabolic syndrome. Nature. 2016; 534 (7606), 213-7.
[3] Costa F, Hagan JE, Calcagno J, et al. Global morbidity and mortality of leptospirosis: a systematic review. PLoS Negl Trop Dis. 2015; 9 (9), e0003898.
[4] Torgerson PR, Hagan JE, Costa F, et al. Global burden of leptospirosis: estimated in terms of disability adjusted life years. PLoS Negl Trop Dis. 2015; 9 (10), e0004122.
[5] Kravchuk YA, Vasylieva N. Epizootolohoepidemiolohichni osoblyvosti leptospirozu v Ternopilskii oblasti [Epizootological and epidemiological features of leptospirosis in the Ternopil region]. Analy Mechnykovskoho Instytutu. 2015; 2, 165-71.
[6] Hopko N. Epidemiolohichni osoblyvosti leptospirozu v Ukraini v umovakh sohodennia. Visnyk Problem Biolohii i Medytsyny. 2017; 4 (3), 84-6.
[7] Ko AI, Goarant C, Picardeau M. Leptospira: the dawn of the molecular genetics era for an emerging zoonotic pathogen. Nature Reviews Microbiology. 2009; 7 (10), 736-47.
[8] Yersin C, Bovet P, Mérien F, et al. Pulmonary haemorrhage as a predominant cause of death in leptospirosis in Seychelles. Trans R Soc Trop Med Hyg. 2000; 94 (1), 71-6.
[9] Goswami RP, Goswami RP, Basu A, Tripathi SK, Chakrabarti S, Chattopadhyay I. Predictors of mortality in leptospirosis: an observational study from two hospitals in Kolkata, eastern India. Trans R Soc Trop Med Hyg. 2014; 108 (12), 791-6.
[10] Spichler AS, Vilaça PJ, Athanazio DA, et al. Predictors of lethality in severe leptospirosis in urban Brazil. Am J Trop Med Hyg 2008; 79 (6), 911.
[11] Dupont H, Dupont-Perdrizet D, Perie JL, Zehner-Hansen S, Jarrige B, Daijardin JB. Leptospirosis: prognostic factors associated with mortality. Clin Infect Dis. 1997; 25 (3), 720-4.
[12] Daher EDF, Soares DS, de Menezes Fernandes ATB, et al. Risk factors for intensive care unit admission in patients with severe leptospirosis: a comparative study according to patients’ severity. BMC infectious diseases. 2015; 16 (1), 1-7.
[13] Hochedez P, Theodose R, Olive C, et al. Factors associated with severe leptospirosis, Martinique, 2010–2013. Emerg Infect Dis. 2015; 21 (12), 2221.
[14] Markovych O, Tymchyk V, Kolesnikova I. Leptospirosis in Zakarpattia Oblast (2005–2015). Vector Borne Zoonotic Dis. 2019; 19 (5), 333-40.
[15] Organization WH. Human leptospirosis: guidance for diagnosis, surveillance and control: World Health Organization; 2003.
[16] Lim V. Leptospirosis: a re-emerging infection. Malays J Pathol. 2011; 33 (1), 1.
[17] Bharti AR, Nally JE, Ricaldi JN, et al. Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis. 2003; 3, 757-71.
[18] Sandhu RS, Ismail HB, Ja’afar MHB, Rampal S. The predictive factors for severe leptospirosis cases in Kedah. Trop Med Infect Dis. 2020; 5 (2), 79.
[19] Chang ML, Yang CW, Chen JC, et al. Disproportional exaggerated aspartate transaminase is a useful prognostic parameter in late leptospirosis. World J Gastroenterol. 2005; 11 (35), 5553-6.
[20] Durmaz Cetin B, Harmankaya O, Hasman H, Gunduz A, Oktar M, Seber E. Acute renal failure: a common manifestation of leptospirosis. Renal failure. 2004; 26 (6), 655-61.
[21] Daher EF, Silva Jr GB, Karbage NN, et al. Predictors of oliguric acute kidney injury in leptospirosis. Nephron Clin Pract. 2009; 112 (1), c25-c30.
[22] Daher EF, Silva GB, Silveira CO, et al. Factors associated with thrombocytopenia in severe leptospirosis (Weil’s disease). Clinics. 2014; 69, 106-10.
[23] Wagenaar JF, Goris MG, Sakundarno M, et al. What role do coagulation disorders play in the pathogenesis of leptospirosis? Trop Med Int Health. 2007; 12 (1), 111-22.
[24] Panaphut T, Domrongkitchaiporn S, Thinkamrop B. Prognostic factors of death in leptospirosis: a prospective cohort study in Khon Kaen, Thailand. Int J Infect Dis. 2002; 6 (1), 52-9.
[25] Tantitanawat S, Tanjatham S. Prognostic factors associated with severe leptospirosis. J Med Ass Thai. 2003; 86 (10), 925-31.
[26] Boccardo P, Remuzzi G, Galbusera M. Platelet dysfunction in renal failure. Semin Thromb Hemost. 2004; 30 (5), 579-89. doi: 10.1055/s-2004-835678.
[27] Tubiana S, Mikulski M, Becam J, et al. Risk factors and predictors of severe leptospirosis in New Caledonia. PLoS Negl Trp Dis. 2013; 7 (1), e1991.