Le Infezioni in Medicina, n. 1, 1-10, 2022
doi: 10.53854/liim-3001-1
REVIEWS
A review of adverse effects of COVID-19 vaccines
Hisham Ahmed Mushtaq1, Anwar Khedr1, Thoyaja Koritala2, Brian N. Bartlett3, Nitesh K. Jain4, Syed Anjum Khan4
1Research Trainee, Mayo Clinic Health System - Southwest Minnesota Region, Mankato, Minnesota, United States;
2Department of Hospital Internal Medicine, Mayo Clinic Health System - Southwest Minnesota Region,
Mankato, Minnesota, United States;
3Department of Emergency Medicine, Mayo Clinic Health System - Southwest Minnesota Region, Mankato, Minnesota, United States;
4Department of Critical Care Medicine, Mayo Clinic Health System - Southwest Minnesota Region, Mankato, Minnesota, United States
Article received 10 December 2022, accepted 7 February 2022
Corresponding author
Syed Anjum Khan
E-mail: khan.syed@mayo.edu
SummaRY
The COVID-19 pandemic has led to unanticipated pressures on all aspects of human life. Multiple approaches to eliciting protective immunity must be rapidly evaluated. Numerous efforts have been made to develop an effective vaccine for this novel coronavirus, resulting in a race for vaccine development. To combat COVID-19, all nations must focus their efforts on widespread vaccination with an effective and safe vaccine. Globally, concerns about potential long-term adverse effects of vaccines have led to some apprehension about vaccine use. A vaccine’s adverse effect has an integral role in the public’s confidence and vaccine uptake. This article reviews the current primary literature regarding adverse effects associated with different COVID-19 vaccines in use worldwide.
Keywords: adverse effects, adverse events, complications, COVID-19, vaccine.
BACKGROUND
As of January 14, 2022, the World Health Organization (WHO) has confirmed about 318,648,834 cases of COVID-19 worldwide, including 5,518,343 fatalities [1]. The COVID-19 pandemic has resulted in a global economic disruption. To restore normalcy and enable economic growth, vaccines are the best option. The first COVID-19 vaccine introduced in December 2020 has become a milestone in the fight against this pandemic. On December 2, 2020, using an Emergency Use Authorization (EUA), the UK became the first country to approve Pfizer-BioNTech’s COVID-19 vaccine, BNT162 [2]. As of December 31, 2020, the WHO approved BNT162 for emergency use, making its global production and supply more efficient [3]. Different vaccine candidates for COVID-19 have been approved using similar EUA processes, and the list continues to grow.
A historic vaccination campaign is taking place in the US currently. In 1 week, 1.12 million doses were administered daily, on average. More than 523 million doses have been given in the US to date (Figure 1) [4, 5]. As of January 14, 2022, 194 vaccines are in preclinical development, and 139 are in clinical trials [6].
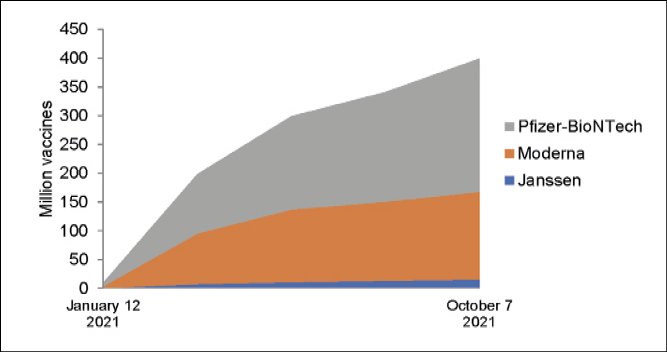
Figure 1 - COVID-19 Vaccines Administered in the US by Manufacturer [5].
Vaccine uptake must be accelerated in the coming months to continue to decrease infection rates [7]. However, some people question whether the speed at which a vaccine is developed will compromise its efficiency and safety. This, in turn, may lead to vaccine hesitancy, which further inhibits attaining the goal of having 70% of the population fully vaccinated, after which herd immunity can effectively be achieved [8]. Therefore, it is crucial to establish the safety of the vaccines in these circumstances to perhaps promote wider vaccine acceptance among hesitant people. Adverse effects, however, are associated with every vaccination [9]. The purpose of this article is to review the current primary literature regarding adverse affects associated with the different COVID-19 vaccines. Our aim is to provide insights into the safety of the vaccines to help address misinformation and vaccine hesitancy. We discuss the adverse effects of the most common vaccines, which were chosen based on the number of countries they are approved in.
This article is intended to be a narrative review. Searches were conducted in PubMed and Google Scholar to identify related literature from 2020 to 2021. Keywords such as “adverse effects,” “adverse events,” “complications,” “COVID-19,” and “vaccine,” were searched individually or in combination to yield relevant information. The results were reviewed for relevance to the topic, and the articles were screened by 2 authors. We had no language restrictions because of the relatively few articles on the topic. Duplicated studies and studies providing insufficient and irrelevant information were excluded.
mRNA Vaccines
Pfizer-BioNTech
Pfizer-BioNTech’s BNT162 vaccine is a lipid nanoparticle-derived, nucleoside-modified mRNA vaccine that encodes the SARS-CoV-2 glycoprotein spike [10]. The UK was the first nation to approve BNT162 on December 2, 2020 [2]. A first EUA for BNT162 was issued by the US Food and Drug Administration (FDA) on December 11, 2020 [11]. BNT162 was later approved by Canada and Mexico via their respective EUAs. The WHO approved the first vaccine candidate, BNT162, on December 31, 2020, for emergency use, therefore facilitating easy production and distribution globally [3]. A total of 232.52 million doses of the Pfizer-BioNTech vaccine have been given in the US through October 7, 2021 [12].
Moderna
mRNA-1273 from Moderna is a lipid-encapsulated mRNA vaccine that encodes the SARS-CoV-2 prefusion-stabilized spike protein [13]. The FDA issued an EUA for mRNA-1273 on December 18, 2020 [11]. It was the second COVID-19 vaccine in the US to be authorized under an EUA [14]. As of October 7, 2021, 152.51 million doses of Moderna vaccine have been given in the US [12].
Considerations with mRNA vaccines
For the 2 mRNA vaccines, the second dose was associated with more adverse effects than the first dose [15]. A higher rate of systemic events was reported by younger vaccine recipients (aged 16-55 years) than those older than 55 years, which may be due to a more robust immunogenic response in younger persons [15].
Evaluation of the vaccines vs placebo (normal saline) showed a higher incidence of mild local adverse effects such as pain, heat, swelling, and redness [15]. The vaccines were also associated with other systemic adverse effects such as fever, fatigue, arthralgias, myalgias, and headache. These adverse effects usually developed within 1 to 2 days of vaccination [15]. In initial trials, the localized symptoms were mild to moderate in severity and lasted 1 to 2 days. Moderate to severe systemic symptoms, such as headache, myalgia, arthralgia, and fatigue, also lasted 1 to 2 days [15]. More local reactions were seen among the vaccine group than the placebo group. The most common localized symptom was pain at the injection site, which was seen within 1 week of vaccination [15]. Anaphylaxis and edema of the labial, facial, and glossal areas were among the adverse events noted [16].
Adenoviral vaccines
Oxford/AstraZeneca ChAdOx1 nCoV-19 Vaccine (AZD1222)
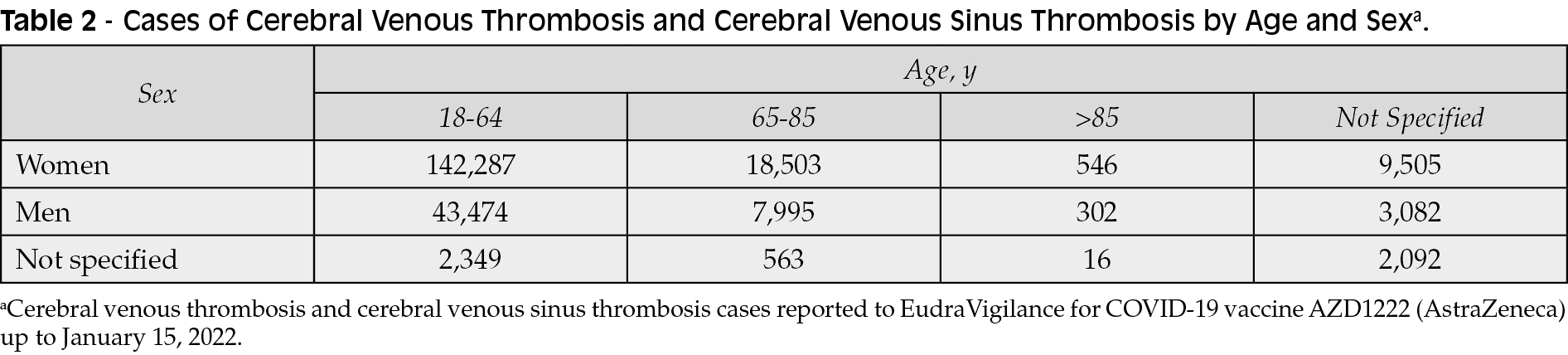
The SARS-CoV-2 structural surface spike protein gene is integrated into the ChAdOx1 nCoV-19 vaccine (AZD1222; trade name Vaxzevria) from Oxford/AstraZeneca, which is made from replication-deficient chimpanzee adenovirus ChAdOx1 [17]. Efficacy and safety results for AZD1222 have been documented in 4 randomized clinical trials in the UK, South Africa, and Brazil [17]. Overall, the vaccine was safe across all 4 studies, and serious adverse events were evenly distributed among all study groups. A total of 168 serious adverse events were reported among 79 recipients of AZD1222 and 89 recipients of saline control [17]. One case of transverse myelitis was reported 14 days after the second dose of AZD1222; this was viewed as possibly related to vaccination, and a diagnosis of an idiopathic, short-segment, spinal cord demyelination was made. In South Africa, 1 patient had a fever higher than 40°C 2 days after vaccination, but the patient recovered quickly [17]. In another study, laboratory tests in 11 patients in Austria and Germany indicated either thrombocytopenia or thrombosis after being vaccinated with AZD1222 [18]. The Supplemental Figure shows the number of individual events by reaction group identified in the European database of suspected adverse drug reaction reports (EudraVigilance) for AZD1222 (up to January 15, 2022) [19].
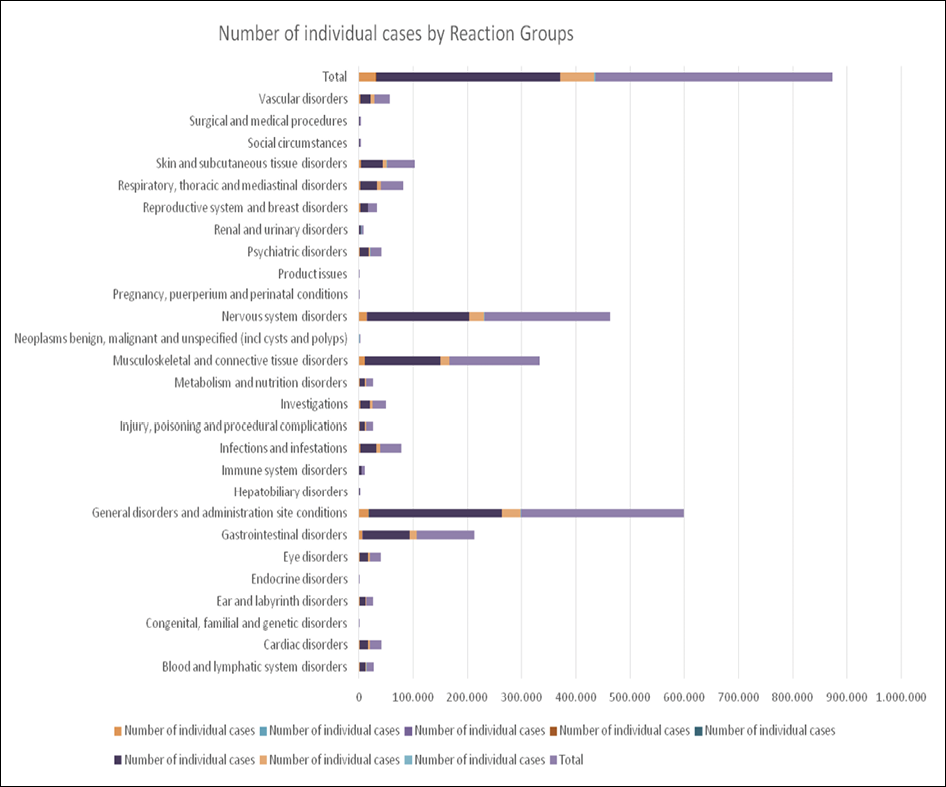
Supplemental Figure
Johnson & Johnson (Janssen) Ad26.COV2.S
The Ad26.COV2.S vaccine from Johnson & Johnson (Janssen) was the third COVID-19 vaccine approved to be used in the US. Ad.26.COV2.S employs a human adenoviral type 26 vector platform [20]. The first 2 approved mRNA vaccines require 2 doses, whereas the Janssen vaccine is given as a single dose intramuscularly. Ad.26.COV2.S was granted an EUA by the FDA on Feb 27, 2021 [14]. Low- and middle-income countries prefer adenoviral vaccines because they do not require high-level cold-chain storage, and Ad26.COV2.S requires only 1 dose [20].
After 6 recipients were diagnosed with cerebral venous sinus thrombosis and thrombocytopenia, the FDA and the Centers for Disease Control and Prevention (CDC) recommended a pause in the administration of Janssen vaccines [21]. In Europe, reports of similar thrombotic events have been observed primarily among women younger than 60 years after receiving the AstraZeneca AZD1222 vaccine [20].
Sputnik V
Sputnik V (Gam-COVID-Vac) is a 2-part adenoviral vaccine against SARS-CoV-2. Specifically, it contains the DNA for the spike protein encoded by SARS-CoV-2 that the virus uses to infect human cells. An immune response is triggered to the spike protein [22]. This vaccine consists of 2 adenoviral vectors (rAd26 and rAd5) administered in separate doses, 21 days apart. [23] The use of recombinant adenovirus is similar to the Oxford AstraZeneca and the Janssen vaccines [17, 24].
The Gamaleya National Center of Epidemiology and Microbiology in Moscow was already devising prototypes of Sputnik V when the WHO declared COVID-19 a pandemic [25]. In September 2020, researchers published results from phases I and II of an open, nonrandomized trial of 76 participants [26]. All participants were reported to have developed antibodies against SARS-CoV-2. Pain at the injection site (44 [58%]), asthenia (21 [28%]), headache (32 [42%]), hyperthermia (38 [50%]), and muscle pain (18 [24%]) were among the most common adverse events. Serious adverse events were not observed [26]. The rapidity and lack of transparency in the development of the Sputnik V vaccine have been criticized, however [27].
The phase III interim report included results for more than 20,000 participants. The vaccine was not directly linked to any serious adverse events. However, 45 participants who were given the vaccine and 23 who were given the placebo experienced serious adverse effects that were not related to the vaccine [26].
Surveillance programs
To confirm vaccine safety, an objective analysis of adverse effects and potential adverse reactions is required. To this effect, several surveillance programs are used. The Vaccine Adverse Event Reporting System (VAERS), created by the CDC and FDA, monitors adverse reactions after vaccination (Figure 2) [28, 29]. Reports can be submitted by vaccine manufacturers, health care providers, and the general public. VAERS requires reporting of various adverse events by health care providers, including deaths, as part of the European Union Agreements on COVID-19 vaccines [28].
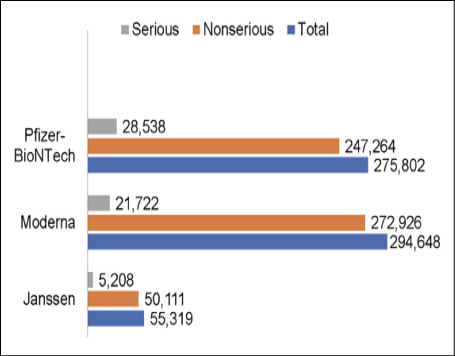
Figure 2 - Vaccine Adverse Events According to the Vaccine Adverse Event Reporting System [29]. Events reported through October 7, 2021.
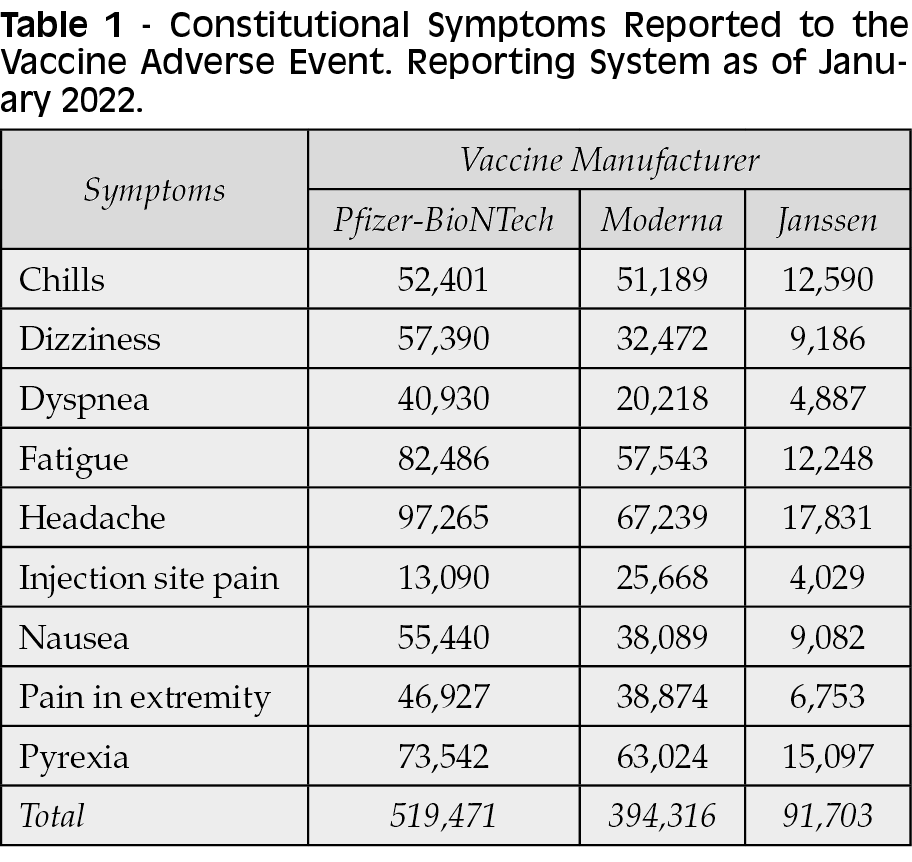
VAERS database entries do not indicate a causal relationship between vaccination and the cases. In addition, the VAERS database is based on passive surveillance and therefore could be biased or contain errors resulting from reporting bias. Because of the large number of vaccines administered and the prevalence of serious adverse events in the population, some cases of these conditions may occur by chance during the postvaccination period, unrelated to the vaccines themselves [30]. Constitutional symptoms reported to VAERS as of January 7, 2022, are shown in Table 1 [31, 32].
The v-safe program is a system of surveillance using text messages to collect information regarding vaccine adverse effects. In v-safe, vaccine recipients are consistently prompted to complete short medical surveys, including an inquiry about the injection site and systemic reactions and health effects. When enrollees seek medical care, the v-safe call center notifies them and encourages them to fill out a VAERS report [28].
Specific Adverse Events
Thrombosis
Recently, several reports of thrombocytopenia with thrombosis, most notably cerebral venous sinus thrombosis or cerebral venous thrombosis (CVT) within 28 days of vaccination, have been associated with Ad26.COV2.S (Janssen) and AZD1222 (AstraZeneca) (Table 2), both of which use the adenovirus-vector platform [19, 33]. Reports of thrombosis could have implications for vaccine uptake all over the world. Consequently, many nations have altered their vaccination guidelines. AZD1222 was made available only to adults older than 40 years in the UK, older than 55 years in Canada, and older than 60 years in Germany [33-35]. As a result of 6 reports of CVT, the FDA and CDC recommended a pause in the administration of Ad26.COV2.S vaccine in the US on April 13, 2021 [21].
New-onset severe headache is an important symptom of CVT and occurs in up to 67% of persons within the first few days after COVID-19 immunization [36]. It is critical for health care providers to diagnose CVT in vaccinated patients and to evaluate and treat patients with suspicion of immune-mediated thrombocytopenia with thrombosis resulting from vaccination. A CVT event occurs when the smaller draining cortical veins or the cerebral venous sinus system are completely or partially occluded [37]. It is more likely to occur in young adults and is 3 times as common among women than men [38].
Antibodies to platelet factor 4 (PF4) were detected in several patients who had CVT events after vaccination with Ad26.COV2.S and AZD1222 vaccines, which mimicked autoimmune heparin-induced thrombocytopenia [39]. Antibody complexes involving PF4 are formed which bind the Fc gamma receptor of platelets, form crosslinks, and activate the platelets [18]. Similar to heparin-induced thrombocytopenia, when platelets are consumed, thrombocytopenia is precipitated, and when monocytes and platelets are activated, thrombin production increases, which leads to thrombosis. In addition, an increase in D-dimer levels is seen, and eventually, it leads to disseminated intravascular coagulation [18]. The reason for adenoviral vector vaccines being associated with PF4 antibody production and thrombosis is unknown, but animal trials have shown that adenoviral vaccines can be biodistributed in the brain. Therefore, the presence of spike protein in the cerebral tissues can trigger an autoimmune reaction and eventual thrombosis [40].
The development of CVT is 41 times more likely in patients with COVID-19 than those without COVID-19, according to analyzed TriNetX data [41]. Thus, COVID-19 vaccination provides an overall benefit. In the US, on April 27, 2021, authorities decided to resume the use of the Ad26.COV2.S vaccine in all adults older than 18 years [42]. However, the CDC included a warning for women younger than 50 years on the risks of thrombosis associated with this vaccine [43].
Guillain-Barré syndrome
In developed countries, Guillain-Barré syndrome (GBS) is one of the leading causes of acute flaccid paralysis, characterized by autonomic dysfunction, sensory abnormalities, and varying degrees of weakness. Although the specific pathophysiology is not known, this disorder is believed to result from an autoimmune response [44].
mRNA from the approved mRNA vaccines gains access into the human cell and directs it to synthesize a copy of the spike protein found on the virus’s surface and produce antibodies against it. These antibodies become primed to inactivate the virus before it can cause the disease. Sometimes, however, a patient’s immune response can trigger the synthesis of antibodies against myelin, causing GBS [45].
A case of GBS was seen in the UK in a 62-year-old woman who had paraesthesias and weakness of the lower limbs 11 days after her initial dose of AZD1222 vaccine [46]. Another 82-year-old woman received her initial dose of the BNT162 vaccine 2 weeks before the diagnosis of GBS [45]. Approximately 17 cases of GBS develop per million people worldwide each year. With previous 1976 Swine flu and 2009 H1N1 vaccines, studies showed no increase in cases of GBS after vaccination [47]. To date, there is no substantial evidence that any of the COVID-19 vaccines cause GBS. Furthermore, no association was found between infection with COVID-19 and GBS. As a result, there is a low probability that GBS incidence will increase after COVID-19 vaccination [48]. COVID-19 poses a much greater risk of mortality and morbidity for adults than GBS does [49].
Acute transverse myelitis
Acute transverse myelitis is an uncommon neurologic condition affecting people aged 35 to 40 years at an incidence of 1.34 to 4.6 cases/million adults per year [50]. Of the reported adverse events after immunization recorded in the VAERS, 341 were neurologic events, 122 of which were cases of transverse myelitis [31]. Interleukin (IL)-17 and IL-6 appear to be involved in the pathogenesis of transverse myelitis. In myelitis, cerebrospinal fluid analysis findings show increased IL-6 levels [51]. By regulating cytokines, IL-17 stimulates astrocytes to produce IL-6, which forms nitric oxide metabolites and causes CNS damage [51].
Three cases of transverse myelitis were reported in the trial phase of the recombinant AZD1222 vaccine. Among these 3, 1 case had a background of multiple sclerosis; another was initially termed a potentially related case, but this was later ruled out by experts [17]. The presence of chimpanzee adenovirus antigen in AZD1222 may instigate immune responses targeting the spinal cord, which may in turn result in acute transverse myelitis [52]. COVID-19 - associated acute transverse myelitis should be investigated to identify the responsible antigen and explore immunopathogenesis.
Myocarditis and pericarditis
Myocarditis is an inflammation of the myocardial tissue without signs of ischemia and has various causes and diverse patterns [53]. In a study involving 7 patients with myocarditis between February 1 and April 30, 2021, 4 were diagnosed within 5 days after receiving COVID-19 vaccination. These 4 patients, who had received the second dose of an mRNA vaccine, reported chest pain and had increased biomarker levels suggestive of myocardial tissue injury. Cardiac magnetic resonance imaging results showed characteristics of myocarditis [54].
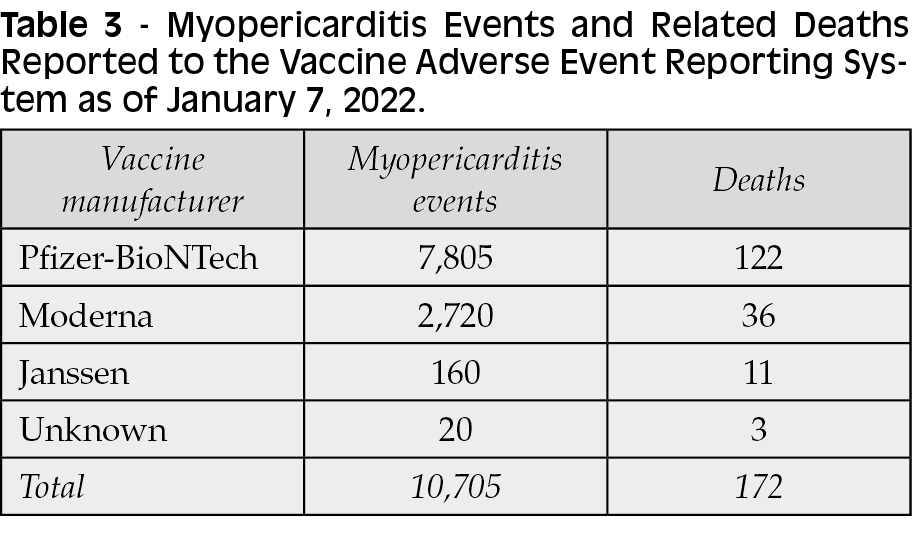
According to the CDC, more than 10,000 reports of myocarditis were reported to the VAERS after COVID-19 vaccination (Pfizer-BioNTech and Moderna) in the US (Table 3) [29]. These reports, however, are infrequent compared with the hundreds of millions of vaccine doses that were administered without adverse effects. The majority of the confirmed cases have been in teenagers and young adults 16 years or older and were often seen after receiving the second dose of the vaccine [55]. In a study of 200,287 persons, medical records from 40 hospitals in California, Montana, Los Angeles County, Oregon, and Washington were reviewed to identify cases of myocarditis and pericarditis after vaccination [56]. Myocarditis developed in 20 persons and pericarditis in 37. The incidence of myocarditis was highest among younger patients, generally after the second dose. However, older patients had development of pericarditis after the first or second dose [56]. In another study, vaccination for COVID-19 led to myocarditis in 23 male patients, 22 of whom were healthy members of the military [57]. In the majority of the patients, the diagnosis was made at least 4 days after the second dose of vaccination [57]. The clinical course and presentation suggest an association with vaccination-induced inflammation.
Cutaneous reactions
In a study from December 2020 to February 2021, 414 cutaneous symptoms were noted after administration of an mRNA vaccine [58]. Injection-site reactions, with delayed local reactions and urticarial and morbilliform eruptions, were the most commonly observed findings. Among recipients with first-dose reactions, 43% also had recurrences after their second dose [58]. Other reactions less commonly reported were pernio/chilblain, pityriasis rosea-like reactions, zoster, cosmetic filler reactions, and herpes simplex exacerbations. Some dermatologic symptoms, like pernio/chilblain, imitated COVID-19 symptoms. None of the patients reported serious adverse effects after receiving either of the doses [58]. As a result, researchers concluded that COVID-19 vaccination generally causes only mild and self-limiting reactions, and people should not be discouraged from the vaccination because of them [58].
Glomerular disease
Since mass-vaccination campaigns began in January 2021, the incidence of vaccine-associated glomerular disease has increased [59]. Symptoms of recurrent glomerular diseases or new glomerular diseases have appeared, especially after administration of the mRNA vaccines. The pathogenesis behind vaccine-associated glomerular disorders is not clearly understood. However, an immunogenic response to vaccines has been noted as a possible cause [60]. Minimal change disease, anti-glomerular basement membrane disease, membranous glomerular disease, and immunoglobulin A nephropathy are some of the glomerular lesions observed after vaccination [60]. Some case reports have described patients with gross hematuria after vaccination who were later found to have immunoglobulin A nephropathy. The majority of vaccine-related cases were typically seen within 1 to 3 weeks after vaccination [59]. Management of the glomerular disease must be on a case-by-case basis depending on the severity and remission status, because the benefits of vaccination outweigh the rare risk of glomerular disease.
CONCLUSIONS
COVID-19 is a global health concern that has spread worldwide [61] and has dramatically changed global sociopolitical, economic, and cultural aspects of humanity [62]. COVID-19 vaccines became more and more critical due to the limited prevention and treatment options available [63]. To end the pandemic crisis, the development of affordable, effective, safe, and transportable vaccines has become necessary. Some risks are associated with COVID-19 vaccinations, but no vaccination is entirely safe. Generally, short-term adverse effects of the COVID-19 vaccines present with mild symptoms. The most common symptoms are localized pain and swelling at the injection site, fever, headache, myalgia, and chills. Cases of thrombosis, notably CVT, are mostly seen with the adenoviral vector vaccines. Adverse effects such as myocarditis, glomerular diseases, and cutaneous eruptions are seen with the mRNA vaccines. The majority of vaccination reactions peak within the first 6 weeks after receiving the vaccine, but tracking over a longer time frame may provide insight into any future adverse reactions and rule out reactions that are falsely attributed to vaccinations. It is essential to identify the underlying immunologic and nonimmunologic mechanisms of adverse events so that appropriate policies are adopted, keeping safety in mind.
Acknowledgments
The Scientific Publications staff at Mayo Clinic provided editorial consultation, proofreading, and administrative and clerical support.
Conflict of interest
None to declare.
Funding
None to decleare.
REFERENCES
[1] World Health Organization. WHO coronavirus (COVID-19) dashboard. Geneva (Switzerland): World Health Organization. Available at: https://covid19.who.int. [Accessed 2021 Nov 15].
[2] Elbaum R, Smith A. UK approves Pfizer vaccine: first country to approve as rollout begins next week. Universal City (CA): NBC Universal; 2020. Available at: https://www.nbcnews.com/news/world/u-k-becomes-first-country-approve-pfizer-biontech-covid-19-n1249651. [Accessed 2021 Nov 15].
[3] World Health Organization. WHO issues its first emergency use validation for a COVID-19 vaccine and emphasizes need for equitable global access. Geneva (Switzerland): World Health Organization. Available at: https://www.who.int/news/item/31-12-2020-who-issues-its-first-emergency-use-validation-for-a-covid-19-vaccine-and-emphasizes-need-for-equitable-global-access. [Accessed 2021 Nov 15].
[4] Bloomberg. More than 7.49 billion shots given: COVID-19 tracker. New York (NY): Bloomberg; 2021. Available at: https://www.bloomberg.com/graphics/covid-vaccine-tracker-global-distribution/. [Accessed 2021 Nov 15].
[5] Ritchie H, Mathieu E, Rodes-Guiao L, et al. Coronavirus pandemic (COVID-19). United Kingdom: OurWorldInData.org; 2020. Available at: https://ourworldindata.org/coronavirus. [Accessed 2021 Nov 15].
[6] World Health Organization. COVID-19 vaccine tracker and landscape. Geneva (Switzerland): World Health Organization; 2021. Available at: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines. [Accessed 2021 Nov 15].
[7] Jarrett C, Wilson R, O’Leary M, Eckersberger E, Larson HJ, Sage Working Group on Vaccine Hesitancy. Strategies for addressing vaccine hesitancy: a systematic review. Vaccine. 2015; 33 (34), 4180-90.
[8] Koritala T, Hussain A, Pleshkova Y, et al. A narrative review of emergency use authorization versus full FDA approval and its effect on COVID-19 vaccination hesitancy. Infez Med. 2021; 3, 339-44.
[9] Kimmel SR. Vaccine adverse events: separating myth from reality. Am Fam Physician. 2002; 66 (11), 2113-20.
[10] Oliver SE, Gargano JW, Marin M, et al. The Advisory Committee on Immunization Practices’ Interim Recommendation for use of Pfizer-BioNTech COVID-19 vaccine: United States, December 2020. MMWR Morb Mortal Wkly Rep. 2020; 69 (50), 1922-4.
[11] US Food and Drug Administration. FDA takes key action in fight against COVID-19 by issuing emergency use authorization for first COVID-19 vaccine. Silver Spring (MD): US Food and Drug Administration; 2020. Available at: https://www.fda.gov/news-events/press-announcements/fda-takes-key-action-fight-against-covid-19-issuing-emergency-use-authorization-first-covid-19. [Accessed 2021 Nov 15].
[12] OurWorldInData. Coronavirus (COVID-19) vaccinations. United Kingdom: OurWorldInData; 2021. Available at: https://ourworldindata.org/covid-vaccinations. [Accessed 2021 Nov 15].
[13] World Health Organization. The Moderna COVID-19 (mRNA-1273) vaccine: what you need to know. World Health Organization: Geneva (Switzerland); 2021. Available at: https://www.who.int/news-room/feature-stories/detail/the-moderna-covid-19-mrna-1273-vaccine-what-you-need-to-know?gclid=Cj0KCQjw8vqGBhC_ARIsADMSd1BoyQ_zbHko4wHLbCqKhIlf9l62R-gphelomU8-Z2W7XDRLhg5FNqEaAkzwEALw_wcB. [Accessed 2021 Nov 15].
[14] US Food and Drug Administration. COVID-19 Vaccines: the FDA has regulatory processes in place to facilitate the development of COVID-19 vaccines that meet the FDA’s rigorous scientific standards. Silver Spring (MD): US Food and Drug Administration. Available at: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/covid-19-vaccines. [Accessed 2021 Nov 15].
[15] Baden LR, El Sahly HM, Essink B, et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2021; 384 (5), 403-16.
[16] Cirillo N. Reported orofacial adverse effects of COVID-19 vaccines: the knowns and the unknowns. J Oral Pathol Med. 2021; 50 (4), 424-7.
[17] Voysey M, Clemens SAC, Madhi SA, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021; 397(10269), 99-111.
[18] Greinacher A, Thiele T, Warkentin TE, Weisser K, Kyrle PA, Eichinger S. Thrombotic thrombocytopenia after ChAdOx1 nCov-19 vaccination. N Engl J Med. 2021; 384 (22), 2092-101.
[19] European Medicines Agency. Dashboards. Amsterdame (The Netherlands): European Medicines Agency. Available at: https://dap.ema.europa.eu/analytics/saw.dll?PortalPages [Accessed 2022 Jan 27].
[20] Shay DK, Gee J, Su JR, et al. Safety monitoring of the Janssen (Johnson & Johnson) COVID-19 vaccine: United States, March-April 2021. MMWR Morb Mortal Wkly Rep. 2021; 70 (18), 680-4.
[21] US Food and Drug Administration. FDA and CDC lift recommended pause on Johnson & Johnson (Janssen) COVID-19 vaccine use following thorough safety review. Silver Spring (MD): US Food and Drug Administration; 2021. Available at: https://www.fda.gov/news-events/press-announcements/fda-and-cdc-lift-recommended-pause-johnson-johnson-janssen-covid-19-vaccine-use-following-thorough. [Accessed 2021 Nov 15].
[22] Lawton G. Sputnik V vaccine goes global. New Sci. 2021; 250 (3331), 10-1.
[23] King A. Vector-based vaccines come to the fore in the COVID-19 pandemic Wilmington (DE): Group Publishing; 2020. Available at: https://www.the-scientist.com/news-opinion/vector-based-vaccines-come-to-the-fore-in-the-covid-19-pandemic-67915. [Accessed 2021 Nov 15].
[24] Sadoff J, Le Gars M, Shukarev G, et al. Interim Results of a Phase 1-2a Trial of Ad26.COV2.S Covid-19 Vaccine. N Engl J Med. 2021; 384 (19), 1824-35.
[25] World Health Organization. WHO director-general’s opening remarks at the media briefing on COVID-19, 23 October 2020. Geneva (Switzerland): World Health Organization; 2020. Available at: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---23-october-2020. [Accessed 2021 Nov 15].
[26] Logunov DY, Dolzhikova IV, Shcheblyakov DV, et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. Lancet. 2021; 397 (10275), 671-81.
[27] Cohen C. Russia’s claim of a successful COVID-19 vaccine doesn’t pass the ‘smell test,’ critics say. Washington (DC): American Association for the Advancement of Science; 2020. Available at: https://www.sciencemag.org/news/2020/11/russia-s-claim-successful-covid-19-vaccine-doesn-t-pass-smell-test-critics-say. [Accessed 2021 Nov 15].
[28] Shimabukuro TT, Nguyen M, Martin D, DeStefano F. Safety monitoring in the Vaccine Adverse Event Reporting System (VAERS). Vaccine. 2015; 33 (36), 4398-405.
[29] Centers for Disease Control and Prevention, US Department of Health & Human Services. The Vaccine Adverse Event Reporting System (VAERS). Atlanta (GA): Centers for Disease Control and Prevention, US Department of Health & Human Services. Available at: http://wonder.cdc.gov/vaers.html. [Accessed 2021 Nov 15].
[30] Goss AL, Samudralwar RD, Das RR, Nath A. ANA investigates: neurological complications of COVID-19 vaccines. Ann Neurol. 2021; 89 (5), 856-7.
[31] Centers for Disease Control and Prevention, US Department of Health & Human Services. Vaccine adverse event reporting system. Atlanta (GA): Centers for Disease Control and Prevention, US Department of Health & Human Services. Available at: https://vaers.hhs.gov/ [Accessed 2021 Nov 15].
[32] Pottegard A, Lund LC, Karlstad O, et al. Arterial events, venous thromboembolism, thrombocytopenia, and bleeding after vaccination with Oxford-AstraZeneca ChAdOx1-S in Denmark and Norway: population based cohort study. BMJ. 2021; 373, n1114.
[33] Wise J. Covid-19: European countries suspend use of Oxford-AstraZeneca vaccine after reports of blood clots. BMJ. 2021; 372, n699.
[34] Government of Canada, National Advisory Committee on Immunization. Archived 8: NACI rapid response: recommended use of AstraZeneca COVID-19 vaccine in younger adults [2021-03-29]. Ottawa (ON): Government of Canada, National Advisory Committee on Immunization; 2021. Available at: https://www.canada.ca/en/public-health/services/immunization/national-advisory-committee-on-immunization-naci/rapid-response-recommended-use-astrazeneca-covid-19-vaccine-younger-adults.html. [Accessed 2021 Nov 15].
[35] Dyer O. COVID-19: EMA defends AstraZeneca vaccine as Germany and Canada halt rollouts. BMJ. 2021; 373, n883.
[36] Chapin-Bardales J, Gee J, Myers T. Reactogenicity following receipt of mRNA-based COVID-19 vaccines. JAMA. 2021; 325 (21), 2201-2.
[37] Ulivi L, Squitieri M, Cohen H, Cowley P, Werring DJ. Cerebral venous thrombosis: a practical guide. Pract Neurol. 2020; 20(5), 356-67.
[38] Ferro JM, Canhao P, Stam J, Bousser MG, Barinagarrementeria F, ISCVT Investigators. Prognosis of cerebral vein and dural sinus thrombosis: results of the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT). Stroke. 2004; 35 (3), 664-70.
[39] Scully M, Singh D, Lown R, et al. Pathologic antibodies to platelet factor 4 after ChAdOx1 nCoV-19 vaccination. N Engl J Med. 2021; 384 (23), 2202-11.
[40] Merchant H. Might post-injection distribution of CoViD vaccines to the brain explain the rare fatal events of cerebral venous sinus thrombosis (CVST)? [Letter]. BMJ2021. Available at: https://www.bmj.com/content/373/bmj.n958/rr-1. [Accessed 2021 Nov 15].
[41] Hinduja A, Nalleballe K, Onteddu S, Kovvuru S, Hussein O. Impact of cerebral venous sinus thrombosis associated with COVID-19. J Neurol Sci. 2021; 425, 117448.
[42] MacNeil JR, Su JR, Broder KR, et al. Updated recommendations from the Advisory Committee on Immunization Practices for Use of the Janssen (Johnson & Johnson) COVID-19 vaccine after reports of thrombosis with thrombocytopenia syndrome among vaccine recipients: United States, April 2021. MMWR Morb Mortal Wkly Rep. 2021; 70 (17), 651-6.
[43] Centers for Disease Control and Prevention, US Department of Health & Human Services. CDC recommends use of Johnson & Johnson’s Janssen COVID-19 vaccine resume. Atlanta (GA): Centers for Disease Control and Prevention, US Department of Health & Human Services; 2021. Available at: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/JJUpdate.html. [Accessed 2021 Nov 15].
[44] Haber P, Sejvar J, Mikaeloff Y, DeStefano F. Vaccines and Guillain-Barre syndrome. Drug Saf. 2009; 32 (4), 309-23.
[45] Waheed S, Bayas A, Hindi F, Rizvi Z, Espinosa PS. Neurological complications of COVID-19: Guillain-Barre syndrome following Pfizer COVID-19 vaccine. Cureus. 2021; 13 (2), e13426.
[4] Hasan T, Khan M, Khan F, Hamza G. Case of Guillain-Barre syndrome following COVID-19 vaccine. BMJ Case Rep. 2021; 14(6).
[47] Sejvar JJ, Pfeifer D, Schonberger LB. Guillain-barre syndrome following influenza vaccination: causal or coincidental? Curr Infect Dis Rep. 2011; 13 (4), 387-98.
[48] Lunn MP, Cornblath DR, Jacobs BC, et al. COVID-19 vaccine and Guillain-Barre syndrome: let’s not leap to associations. Brain. 2021; 144 (2), 357-60.
[49] The GBS/CIDP Foundation International. COVID-19 vaccines and the GBS|CIDP community. Conshohocken (PA): GBS/CIDP Foundation International; 2021. Available at: https://www.gbs-cidp.org/covid-19-vaccines-and-the-gbscidp-community/. [Accessed 2021 Nov 15].
[50] Roman GC, Gracia F, Torres A, Palacios A, Gracia K, Harris D. Acute Transverse Myelitis (ATM): clinical review of 43 patients with COVID-19-associated ATM and 3 post-vaccination ATM serious adverse events with the ChAdOx1 nCoV-19 vaccine (AZD1222). Front Immunol. 2021; 12, 653786.
[51] Graber JJ, Allie SR, Mullen KM, et al. Interleukin-17 in transverse myelitis and multiple sclerosis. J Neuroimmunol. 2008; 196 (1-2), 124-32.
[52] Ramasamy MN, Minassian AM, Ewer KJ, et al. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults (COV002): a single-blind, randomised, controlled, phase 2/3 trial. Lancet. 2021; 396 (10267), 1979-93.
[53] Tschope C, Ammirati E, Bozkurt B, et al. Myocarditis and inflammatory cardiomyopathy: current evidence and future directions. Nat Rev Cardiol. 2021; 18 (3), 169-93.
[54] Kim HW, Jenista ER, Wendell DC, et al. Patients with acute myocarditis following mRNA COVID-19 vaccination. JAMA Cardiol. 2021; 6 (10), 1196-201.
[55 Centers for Disease Control and Prevention, US Department of Health & Human Services. Myocarditis and pericarditis after mRNA COVID-19 vaccination. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health & Human Services; 2021. Available at: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/myocarditis.html. [Accessed 2021 Nov 15].
[56] Diaz GA, Parsons GT, Gering SK, Meier AR, Hutchinson IV, Robicsek A. Myocarditis and pericarditis after vaccination for COVID-19. JAMA. 2021; 326 (12), 1210-2.
[57] Montgomery J, Ryan M, Engler R, et al. Myocarditis following immunization with mRNA COVID-19 vaccines in members of the US military. JAMA Cardiol. 2021; 6 (10), 1202-6.
[58] McMahon DE, Amerson E, Rosenbach M, et al. Cutaneous reactions reported after Moderna and Pfizer COVID-19 vaccination: a registry-based study of 414 cases. J Am Acad Dermatol. 2021; 85 (1), 46-55.
[59] Bomback AS, Kudose S, D’Agati VD. De novo and relapsing glomerular diseases after COVID-19 vaccination: what do we know so far? Am J Kidney Dis. 2021; 78 (4), 477-80.
[60] Gutierrez S, Dotto B, Petiti JP, et al. Minimal change disease following influenza vaccination and acute renal failure: just a coincidence? Nefrologia. 2012; 32 (3), 414-5.
[61] worldometer. COVID-19 coronavirus pandemic. worldometer; 2021. Available at: https://www.worldometers.info/coronavirus/#countries. [Accessed 2021 Nov 15].
[62] Sun J, He WT, Wang L, et al. COVID-19: epidemiology, evolution, and cross-disciplinary perspectives. Trends Mol Med. 2020; 26 (5), 483-95.
[63] Provenzani A, Polidori P. Covid-19 and drug therapy, what we learned. Int J Clin Pharm. 2020; 42 (3), 833-6.